ASMANEX HFA
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use ASMANEX HFA safely and effectively. See full prescribing information for ASMANEX HFA. ASMANEX® HFA 100 mcg (mometasone furoate) Inhalation Aerosol ASMANEX® HFA 200 mcg (mometasone furoate) Inhalation Aerosol FOR ORAL INHALATION Initial U.S. Approval: 1987INDICATIONS AND USAGEASMANEX HFA is a corticosteroid indicated for: Maintenance treatment of asthma as prophylactic therapy in patients 12 years of age and older. (1.1) Important limitations: Not indicated for the relief of acute bronchospasm. (1.1) DOSAGE AND ADMINISTRATIONFor oral inhalation only. (2.1) Treatment of asthma in patients 12 years of age and older: 2 inhalations twice daily of ASMANEX HFA 100 mcg or 200 mcg. (2.2) Starting dosage is based on prior asthma therapy. (2.2) DOSAGE FORMS AND STRENGTHS Inhalation aerosol containing 100 mcg or 200 mcg of mometasone furoate per actuation. (3) CONTRAINDICATIONS Primary treatment of status asthmaticus or acute episodes of asthma requiring intensive measures. (4.1) Hypersensitivity to any of the ingredients of ASMANEX HFA. (4.2) WARNINGS AND PRECAUTIONS Deterioration of asthma and acute episodes: ASMANEX HFA should not be used for relief of acute symptoms. Patients require immediate re-evaluation during rapidly deteriorating asthma. (5.1) Localized infections: Candida albicans infection of the mouth and throat may occur. Monitor patients periodically for signs of adverse effects on the oral cavity. Advise patients to rinse the mouth following inhalation. (5.2) Immunosuppression: Potential worsening of existing tuberculosis, fungal, bacterial, viral, or parasitic infection; or ocular herpes simplex infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. Use with caution in patients with these infections because of the potential for worsening of these infections. (5.3) Transferring patients from systemic corticosteroids: Risk of impaired adrenal function when transferring from oral steroids. Wean patients slowly from systemic corticosteroids if transferring to ASMANEX HFA. (5.4) Hypercorticism and adrenal suppression: May occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue ASMANEX HFA slowly. (5.5) Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir): Risk of increased systemic corticosteroid effects. Exercise caution when used with ASMANEX HFA. (5.6) Paradoxical bronchospasm: Discontinue ASMANEX HFA and institute alternative therapy if paradoxical bronchospasm occurs. (5.7) Hypersensitivity reactions including anaphylaxis: Hypersensitivity reactions, such as urticaria, flushing, allergic dermatitis, bronchospasm, rash, pruritus, angioedema, and anaphylactic reaction may occur. Discontinue ASMANEX HFA if such reactions occur. (5.8) Decreases in bone mineral density: Monitor patients with major risk factors for decreased bone mineral content. (5.9) Effects on growth: Monitor growth of pediatric patients. (5.10) Glaucoma and cataracts: Monitor patients with change in vision or with a history of increased intraocular pressure, glaucoma, and/or cataracts closely. (5.11) Side EffectsMost common adverse reactions (reported in greater than or equal to 3% of patients) included: nasopharyngitis, headache, sinusitis, bronchitis, and influenza. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir): Use with caution. May cause increased systemic corticosteroid effects. (7.1) USE IN SPECIFIC POPULATIONS Hepatic impairment: Monitor patients for signs of increased drug exposure. (8.6)
FULL PRESCRIBING INFORMATION: CONTENTS*
- 1 ASMANEX HFA INDICATIONS AND USAGE
- 2 ASMANEX HFA DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORMS AND STRENGTHS
- 4 ASMANEX HFA CONTRAINDICATIONS
- 5 WARNINGS AND PRECAUTIONS
- 5.1 Deterioration of Asthma and Acute Episodes
- 5.2 Local Effects
- 5.3 Immunosuppression
- 5.4 Transferring Patients from Systemic Corticosteroid Therapy
- 5.5 Hypercorticism and Adrenal Suppression
- 5.6 Drug Interactions with Strong Cytochrome P450 3A4 Inhibitors
- 5.7 Paradoxical Bronchospasm and Upper Airway Symptoms
- 5.8 Hypersensitivity Reactions Including Anaphylaxis
- 5.9 Reduction in Bone Mineral Density
- 5.10 Effect on Growth
- 5.11 Glaucoma and Cataracts
- 6 ASMANEX HFA ADVERSE REACTIONS
- 7 DRUG INTERACTIONS
- 8 USE IN SPECIFIC POPULATIONS
- 10 OVERDOSAGE
- 11 ASMANEX HFA DESCRIPTION
- 12 CLINICAL PHARMACOLOGY
- 13 NONCLINICAL TOXICOLOGY
- 14 CLINICAL STUDIES
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
- Patient Information
- Instructions for Use
- PRINCIPAL DISPLAY PANEL - 100 mcg Canister Carton
- PRINCIPAL DISPLAY PANEL - 200 mcg Canister Carton
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Treatment of Asthma
ASMANEX® HFA is indicated for the maintenance treatment of asthma as prophylactic therapy in patients 12 years of age and older.
Important Limitations of Use
- ASMANEX HFA is NOT indicated for the relief of acute bronchospasm.
2 DOSAGE AND ADMINISTRATION
2.1 General
ASMANEX HFA should be administered only by the orally inhaled route [see Instructions for Use in the Patient Information leaflet]. After each dose, the patient should be advised to rinse his/her mouth with water without swallowing.
The cap from the mouthpiece of the actuator should be removed before using ASMANEX HFA.
ASMANEX HFA should be primed before using for the first time by releasing 4 test sprays into the air, away from the face, shaking well before each spray. In cases where the inhaler has not been used for more than 5 days, prime the inhaler again by releasing 4 test sprays into the air, away from the face, shaking well before each spray.
