Acetaminophen, Diphenhydramine Hydrochloride
Acetaminophen, Diphenhydramine HCl Tablets
FULL PRESCRIBING INFORMATION: CONTENTS*
- Acetaminophen, Diphenhydramine Hydrochloride Uses
- Warnings
- Directions
- Acetaminophen, Diphenhydramine Hydrochloride Other information
- Inactive ingredients
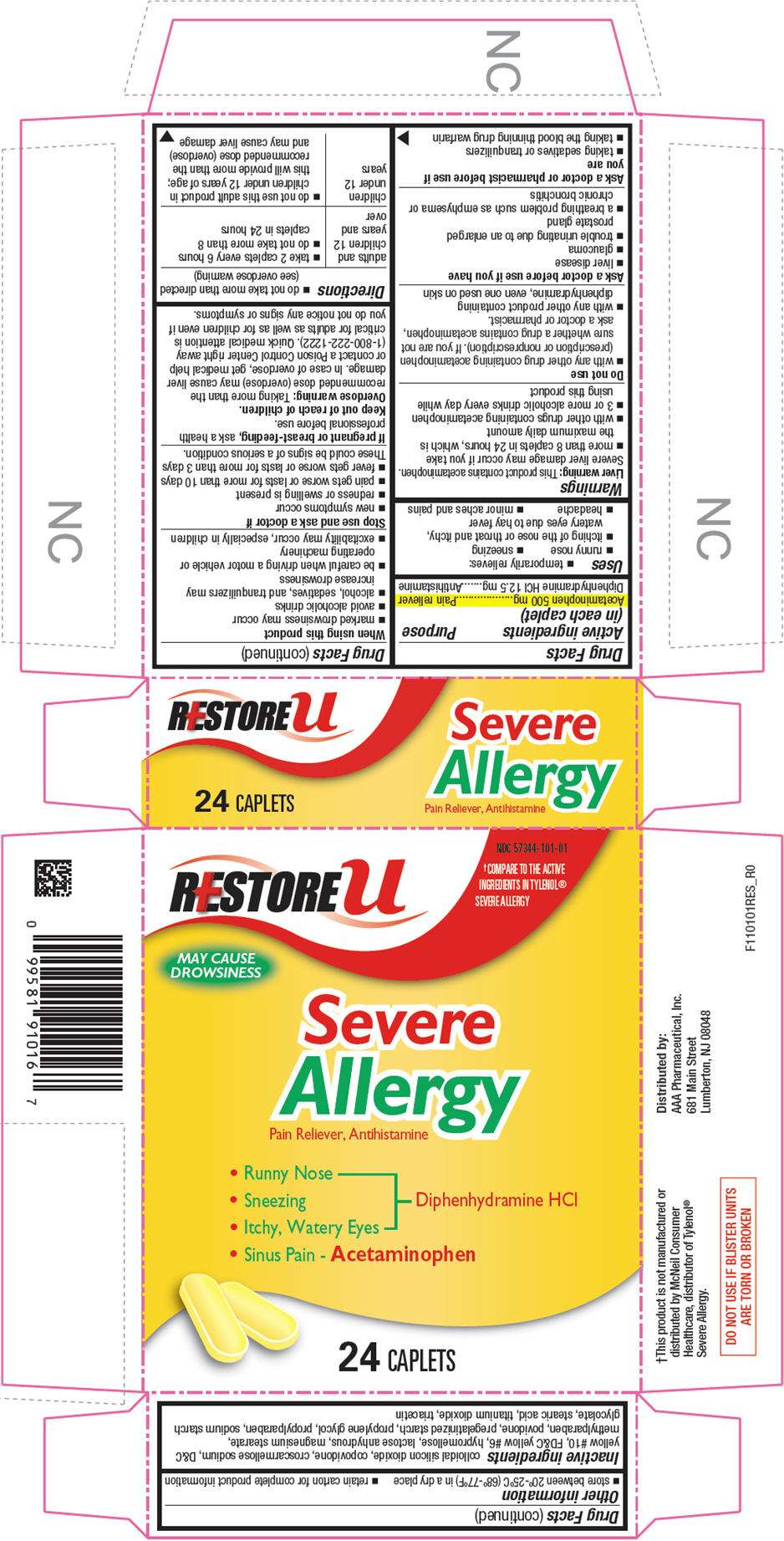
- PRINCIPAL DISPLAY PANEL - 24 Caplet Blister Pack Carton
FULL PRESCRIBING INFORMATION
Drug Facts
Active ingredient
Purpose
|
Active ingredients (in each caplet) |
Purpose |
|---|---|
| Acetaminophen 500 mg | Pain reliever |
| Diphenhydramine HCl 12.5 mg | Antihistamine |
Acetaminophen, Diphenhydramine Hydrochloride Uses
- temporarily relieves:
- runny nose
- sneezing
- itching of the nose or throat and itchy, watery eyes due to hay fever
- headache
- minor aches and pains
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if you take
- more than 8 caplets in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- liver disease
- glaucoma
- trouble urinating due to an enlarged prostate gland
- a breathing problem such as emphysema or chronic bronchitis
Ask a doctor or pharmacist before use if you are
- taking sedatives or tranquilizers
- taking the blood thinning drug warfarin
When using this product
- marked drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Stop use and ask a doctor if
- new symptoms occur
- redness or swelling is present
- pain gets worse or lasts for more than 10 days
- fever gets worse or lasts for more than 3 days
These could be signs of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children.
Overdose warning
Taking more than the recommended dose (overdose) may cause liver damage. In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Directions
- do not take more than directed (see overdose warning)
| adults and children 12 years and over |
|
| children under 12 years |
|
Acetaminophen, Diphenhydramine Hydrochloride Other information
- store between 20º-25ºC (68º-77ºF) in a dry place
- retain carton for complete product information
Inactive ingredients
colloidal silicon dioxide, copovidone, croscarmellose sodium, D&C yellow #10, FD&C yellow #6, hypromellose, lactose anhydrous, magnesium stearate, methylparaben, povidone, pregelatinized starch, propylene glycol, propylparaben, sodium starch glycolate, stearic acid, titanium dioxide, triacetin
Distributed by:
AAA Pharmaceutical, Inc.
681 Main Street
Lumberton, NJ 08048
PRINCIPAL DISPLAY PANEL - 24 Caplet Blister Pack Carton
NDC 57344-101-01
RESTORE u
†COMPARE TO THE ACTIVE
INGREDIENTS IN TYLENOL®
SEVERE ALLERGY
MAY CAUSE
DROWSINESS
Severe
Allergy
Pain Reliever, Antihistamine
- Runny Nose
- Sneezing
- Itchy, Watery Eyes
Diphenhydramine HCl - Sinus Pain - Acetaminophen
24 CAPLETS
Acetaminophen, Diphenhydramine HydrochlorideACETAMINOPHEN and DIPHENHYDRAMINE HYDROCHLORIDE TABLET, COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||