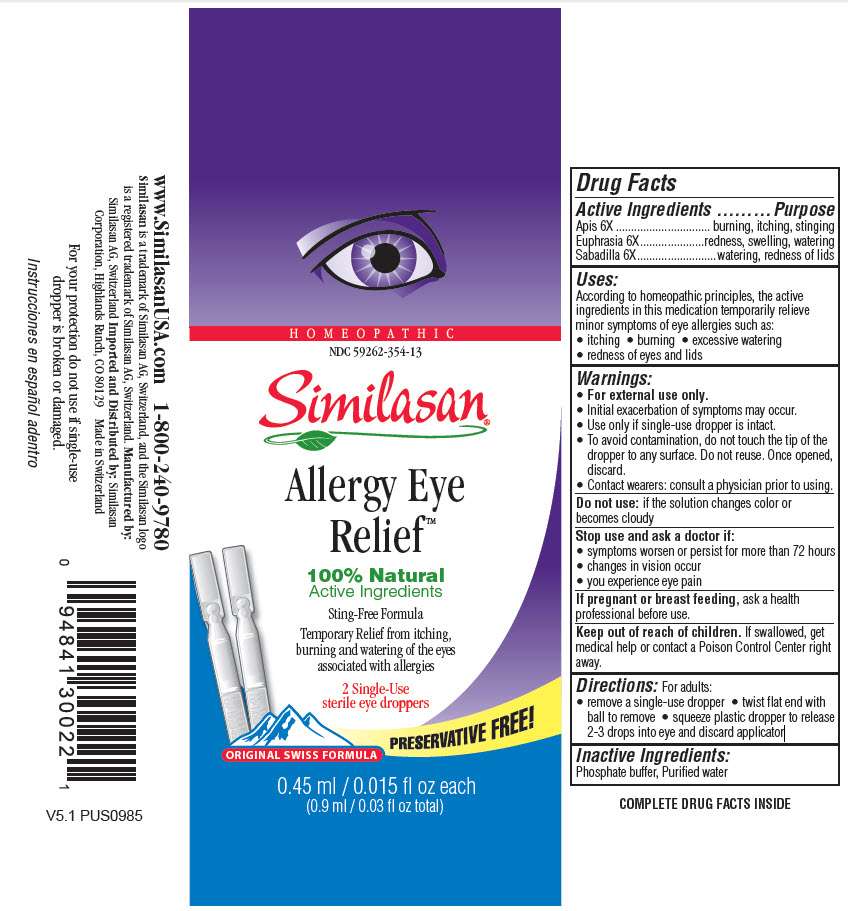
Allergy Eye Relief
Similasan Allergy Eye Relief 10 ml
FULL PRESCRIBING INFORMATION
Apis 6X
burning, itching, stinging
Euphrasia 6X
redness, swelling, watering
Sabadilla 6X
watering, redness of lids
According to homeopathic principles, the active ingredients in this medication temporarily relieve minor symptoms of eye allergies such as:
- itching
- burning
- excessive watering
- redness of eyes and lids
- For external use only.
- Initial exacerbation of symptoms may occur.
- Use only if single-use dropper is intact.
- To avoid contamination, do not touch the tip of the dropper to any surface. Do not reuse. Once opened, discard.
- Contact wearers: consult a physician prior to using.
if solution changes color or becomes cloudy
- symptoms worsen or persist for more than 72 hours
- changes in vision occur
- you experience eye pain
If pregnant or breastfeeding,
ask a health professional before use.
If swallowed, get medicl help or contact a Poison Control Center right away.
For adults:
- remove a single-use dropper
- twist flat end with ball to remove
- squeeze plastic dropper to release 2-3 drops into eye and discard applicator
Phosphate buffer, Purified water
Homeopathic
NDC 59262-354-13
Similasan Allergy Eye
Relief
2 Single-Use
sterile eye droppers
SIMILASAN / USA
30022
Allergy Eye Relief, eye drops,
2 x 0.45 ml, 2 Single-Use
Droppers, Trial size buffered
270 pieces (Stk.)
Allergy Eye ReliefApis and Euphrasia and Sabadilla SOLUTION/ DROPS
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!