ACYCLOVIR
Acyclovir Capsules
FULL PRESCRIBING INFORMATION: CONTENTS*
- ACYCLOVIR DESCRIPTION
- VIROLOGY
- CLINICAL PHARMACOLOGY
- CLINICAL TRIALS
- ACYCLOVIR INDICATIONS AND USAGE
- ACYCLOVIR CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- ACYCLOVIR ADVERSE REACTIONS
- OVERDOSAGE
- ACYCLOVIR DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
FULL PRESCRIBING INFORMATION
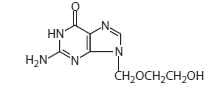
ACYCLOVIR DESCRIPTION
VIROLOGY
Mechanism of Antiviral Action: Acyclovir is a synthetic purine nucleoside analogue with in vitro in in vivo inhibitory activity against herpes simples virus types 1 (HSV-1), 2 (HSV-2), and varicella-zoster virus (VZV). The inihibitory activity of acyclovir is highly selective due to its affinity for the enzyme thymidine kinase (TK) encoded by HSV and VZV. This viral enzyme converts acyclovir into acyclovir monophosphate, a nucleoside analogue. The monophosphate is further converted into diphosphate by cellular guanylate kinase and into triphosphate by a number of cellular enzymes. In vitro, acyclovir triphosphate stops replication of herpes viral DNA. This is accomplished in 3 ways: 1) competitive inhibition of viral DNA polymerase, 2) incorporation into and termination of growing viral DNA chain, and 3) inactivation of the viral DNA polymerase. The greater antiviral activity of acyclovir against HSV compared to VZV is due to its more efficient phosphorylation by the viral TK.
Antiviral Activiti es: The quantitative relationship between the in vitro susceptibility of herpes viruses to antivirals and the clinical response to therapy has not been established in humans, and virus sensitivity testing has not been standardized. Sensitivity testing results, expressed as the concentration of drug required to inhibit by 50% the growth of virus in cell culture (IC50), vary greatly depending upon a number of factors. Using plaque-reduction assays, the IC50 against herpes simplex virus isolates ranges from 0.02 to 13.5 mcg/mL for HSV-1 and from 0.01 to 9.9 mcg/mL for HSV-2. The IC50 for acyclovir against most laboratory strains and clinical isolates of VZV ranges from 0.12 to 10.8 mcg/mL. Acyclovir also demonstrates activity against the Oka vaccine strain of VZV with a mean IC50 of 1.35mcg/mL.
Drug Resistance: Resistance of HSV and VZV to acyclovir can result from qualitative and quantitative changes in the viral TK and/or DNA polymerase. Clinical isolates of HSV and VZV with reduced susceptibility to acyclovir have been recovered from immunocompromised patients, especially with advanced HIV infection . While most of the acyclovir-resistant mutants isolated thus far from immunocompromised patients have been found to be TK-deficient mutants, other mutants involving the viral TK gene (Tk partial and Tk altered) and DNA polymerase have been isolated. TK-negative mutants may cause severe disease in infants and immunocompromised adults. The possibility of viral resistance to acyclovir should be considered in patients who show poor clinical response during therapy.
CLINICAL PHARMACOLOGY
Pharmacokinetics:
Table 1. Acyclovir Pharmacokinetic Characteristics (Range)
----------------------------------------------------------------------------------------------------------------------------------------------------------------------
Parameter Range
Plasma protein binding 9% to 33%
Plasma elimination half-life 2.5 to 3.3 hr
Average oral bioavailability 10% to 20%*
*Bioavailability decreases with increasing dose
----------------------------------------------------------------------------------------------------------------------------------------------------------------------
In one multiple-dose, crossover study in healthy subjects (n=23), it was shown that increases in plasma acyclovir concentrations were less than those proportional with increasing dose, as shown in Table 2. The decrease in bioavailability is a function of the dose and not the dosage form.
Table 2. Acyclovir Peak and Trough Concentrations at Steady State
---------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Parameter 200 mg 400 mg 800 mg
Css max 0.83 mcg/ml 1.21 mcg/ml 1.61 mcg/ml
Css trough 0.46 mcg/ml 0.63 mcg/ml 0.83 mcg/ml
---------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
There was no effect of food on the absorption of acyclovir (n=6); therefore, acyclovir capsules may be administered with or without food. The only known urinary metabolite is 9-[(carboxymethoxy)methyl]guanine.