The ASMANEX HFA canister should only be used with the ASMANEX HFA actuator. The ASMANEX HFA actuator should not be used with any other inhalation drug product. Actuators from other products should not be used with the ASMANEX HFA canister.
2.2 Dosing
ASMANEX HFA should be administered as two inhalations twice daily every day (morning and evening) by the orally inhaled route.
Shake well prior to each inhalation.
The recommended doses for ASMANEX HFA treatment based on prior asthma therapy are provided in Table 1.
| Previous Therapy | Recommended Dose |
|---|---|
| Inhaled medium-dose corticosteroids | ASMANEX HFA 100 mcg, 2 inhalations twice daily |
| Inhaled high-dose corticosteroids | ASMANEX HFA 200 mcg, 2 inhalations twice daily |
| Oral corticosteroids |
ASMANEX HFA 200 mcg, 2 inhalations twice daily |
If a dosage regimen of ASMANEX HFA fails to provide adequate control of asthma, the therapeutic regimen should be re-evaluated and additional therapeutic options, e.g., replacing the current strength of ASMANEX HFA with a higher strength, initiating an inhaled corticosteroid and long-acting beta2-agonist combination product, or initiating oral corticosteroids, should be considered.
The maximum daily recommended dose is two inhalations of ASMANEX HFA 200 mcg twice daily (maximum of 800 mcg a day). If symptoms arise between doses, an inhaled short-acting beta2-agonist should be taken for immediate relief.
The maximum benefit may not be achieved for 1 week or longer after beginning treatment. Individual patients may experience a variable time to onset and degree of symptom relief. For patients who do not respond adequately after 2 weeks of therapy, higher strength may provide additional asthma control.
After asthma stability has been achieved, it is desirable to titrate to the lowest effective dosage to reduce the possibility of side effects.
3 DOSAGE FORMS AND STRENGTHS
ASMANEX HFA is a pressurized metered dose inhaler that is available in 2 strengths.
ASMANEX HFA 100 mcg delivers 100 mcg of mometasone furoate per actuation.
ASMANEX HFA 200 mcg delivers 200 mcg of mometasone furoate per actuation.
4 CONTRAINDICATIONS
4.1 Status Asthmaticus
ASMANEX HFA is contraindicated in the primary treatment of status asthmaticus or other acute episodes of asthma where intensive measures are required.
4.2 Hypersensitivity
ASMANEX HFA is contraindicated in patients with known hypersensitivity to mometasone furoate or any of the ingredients in ASMANEX HFA [see Warnings and Precautions (5.8)].
5 WARNINGS AND PRECAUTIONS
5.1 Deterioration of Asthma and Acute Episodes
ASMANEX HFA is not indicated for the relief of acute symptoms, i.e., as rescue therapy for the treatment of acute episodes of bronchospasm. An inhaled, short-acting beta2-agonist, not ASMANEX HFA, should be used to relieve acute symptoms such as shortness of breath. When prescribing ASMANEX HFA, the physician must also provide the patient with an inhaled, short-acting beta2-agonist (e.g., albuterol) for treatment of acute symptoms, despite regular twice-daily (morning and evening) use of ASMANEX HFA. Instruct patients to contact their physician immediately if episodes of asthma that are not responsive to bronchodilators occur during the course of treatment with ASMANEX HFA. During such episodes, patients may require therapy with oral corticosteroids.
5.2 Local Effects
In clinical trials, the development of localized infections of the mouth and pharynx with Candida albicans have occurred in patients treated with ASMANEX HFA. If oropharyngeal candidiasis develops, it should be treated with appropriate local or systemic (i.e., oral) antifungal therapy while remaining on treatment with ASMANEX HFA therapy, but at times therapy with ASMANEX HFA may need to be interrupted. Advise patients to rinse the mouth after inhalation of ASMANEX HFA.
5.3 Immunosuppression
Persons who are using drugs that suppress the immune system are more susceptible to infections than healthy individuals.
Chickenpox and measles, for example, can have a more serious or even fatal course in susceptible children or adults using corticosteroids. In such children or adults who have not had these diseases or who are not properly immunized, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) or pooled intravenous immunoglobulin (IVIG) may be indicated. If exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the respective package inserts for complete VZIG and IG prescribing information.) If chickenpox develops, treatment with antiviral agents may be considered.
Inhaled corticosteroids should be used with caution, if at all, in patients with active or quiescent tuberculosis infection of the respiratory tract, untreated systemic fungal, bacterial, viral, or parasitic infections; or ocular herpes simplex.
5.4 Transferring Patients from Systemic Corticosteroid Therapy
Particular care is needed for patients who are transferred from systemically active corticosteroids to ASMANEX HFA because deaths due to adrenal insufficiency have occurred in asthmatic patients during and after transfer from systemic corticosteroids to less systemically available inhaled corticosteroids. After withdrawal from systemic corticosteroids, a number of months are required for recovery of hypothalamic-pituitary-adrenal (HPA) function.
Patients who have been previously maintained on 20 mg or more per day of prednisone (or its equivalent) may be most susceptible, particularly when their systemic corticosteroids have been almost completely withdrawn. During this period of HPA suppression, patients may exhibit signs and symptoms of adrenal insufficiency when exposed to trauma, surgery, or infection (particularly gastroenteritis) or other conditions associated with severe electrolyte loss. Although ASMANEX HFA may improve control of asthma symptoms during these episodes, in recommended doses it supplies less than normal physiological amounts of corticosteroid systemically and does NOT provide the mineralocorticoid activity necessary for coping with these emergencies.
During periods of stress or severe asthma attack, patients who have been withdrawn from systemic corticosteroids should be instructed to resume oral corticosteroids (in large doses) immediately and to contact their physicians for further instruction. These patients should also be instructed to carry a medical identification card indicating that they may need supplementary systemic corticosteroids during periods of stress or severe asthma attack.
