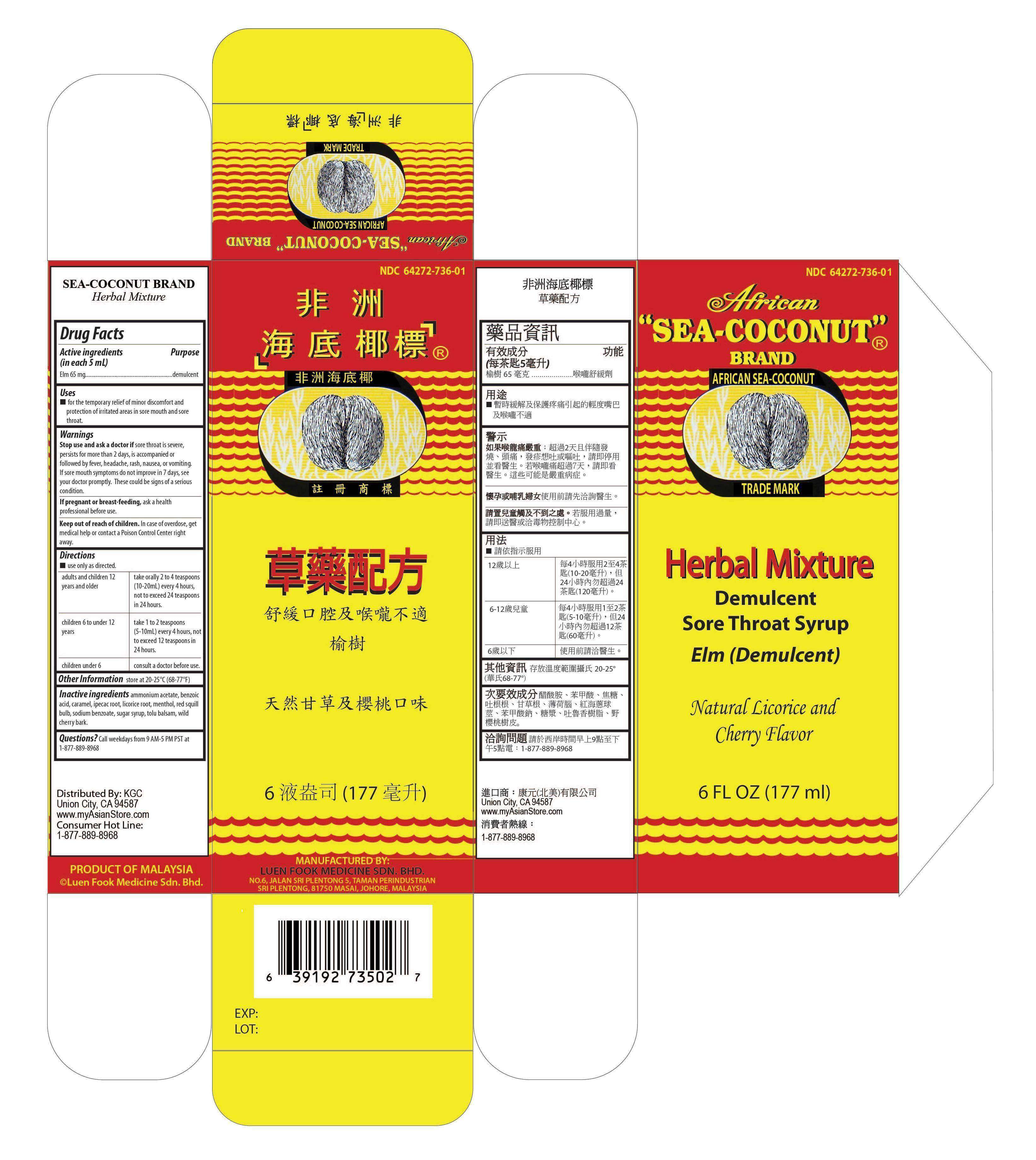
AFRICAN SEA-COCONUT
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredients (in each 5 mL tsp)
- Purpose
- Use
- Directions
- Other Information
- Inactive Ingredients
- Questions?
FULL PRESCRIBING INFORMATION
Active ingredients (in each 5 mL tsp)
Elm 65 mg
Purpose
Demulcent
Use
for the temporary relief of minor discomfort and protection of irritated areas in sore mouth and sore throat.
Warnings
Stop use and ask a doctor if sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting. If sore mouth symptoms do not improve in 7 days, see your doctor promptly. These could be signs of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- use only as directed
|
adults and children 12 years and
older |
take orally 2 to 4 teaspoons (10-20mL) every 4 hours, not to exceed 24 teaspoons in 24 hours. |
| children 6 to under 12 years | take 1 to 2 teaspoons (5-10mL) every 4 hours, not to exceed 12 teaspoons in 24 hours. |
| children under 6 years | consult a doctor before use. |
Other Information
store at 20-25∘C (68-77∘F)
Inactive Ingredients
ammonium acetate, benzoic acid, caramel, ipecac, licorice, menthol, drimia maritima bulb, sodium benzoate, sucrose, tolu balsam, prunus serotina bark.
Questions?
Call weekdays from 9 AM-5 PM PST at 1-877-889-8968.
MM2
AFRICAN SEA-COCONUTElm SYRUP
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||