Special Populations: Adults With Impaired Renal Function: The half-life and total body clearance of acyclovir are dependent on renal function. A dosage adjustment is recommended for patients with reduced renal function (see DOSAGE AND ADMINISTRATION).
Pediatrics:
Drug Interactions:
CLINICAL TRIALS
Initial Genital Herpes: Double-blind, placebo-controlled studies have demonstrated that orally administered acyclovir significantly reduced the duration of acute infection and duration of lesion healing. The duration of pain and new lesion formation was decreased in some patient groups.
Recurrent Genital Herpes: Double-blind, placebo-controlled studies in patients with frequent recurrences (6 or more episodes per year) have shown that orally administered acyclovir given daily for 4 months to 10 years prevented or reduced the frequency and/or severity of recurrences in greater than 95% of patients. In a study of patients who received acyclovir 400mg twice daily for 3 years, 45%, 52%, and 63% of patients remained free of recurrences in the first, second, and third years, respectively. Serial analyses of the 3-month recurrence rates for the patients showed that 71% to 87% were recurrence free in each quarter.
Herpes Zoster Infections: In a double-blind, placebo-controlled study of immunocompetent patients with localized cutaneous zoster infection, acyclovir (800mg 5 times daily for 10 days) shortened the times to lesion scabbing, healing, and complete cessation of pain, and reduced the duration of viral shedding and the duration of new lesion formation. In a similar double-blind, placebo-controlled study, acyclovir (800mg 5 times daily for 7 days) shortened the times to complete lesion scabbing, healing, and cessation of pain; reduced the duration of new lesion formation; and reduced the prevalence of localized zoster-associated neurologic symptoms (paresthesia, dysesthesia, or hyperesthesia). Treatment was begun within 72 hours of rash onset and was most effective if started within the first 48 hours. Adults greater than 50 years of age showed greater benefit.
Chickenpox: Three randomized, double-blind, placebo-controlled trials were conducted in 993 pediatric patients aged 2 to 18 years with chickenpox. All patients were treated within 24 hours after the onset of rash. In 2 trials, acyclovir was administered at 20mg/kg 4 times daily (up to 3,200 mg per day) for 5 days. In the third trial, doses of 10, 15, or 20 mg/kg were administered 4 times daily for 5 to 7 days. Treatment acyclovir shortened the time to 50% healing; reduced the maximum number of lesions; reduced the median number of vesicles; decreased the median number of residual lesions on day 28; and decreased the proportion of patients with fever, anorexia, and lethargy by day 2. Treatment with acyclovir did not affect varicella-zoster virus-specific humoral or cellular immune responses at 1 month or 1 year following treatment.
ACYCLOVIR INDICATIONS AND USAGE
Herpes Zoster Infections: acyclovir is indicated for the acute treatment of herpes zoster (shingles).
Genital Herpes: acyclovir is indicated for the treatment of initial episodes and the management of recurrent episodes of genital herpes.
Chickenpox: acyclovir is indicated for the treatment of chickenpox (varicella).
ACYCLOVIR CONTRAINDICATIONS
Acyclovir is contraindicated for patients who develop hypersensitivity to acyclovir or valacyclovir.
WARNINGS
Acyclovir capsules are intended for oral ingestion only. Renal failure, in some cases resulting in death, has been observed with acyclovir therapy (see (ADVERSE REACTIONS: Observed During Clinical Practice and OVERDOSAGE). Thrombotic thrombocytopenic purpura/hemolytic uremic syndrome (TTP/HUS), which has resulted in death, has occurred in immunocompromised patients receiving acyclovir therapy.