Patients requiring oral or other systemic corticosteroids should be weaned slowly from oral or other systemic corticosteroid use after transferring to ASMANEX HFA. Lung function (FEV1 or PEF), beta-agonist use, and asthma symptoms should be carefully monitored during withdrawal of oral or other systemic corticosteroids. In addition to monitoring asthma signs and symptoms, patients should be observed for signs and symptoms of adrenal insufficiency such as fatigue, lassitude, weakness, nausea and vomiting, and hypotension.
Transfer of patients from systemic corticosteroid therapy to ASMANEX HFA may unmask allergic conditions previously suppressed by the systemic corticosteroid therapy, e.g., rhinitis, conjunctivitis, eczema, arthritis, and eosinophilic conditions.
During withdrawal from oral corticosteroids, some patients may experience symptoms of systemically active corticosteroid withdrawal, e.g., joint and/or muscular pain, lassitude, and depression, despite maintenance or even improvement of respiratory function.
5.5 Hypercorticism and Adrenal Suppression
ASMANEX HFA will often help control asthma symptoms with less suppression of HPA function than therapeutically equivalent oral doses of prednisone. Since mometasone furoate is absorbed into the circulation and can be systemically active at higher doses, the beneficial effects of ASMANEX HFA in minimizing HPA dysfunction may be expected only when recommended dosages are not exceeded and individual patients are titrated to the lowest effective dose.
Because of the possibility of systemic absorption of inhaled corticosteroids, patients treated with ASMANEX HFA should be observed carefully for any evidence of systemic corticosteroid effects. Particular care should be taken in observing patients postoperatively or during periods of stress for evidence of inadequate adrenal response.
It is possible that systemic corticosteroid effects such as hypercorticism and adrenal suppression (including adrenal crisis) may appear in a small number of patients, particularly when mometasone furoate is administered at higher than recommended doses over prolonged periods of time. If such effects occur, the dosage of ASMANEX HFA should be reduced slowly, consistent with accepted procedures for reducing systemic corticosteroids and for management of asthma symptoms.
5.6 Drug Interactions with Strong Cytochrome P450 3A4 Inhibitors
Caution should be exercised when considering the coadministration of ASMANEX HFA with ketoconazole, and other known strong cytochrome P450 (CYP) isoenzyme 3A4 (CYP3A4) inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, telithromycin) because adverse effects related to increased systemic exposure to mometasone furoate may occur [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
5.7 Paradoxical Bronchospasm and Upper Airway Symptoms
ASMANEX HFA may produce inhalation induced bronchospasm with an immediate increase in wheezing after dosing that may be life-threatening. If inhalation induced bronchospasm occurs, it should be treated immediately with an inhaled, short-acting bronchodilator. ASMANEX HFA should be discontinued immediately and alternative therapy instituted.
5.8 Hypersensitivity Reactions Including Anaphylaxis
Hypersensitivity reactions such as urticaria, flushing, allergic dermatitis, and bronchospasm, may occur after administration of ASMANEX HFA. Discontinue ASMANEX HFA if such reactions occur [see Contraindications (4.2)].
The following additional hypersensitivity reactions, such as rash, pruritus, angioedema, and anaphylactic reaction, have been reported after administration of mometasone furoate dry powder inhaler [see Adverse Reactions (6.2)].
5.9 Reduction in Bone Mineral Density
Decreases in bone mineral density (BMD) have been observed with long-term administration of products containing inhaled corticosteroids, including mometasone furoate. The clinical significance of small changes in BMD with regard to long-term outcomes, such as fracture, is unknown. Patients with major risk factors for decreased bone mineral content, such as prolonged immobilization, family history of osteoporosis, or chronic use of drugs that can reduce bone mass (e.g., anticonvulsants and corticosteroids) should be monitored and treated with established standards of care.
In a 2-year double-blind study in 103 male and female asthma patients 18 to 50 years of age previously maintained on bronchodilator therapy (Baseline FEV1 85%-88% predicted), treatment with mometasone furoate dry powder inhaler 200 mcg twice daily resulted in significant reductions in lumbar spine (LS) BMD at the end of the treatment period compared to placebo. The mean change from Baseline to Endpoint in the lumbar spine BMD was -0.015 (-1.43%) for the mometasone furoate dry powder inhaler group compared to 0.002 (0.25%) for the placebo group. In another 2-year double-blind study in 87 male and female asthma patients 18 to 50 years of age previously maintained on bronchodilator therapy (Baseline FEV1 82%-83% predicted), treatment with mometasone furoate dry powder inhaler 400 mcg twice daily demonstrated no statistically significant changes in lumbar spine BMD at the end of the treatment period compared to placebo. The mean change from Baseline to Endpoint in the lumbar spine BMD was -0.018 (-1.57%) for the mometasone furoate group compared to -0.006 (-0.43%) for the placebo group.
5.10 Effect on Growth
Orally inhaled corticosteroids, including ASMANEX HFA, may cause a reduction in growth velocity when administered to pediatric patients. Monitor the growth of pediatric patients receiving ASMANEX HFA routinely (e.g., via stadiometry). To minimize the systemic effects of orally inhaled corticosteroids, including ASMANEX HFA, titrate each patient's dose to the lowest dosage that effectively controls his/her symptoms [see Use in Specific Populations (8.4)].
5.11 Glaucoma and Cataracts
Glaucoma, increased intraocular pressure, and cataracts have been reported following the use of long-term administration of inhaled corticosteroids, including mometasone furoate. Therefore, close monitoring is warranted in patients with a change in vision or with a history of increased intraocular pressure, glaucoma, and/or cataracts [see Adverse Reactions (6)].
6 ADVERSE REACTIONS
Systemic and local corticosteroid use may result in the following:
- Candida albicans infection [see Warnings and Precautions (5.2)]
- Immunosuppression [see Warnings and Precautions (5.3)]
- Hypercorticism and adrenal suppression [see Warnings and Precautions (5.5)]
- Growth effects in pediatrics [see Warnings and Precautions (5.10)]
- Glaucoma and cataracts [see Warnings and Precautions (5.11)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The safety of ASMANEX HFA was evaluated in 2 randomized placebo and active-controlled trials of 12 and 26 weeks' duration, conducted as part of a mometasone furoate/formoterol fumarate combination product asthma program, which enrolled 1509 patients with persistent asthma. Patient ages ranged from 12 to 84 years of age, 41% were male and 59% female, 73% were Caucasian and 27% non-Caucasian. Of the total population enrolled in the 2 trials, 432 patients received two inhalations twice daily of either ASMANEX HFA, 100 mcg or 200 mcg/actuation. In the 26-week trial (Trial 1) 192 patients received two inhalations twice daily of ASMANEX HFA 100 mcg/actuation and 196 patients received placebo. In the 12 week trial (Trial 2) 240 patients received two inhalations twice daily of ASMANEX HFA 200 mcg/actuation and 233 and 255 patients received mometasone furoate and formoterol fumarate 100 mcg/5 mcg and 200 mcg/5 mcg/actuation combination products, respectively, as comparators.
In these trials, the proportion of patients who discontinued study treatment early due to adverse reactions was 3% and 2% for ASMANEX HFA 100 and 200 mcg treated patients, respectively, and 4% for placebo-treated patients. Serious adverse reactions, whether considered drug-related or not by the investigators, which occurred more frequently in ASMANEX HFA-treated patients included colitis ulcerative, colonic polyp, chest pain, gastroenteritis, endometriosis, asthma, and hemoptysis; all events occurred at rates less than 1%.
The incidence of treatment emergent adverse reactions associated with ASMANEX HFA are shown in Tables 2 and 3. These are based upon data from each of the 2 clinical trials of 12 or 26 weeks in duration in patients 12 years and older treated with two inhalations twice daily of ASMANEX HFA (100 mcg or 200 mcg), mometasone furoate/formoterol fumarate (100 mcg/5 mcg or 200 mcg/5 mcg), or placebo.
| ASMANEX HFA 100 mcg N=192 n (%) |
Placebo N=196 n (%) |
|
|---|---|---|
| Nasopharyngitis | 15 (8) | 7 (4) |
| Headache | 10 (5) | 7 (4) |
| Influenza | 7 (4) | 5 (3) |
| Sinusitis | 6 (3) | 2 (1) |
| ASMANEX HFA 200 mcg N=240 n (%) |
MF/F 100/5 mcg N=233 n (%) |
MF/F 200/5 mcg N=255 n (%) |
|
|---|---|---|---|
| Nasopharyngitis | 13 (5) | 8 (3) | 12 (5) |
| Headache | 8 (3) | 10 (4) | 5 (2) |
| Bronchitis | 6 (3) | 2 (1) | 7 (3) |
Oral candidiasis has been reported in clinical trials at an incidence of 0.5% in patients using ASMANEX HFA 100 mcg, 0.8% in patients using ASMANEX HFA 200 mcg and 0.5% in the placebo group.
6.2 Postmarketing Experience
There are no postmarketing adverse experiences reported to date with ASMANEX HFA. However, the postmarketing safety experience with mometasone furoate dry powder inhaler is relevant to ASMANEX HFA since they contain the same active ingredient. The following adverse reactions have been reported during post-approval use of mometasone furoate dry powder inhaler. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Immediate and delayed hypersensitivity reactions including rash, pruritus, angioedema and anaphylactic reaction [see Contraindications (4.2) and Warnings and Precautions (5.8)].
Respiratory, Thoracic and Mediastinal Disorders: Asthma aggravation, which may include cough, dyspnea, wheezing and bronchospasm.
7 DRUG INTERACTIONS
In clinical trials, concurrent administration of ASMANEX HFA and other drugs, such as short-acting beta2-agonist and intranasal corticosteroids have not resulted in an increased frequency of adverse drug reactions. No formal drug interaction studies have been performed with ASMANEX HFA.
7.1 Inhibitors of Cytochrome P450 3A4
The main route of metabolism of corticosteroids, including mometasone furoate, is via CYP3A4. After oral administration of ketoconazole, a strong inhibitor of CYP3A4, the mean plasma concentration of orally inhaled mometasone furoate increased. Concomitant administration of CYP3A4 inhibitors may inhibit the metabolism of, and increase the systemic exposure to, mometasone furoate. Caution should be exercised when considering the coadministration of ASMANEX HFA with long-term ketoconazole and other known strong CYP3A4 inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, telithromycin) [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category C
There are no adequate and well-controlled studies of ASMANEX HFA in pregnant women. Animal reproduction studies of mometasone furoate in mice, rats, and rabbits revealed evidence of teratogenicity. Because animal reproduction studies are not always predictive of human response, ASMANEX HFA should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Teratogenic Effects
When administered to pregnant mice, rats, and rabbits, mometasone furoate increased fetal malformations and decreased fetal growth (measured by lower fetal weights and/or delayed ossification). Dystocia and related complications were also observed when mometasone furoate was administered to rats late in gestation. However, experience with oral corticosteroids suggests that rodents are more prone to teratogenic effects from corticosteroid exposure than humans.
In a mouse reproduction study, subcutaneous mometasone furoate produced cleft palate at approximately one-third of the maximum recommended daily human dose (MRHD) on a mcg/m2 basis and decreased fetal survival at approximately 1 times the MRHD. No toxicity was observed at approximately one-tenth of the MRHD on a mcg/m2 basis.
In a rat reproduction study, mometasone furoate produced umbilical hernia at topical dermal doses approximately 6 times the MRHD on a mcg/m2 basis and delays in ossification at approximately 3 times the MRHD on a mcg/m2 basis.
In another study, rats received subcutaneous doses of mometasone furoate throughout pregnancy or late in gestation. Treated animals had prolonged and difficult labor, fewer live births, lower birth weight, and reduced early pup survival at a dose that was approximately 8 times the MRHD on an area under the curve (AUC) basis. Similar effects were not observed at approximately 4 times MRHD on an AUC basis.