PRECAUTIONS
Information for Patients:
Herpes Zoster:
Genital Herpes Infections:
Chickenpox:
Drug Interactions:
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Pregnancy:
Nursing Mothers:
Pediatrics:
Geriatric Use:>
ACYCLOVIR ADVERSE REACTIONS
Herpes Simplex: Short-Term Administration:Long-Term Administration:
Herpes Zoster:
Chickenpox:
Observed During Clinical Practice
General:
Nervous:
Digestive:
Hematologic and Lymphatic:
Hepatobiliary Tract and Pancreas:
Musculoskeletal:
Skin:
Special Senses:
Urogenital:
OVERDOSAGE
Overdoses involving ingestion of up to 100 capsules (20g) have been reported. Adverse events that have been reported in association with overdosage include agitation, coma, seizures, and lethargy. Precipitation of acyclovir in renal tubules may occur when the solubility (2.5mg/mL) is exceeded in the intratubular fluid. Overdosage has been reported following bolus injections or inappropriately high doses and in patients whose fluid and electrolyte balance were not properly monitored. This has resulted in elevated BUN and serum creatinine and subsequent renal failure. In the event of acute renal failure and anuria, the patient may benefit from hemodialysis until renal function is restored (see DOSAGE AND ADMINISTRATION).
ACYCLOVIR DOSAGE AND ADMINISTRATION
Acute Treatment of Herpes Zoster:Genital Herpes: Treatment of Initial Genital Herpes: 200 mg every 4 hours, 5 times daily for 10 days.
Chronic Suppressive Therapy for Recurrent Disease: 400 mg 2 times daily for up to 12 months, followed by re-evaluation. Alternative regimens have included doses ranging from 200 mg 3 times daily to 200 mg 5 times daily. The frequency and severity of episodes of untreated genital herpes may change over time. After 1 year of therapy, the frequency and severity of the patient's genital herpes infection should be re-evaluated to assesst he need for continuation of therapy with acyclovir.
Intermittent Therapy: 200 mg every 4 hours, 5 times daily for 5 days. Therapy should be initiated at the earliest sign or symptom (prodrome) of recurrence.
Treatment of Chickenpox: Children (2 years of age and older): 20 mg/kg per dose orally 4 times daily (80 mg/kg/day) for 5 days. Children over 40 kg should receive the adult dose for chickenpox.
Adults and Children over 40 kg: 800 mg 4 times daily for 5 days. Intravenous acyclovir is indicated for the treatment of varicella-zoster infections in immunocompromised patients. When therapy is indicated, it should be initiated at the earliest sign or symptom of chickenpox. There is no information about the efficacy of therapy initiated more than 24 hours after onset of signs and symptoms. Patients with Acute or Chronic Renal Impairment: In patients with renal impairment, the dose of acyclovir capsules should be modified as shown in Table 3.
Table 3. Dosage Modifications for Renal Impairment
------------------------------------------------------------------------------------------------------------------------------------------------------------
Normal Dosage Creatinine Clearance Adjusted Dosage Dosing Interval
Regimen (mL/min/1.73m2) Regimen Dose (mg)
200 mg every 4 hours >10 200 Every 4 hours, 5x daily
0-10 200 Every 12 hours
400 mg every 12 hours >10 400 Every 12 hours
0-10 200 Every 12 hours
>25 800 Every 4 hours, 5x daily
800 mg every 4 hours 10-25 800 Every 8 hours
0-10 800 Every 12 hours
-----------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Hemodialysis: For patients who require hemodialysis, the mean plasma half-life of acyclovir during hemodialysis is approximately 5 hours. This results in a 60% decrease in plasma concentrations following a 6-hour dialysis period. Therefore, the patient's dosing schedule should be adjusted so that an additional dose is administered after each dialysis.
Peritoneal Dialysis: No supplemental dose appears to be necessary after adjustment of the dosing interval.
Bioequivalence of Dosage Forms: acyclovir Suspension was shown to be bioequivalent to acyclovir Capsules (n=20) and 1 acyclovir 800-mg capsule was shown to be bioequivalent to 4 acyclovir 200-mg capsules (n=24).
HOW SUPPLIED
Acyclovir Capsules USP, 200 mg Oval capsule, light blue opaque cap and aqua blue opaque body imprinted with "HP146" are supplied in bottles of 100s (NDC 62033-0204-0) and 400s (NDC 62033-0204-4).
Store at 150 to 250C (590 to 770F) and protect from moisture. Call you doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Manufactured for:
BOSCOGEN, INC.
11 Morgan
Irvine, CA 92618
Manufactured by:
Stason Pharmaceuticals, Inc.
Irvine, CA 92618

ACYCLOVIRAcyclovir CAPSULE
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||