In rabbits, mometasone furoate caused multiple malformations (e.g., flexed front paws, gallbladder agenesis, umbilical hernia, hydrocephaly) at topical dermal doses approximately 3 times the MRHD on a mcg/m2 basis. In an oral study, mometasone furoate increased resorptions and caused cleft palate and/or head malformations (hydrocephaly and domed head) at a dose less than the MRHD based on AUC. At a dose approximately 2 times the MRHD based on AUC, most litters were aborted or resorbed [see Nonclinical Toxicology (13.2)].
Nonteratogenic Effects
Hypoadrenalism may occur in infants born to women receiving corticosteroids during pregnancy. Infants born to mothers taking substantial corticosteroid doses during pregnancy should be monitored for signs of hypoadrenalism.
8.2 Labor and Delivery
There are no adequate and well-controlled human studies that have studied the effects of ASMANEX HFA during labor and delivery.
8.3 Nursing Mothers
It is not known whether ASMANEX HFA is excreted in human milk. Because other corticosteroids are excreted in human milk, caution should be exercised when ASMANEX HFA is administered to a nursing woman.
Since there are no data from well-controlled human studies on the use of ASMANEX HFA on nursing mothers, a decision should be made whether to discontinue nursing or to discontinue ASMANEX HFA, taking into account the importance of ASMANEX HFA to the mother.
8.4 Pediatric Use
The safety and effectiveness of ASMANEX HFA have been established in patients 12 years of age and older in 2 clinical trials of 12 and 26 weeks in duration. In the 2 clinical trials, 32 patients 12 to 17 years of age were treated with ASMANEX HFA. No overall differences in effectiveness were observed between patients in this age group compared to those observed in patients 18 years of age and older. There were no obvious differences in the type or frequency of adverse drug reactions reported in this age group compared to patients 18 years of age and older. The safety and efficacy of ASMANEX HFA have not been established in children less than 12 years of age.
Controlled clinical studies have shown that inhaled corticosteroids may cause a reduction in growth velocity in pediatric patients. In these studies, the mean reduction in growth velocity was approximately 1 cm per year (range 0.3 to 1.8 per year) and appears to depend upon dose and duration of exposure. This effect was observed in the absence of laboratory evidence of HPA axis suppression, suggesting that growth velocity is a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA axis function. The long-term effects of this reduction in growth velocity associated with orally inhaled corticosteroids, including the impact on final adult height, are unknown. The potential for "catch-up" growth following discontinuation of treatment with orally inhaled corticosteroids has not been adequately studied.
The growth of children and adolescents receiving orally inhaled corticosteroids, including ASMANEX HFA, should be monitored routinely (e.g., via stadiometry). If a child or adolescent on any corticosteroid appears to have growth suppression, the possibility that he/she is particularly sensitive to this effect should be considered. The potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the risks associated with alternative therapies. To minimize the systemic effects of orally inhaled corticosteroids, including ASMANEX HFA, each patient should be titrated to his/her lowest effective dose [see Dosage and Administration (2.2)].
8.5 Geriatric Use
A total of 38 patients 65 years of age and older (3 of whom were 75 years and older) have been treated with ASMANEX HFA in 2 clinical trials of 12 and 26 weeks in duration. No overall differences in safety or effectiveness were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out. Based on available data for ASMANEX HFA, no adjustment of dosage in geriatric patients is warranted.
8.6 Hepatic Impairment
Concentrations of mometasone furoate appear to increase with severity of hepatic impairment [see Clinical Pharmacology (12.3)].
10 OVERDOSAGE
Chronic overdosage may result in signs/symptoms of hypercorticism [see Warnings and Precautions (5.5)]. Single oral doses up to 8000 mcg of mometasone furoate have been studied on human volunteers with no adverse reactions reported.
11 DESCRIPTION
ASMANEX HFA is a metered dose inhaler for oral inhalation only, consisting of 100 mcg and 200 mcg of mometasone furoate per actuation.
Mometasone furoate, the active component of ASMANEX HFA, is a corticosteroid having the chemical name 9,21-dichloro-11(Beta),17-dihydroxy-16 (alpha)-methylpregna-1,4-diene-3,20-dione 17-(2-furoate) with the following chemical structure:
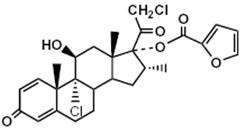
Mometasone furoate is a white powder with an empirical formula of C27H30Cl2O6, and molecular weight 521.44. It is practically insoluble in water; slightly soluble in methanol, ethanol, and isopropanol; soluble in acetone.
Each ASMANEX HFA 100 mcg and 200 mcg is a hydrofluoroalkane (HFA-227: 1,1,1,2,3,3,3-heptafluoropropane) propelled pressurized metered dose inhaler containing sufficient amount of drug for 120 actuations [see How Supplied/Storage and Handling (16)]. After priming, each actuation of the inhaler delivers 115 or 225 mcg of mometasone furoate in 69.6 mg of suspension from the valve and delivers 100 or 200 mcg of mometasone furoate from the actuator. The actual amount of drug delivered to the lung may depend on patient factors, such as the coordination between actuation of the device and inspiration through the delivery system. ASMANEX HFA also contains ethanol as a cosolvent and oleic acid as a surfactant.
ASMANEX HFA should be primed before using for the first time by releasing 4 test sprays into the air, away from the face, shaking well before each spray. In cases where the inhaler has not been used for more than 5 days, prime the inhaler again by releasing 4 test sprays into the air, away from the face, shaking well before each spray.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Mometasone furoate is a corticosteroid demonstrating potent anti-inflammatory activity. The precise mechanism of corticosteroid action on asthma is not known. Inflammation is an important component in the pathogenesis of asthma. Corticosteroids have been shown to have a wide range of inhibitory effects on multiple cell types (e.g., mast cells, eosinophils, neutrophils, macrophages, and lymphocytes) and mediators (e.g., histamine, eicosanoids, leukotrienes, and cytokines) involved in inflammation and in the asthmatic response. These anti-inflammatory actions of corticosteroids may contribute to their efficacy in asthma.
Mometasone furoate has been shown in vitro to exhibit a binding affinity for the human glucocorticoid receptor, which is approximately 12 times that of dexamethasone, 7 times that of triamcinolone acetonide, 5 times that of budesonide, and 1.5 times that of fluticasone. The clinical significance of these findings is unknown.
12.2 Pharmacodynamics
HPA Axis Effects
The effects of inhaled mometasone furoate administered via ASMANEX HFA on adrenal function have not been directly evaluated. However, the effects of inhaled mometasone furoate, administered as part of a mometasone furoate/formoterol fumarate inhalation aerosol combination product, on adrenal function were evaluated in two clinical trials in patients with asthma. As no evidence of a pharmacokinetic drug interaction between mometasone furoate and formoterol was observed when the two drugs were administered in combination, the HPA axis effects from the combination product are applicable to ASMANEX HFA. For the mometasone furoate/formoterol fumarate combination product clinical program, HPA-axis function was assessed by 24-hour plasma cortisol AUC. Although both these trials have open-label design and contain a small number of subjects per treatment arm, results from these trials taken together demonstrated suppression of 24-hour plasma cortisol AUC for the combination mometasone furoate/formoterol fumarate 200 mcg/5 mcg compared to placebo consistent with the known systemic effects of inhaled corticosteroid.
In a 42-day, open-label, placebo- and active-controlled study, the mean change from baseline plasma cortisol AUC(0-24 hr) was 8%, 22% and 34% lower compared to placebo for the mometasone furoate/formoterol fumarate 100 mcg/5 mcg (n=13), mometasone furoate/formoterol fumarate 200 mcg/5 mcg (n=15) and fluticasone propionate/salmeterol xinafoate 230 mcg/21 mcg (n=16) treatment groups, respectively.
In a 52-week, open-label safety study, the mean plasma cortisol AUC(0-24 hr) was 2.2%, 29.6%, 16.7%, and 32.2% lower from baseline for the mometasone furoate/formoterol fumarate 100 mcg/5 mcg (n=18), mometasone furoate/formoterol fumarate 200 mcg/5 mcg (n=20), fluticasone propionate/salmeterol xinafoate 125/25 mcg (n=8), and fluticasone propionate/salmeterol xinafoate 250/25 mcg (n=11) treatment groups, respectively.
The potential effect of mometasone furoate via a dry powder inhaler (DPI) on the HPA axis was also assessed in a 29-day study. A total of 64 adult patients with mild to moderate asthma were randomized to one of 4 treatment groups: mometasone furoate DPI 440 mcg twice daily, mometasone furoate DPI 880 mcg twice daily, oral prednisone 10 mg once daily, or placebo. The 30-minute post-Cosyntropin stimulation serum cortisol concentration on Day 29 was 23.2 mcg/dL for the mometasone furoate DPI 440 mcg twice daily group and 20.8 mcg/dL for the mometasone furoate DPI 880 mcg twice daily group, compared to 14.5 mcg/dL for the oral prednisone 10 mg group and 25 mcg/dL for the placebo group. The difference between mometasone furoate DPI 880 mcg twice daily (twice the maximum recommended dose) and placebo was statistically significant.
12.3 Pharmacokinetics
As no evidence of a pharmacokinetic drug interaction between mometasone furoate and formoterol was observed when the two drugs were administered from a mometasone furoate/formoterol fumarate combination product, the pharmacokinetics information from the combination product is applicable to ASMANEX HFA.
Absorption
Healthy Subjects: Following oral inhalation of single doses of ASMANEX HFA, mometasone furoate was absorbed in healthy subjects with median Tmax values ranging from 0.50 to 2 hours. Following single-dose administration of higher than recommended dose of ASMANEX HFA (4 inhalations of ASMANEX HFA 200 mcg) in healthy subjects, the arithmetic mean (CV%) Cmax and AUC(0-tf) values for mometasone furoate were 53 (102) pg/mL and 992 (80) pg∙hr/mL, respectively. Studies using oral dosing of labeled and unlabeled drug have demonstrated that the oral systemic bioavailability of mometasone furoate is negligible (<1%).
Asthma Patients: Following oral inhalation of single and multiple doses of the mometasone furoate/formoterol fumarate combination product, mometasone furoate was absorbed in asthma patients with median Tmax values ranging from 1 to 2 hours. Following single-dose administration of mometasone furoate/formoterol fumarate 400 mcg/10 mcg, the arithmetic mean (CV%) Cmax and AUC(0-12 hr) values for mometasone furoate were 20 (88) pg/mL and 170 (94) pg∙hr/mL, respectively, while the corresponding estimates following twice daily dosing of mometasone furoate/formoterol fumarate 400 mcg/10 mcg at steady-state were 60 (36) pg/mL and 577 (40) pg∙hr/mL.
Distribution
Based on the study employing a 1000 mcg inhaled dose of tritiated mometasone furoate inhalation powder in humans, no appreciable accumulation of mometasone furoate in the red blood cells was found. Following an intravenous 400 mcg dose of mometasone furoate, the plasma concentrations showed a biphasic decline, with a mean steady-state volume of distribution of 152 liters. The in vitro protein binding for mometasone furoate was reported to be 98% to 99% (in a concentration range of 5 to 500 ng/mL).
Metabolism
Studies have shown that mometasone furoate is primarily and extensively metabolized in the liver of all species investigated and undergoes extensive metabolism to multiple metabolites. In vitro studies have confirmed the primary role of human liver CYP3A4 in the metabolism of this compound; however, no major metabolites were identified. Human liver CYP3A4 metabolizes mometasone furoate to 6-beta hydroxy mometasone furoate.
Excretion
Following an intravenous dosing, the terminal half-life was reported to be about 5 hours. Following the inhaled dose of tritiated 1000 mcg mometasone furoate, the radioactivity is excreted mainly in the feces (a mean of 74%), and to a small extent in the urine (a mean of 8%) up to 7 days. No radioactivity was associated with unchanged mometasone furoate in the urine. Absorbed mometasone furoate is cleared from plasma at a rate of approximately 12.5 mL/min/kg, independent of dose. The effective t½ for mometasone furoate following inhalation with DULERA was 25 hours in healthy subjects and in patients with asthma.
Special Populations
Hepatic/Renal Impairment: There are no data regarding the specific use of ASMANEX HFA in patients with hepatic or renal impairment.
A study evaluating the administration of a single inhaled dose of 400 mcg mometasone furoate by a dry powder inhaler to subjects with mild (n=4), moderate (n=4), and severe (n=4) hepatic impairment resulted in only 1 or 2 subjects in each group having detectable peak plasma concentrations of mometasone furoate (ranging from 50-105 pg/mL). The observed peak plasma concentrations appear to increase with severity of hepatic impairment; however, the numbers of detectable levels were few.
Gender and Race: Specific studies to examine the effects of gender and race on the pharmacokinetics of ASMANEX HFA have not been specifically studied.
Geriatrics: The pharmacokinetics of ASMANEX HFA have not been specifically studied in the elderly population.
Drug-Drug Interactions
A single-dose crossover study was conducted to compare the pharmacokinetics of 4 inhalations of the following: mometasone furoate MDI, formoterol MDI, mometasone furoate/formoterol fumarate MDI combination product, and mometasone furoate MDI plus formoterol fumarate MDI administered concurrently. The results of the study indicated that there was no evidence of a pharmacokinetic interaction between mometasone furoate and formoterol.
Inhibitors of Cytochrome P450 Enzymes: Ketoconazole: In a drug interaction study, an inhaled dose of mometasone furoate 400 mcg delivered by a dry powder inhaler was given to 24 healthy subjects twice daily for 9 days and ketoconazole 200 mg (as well as placebo) were given twice daily concomitantly on Days 4 to 9. Mometasone furoate plasma concentrations were <150 pg/mL on Day 3 prior to coadministration of ketoconazole or placebo. Following concomitant administration of ketoconazole, 4 out of 12 subjects in the ketoconazole treatment group (n=12) had peak plasma concentrations of mometasone furoate >200 pg/mL on Day 9 (211-324 pg/mL). Mometasone furoate plasma levels appeared to increase and plasma cortisol levels appeared to decrease upon concomitant administration of ketoconazole.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year carcinogenicity study in Sprague Dawley rats, mometasone furoate demonstrated no statistically significant increase in the incidence of tumors at inhalation doses up to 67 mcg/kg (approximately 14 times the MRHD on an AUC basis). In a 19-month carcinogenicity study in Swiss CD-1 mice, mometasone furoate demonstrated no statistically significant increase in the incidence of tumors at inhalation doses up to 160 mcg/kg (approximately 9 times the MRHD on an AUC basis).
Mometasone furoate increased chromosomal aberrations in an in vitro Chinese hamster ovary cell assay, but did not have this effect in an in vitro Chinese hamster lung cell assay. Mometasone furoate was not mutagenic in the Ames test or mouse lymphoma assay, and was not clastogenic in an in vivo mouse micronucleus assay, a rat bone marrow chromosomal aberration assay, or a mouse male germ-cell chromosomal aberration assay. Mometasone furoate also did not induce unscheduled DNA synthesis in vivo in rat hepatocytes.
In reproductive studies in rats, impairment of fertility was not produced by subcutaneous doses up to 15 mcg/kg (approximately 8 times the MRHD on an AUC basis).
13.2 Animal Toxicology and/or Pharmacology
Reproductive Toxicology Studies
In mice, mometasone furoate caused cleft palate at subcutaneous doses of 60 mcg/kg and above (approximately one-third of the maximum recommended human dose MRHD on a mcg/m2 basis). Fetal survival was reduced at 180 mcg/kg (approximately equal to the MRHD on a mcg/m2 basis). No toxicity was observed at 20 mcg/kg (approximately one-tenth of the MRHD on a mcg/m2 basis).
In rats, mometasone furoate produced umbilical hernia at topical dermal doses of 600 mcg/kg and above (approximately 6 times the MRHD on a mcg/m2 basis). A dose of 300 mcg/kg (approximately 3 times the MRHD on a mcg/m2 basis) produced delays in ossification, but no malformations.
When rats received subcutaneous doses of mometasone furoate throughout pregnancy or during the later stages of pregnancy, 15 mcg/kg (approximately 8 times the MRHD on an AUC basis) caused prolonged and difficult labor and reduced the number of live births, birth weight, and early pup survival. Similar effects were not observed at 7.5 mcg/kg (approximately 4 times the MRHD on an AUC basis).
In rabbits, mometasone furoate caused multiple malformations (e.g., flexed front paws, gallbladder agenesis, umbilical hernia, hydrocephaly) at topical dermal doses of 150 mcg/kg and above (approximately 3 times the MRHD on a mcg/m2 basis). In an oral study, mometasone furoate increased resorptions and caused cleft palate and/or head malformations (hydrocephaly and domed head) at 700 mcg/kg (less than the MRHD on an AUC basis). At 2800 mcg/kg (approximately 2 times the MRHD on an AUC basis) most litters were aborted or resorbed. No toxicity was observed at 140 mcg/kg (less than the MRHD on an AUC basis).
14 CLINICAL STUDIES
14.1 Asthma
The safety and efficacy of ASMANEX HFA was demonstrated in two randomized, double-blind, placebo- or active-controlled multi-center clinical trials of 12 and 26 weeks' duration, conducted as part of a mometasone furoate/formoterol fumarate 100/5 mcg or 200/5 mcg combination product development program. A total of 1509 patients 12 years of age and older with persistent asthma (mean baseline FEV1 of 66% to 73% predicted) were evaluated.
Trial 1: Clinical Trial with ASMANEX HFA 100 mcg
This 26-week, placebo-controlled trial conducted as part of a mometasone furoate/formoterol fumarate combination product asthma program evaluated 781 patients 12 years of age and older. Of these patients, 192 patients received ASMANEX HFA 100 mcg and 196 patients received placebo, each administered as 2 inhalations twice daily by metered dose inhalation aerosols. All other maintenance therapies were discontinued. The study included a 2- to 3-week run-in period with ASMANEX HFA 100 mcg, 2 inhalations twice daily. Patients ranged from 12 to 76 years of age, 41% were male and 59% female, and 72% were Caucasian and 28% non-Caucasian. Patients had persistent asthma and were not well controlled on medium dose of inhaled corticosteroids prior to randomization. Mean FEV1 and mean percent predicted FEV1 were similar among all treatment groups (2.33 L, 73%). Thirteen (7%) patients receiving ASMANEX HFA 100 mcg and 46 (23%) patients receiving placebo discontinued the study early due to treatment failure.
The change in mean trough FEV1 from baseline to Week 12 compared to placebo was assessed to evaluate the efficacy of ASMANEX HFA 100 mcg. The change from baseline to week 12 in the mean trough FEV1 was greater among patients receiving ASMANEX HFA 100 mcg 2 inhalations twice daily than among those receiving placebo (treatment difference from placebo 0.12 L and 95% confidence interval [0.05, 0.20]).
Clinically judged deteriorations in asthma or reductions in lung function were also assessed to evaluate the efficacy of ASMANEX HFA 100 mcg. Deteriorations in asthma were defined as any of the following: a 20% decrease in FEV1; a 30% decrease in PEF on two or more consecutive days; emergency treatment, hospitalization, or treatment with systemic corticosteroids or other asthma medications not allowed per protocol. Sixty-five (34%) patients who received ASMANEX HFA 100 mcg reported an event compared to 109 (56%) patients who received placebo.
Treatment of asthma patients with ASMANEX HFA 100 mcg, two inhalations twice daily also resulted in fewer nocturnal awakenings and improved morning peak flow compared to those who received placebo.
Trial 2: Clinical Trial with ASMANEX HFA 200 mcg
This 12-week randomized, double-blind, active-controlled trial also conducted as part of a mometasone furoate/formoterol fumarate combination product asthma program evaluated a total of 728 patients 12 years of age and older comparing ASMANEX HFA 200 mcg (n=240 patients), mometasone furoate/formoterol fumarate 200 mcg/5 mcg (n=255 patients), and mometasone furoate/formoterol fumarate 100 mcg/5 mcg (n=233 patients), each administered as 2 inhalations twice daily by metered dose inhalation aerosols. All other maintenance therapies were discontinued. This trial included a 2- to 3-week run-in period with ASMANEX HFA 200 mcg, 2 inhalations twice daily. Patients had persistent asthma and were uncontrolled on high-dose inhaled corticosteroids prior to study entry. Patients ranged from 12 to 84 years of age, 44% were male and 56% female, and 89% were Caucasian and 11% non-Caucasian. Mean FEV1 and mean percent predicted FEV1 values were similar among all treatment groups (2.05 L, 66%). The number of patients who discontinued the trial early due to treatment failure were 11 (5%) in the mometasone furoate/formoterol fumarate 100 mcg/5 mcg group, 8 (3%) in the mometasone furoate/formoterol fumarate 200 mcg/5 mcg group, and 13 (5%) in the ASMANEX HFA 200 mcg group.
In order to assess the added benefit of a higher dose of mometasone in the 200 mcg/actuation mometasone furoate product compared to the lower dose 100 mcg/actuation product, trough FEV1 at 12 weeks was compared between the combination mometasone furoate/formoterol fumarate 200 mcg/5 mcg and 100 mcg/5 mcg treatment groups as a secondary endpoint. Improvement in trough FEV1 from baseline to week 12 in patients who received mometasone furoate 200 mcg in combination with formoterol fumarate 5 mcg was numerically greater than among patients who received mometasone furoate 100 mcg in combination with formoterol fumarate 5 mcg (treatment difference of 0.05 L and 95% confidence interval [-0.02, 0.10]).
Other Studies
In addition to Trial 1 and Trial 2, the safety and efficacy of mometasone furoate MDI 100 mcg and 200 mcg, in comparison to placebo were demonstrated in three other 12-week, placebo-controlled trials which evaluated the mean change in FEV1 from baseline as a primary endpoint.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
ASMANEX HFA is available in two strengths and supplied in the following package size (Table 4):
| Package | NDC |
|---|---|
| ASMANEX HFA 100 mcg 120 metered actuations |
0085-4333-01 |
| ASMANEX HFA 200 mcg 120 metered actuations |
0085-4334-01 |
Each strength is supplied as a pressurized aluminum canister that has a blue plastic actuator integrated with a dose counter and a pink dust cap. Each canister has a net fill weight of 13 grams. Each inhaler is placed into a carton. Each carton contains 1 inhaler.
Initially the dose counter will display "124" actuations. After the initial priming with 4 actuations, the dose counter will read "120" and the inhaler is now ready for use.
16.2 Storage and Handling
The ASMANEX HFA canister should only be used with the ASMANEX HFA actuator. The ASMANEX HFA actuator should not be used with any other inhalation drug product. Actuators from other products should not be used with the ASMANEX HFA canister.
The correct amount of medication in each inhalation cannot be ensured after the labeled number of actuations from the canister has been used, even though the inhaler may not feel completely empty and may continue to operate. The inhaler should be discarded when the labeled number of actuations has been used (the dose counter will read "0").
Store at controlled room temperature 20-25°C (68-77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
For best results, the canister should be at room temperature before use. Shake well and remove the cap from the mouthpiece of the actuator before using. Keep out of reach of children. Avoid spraying in eyes.
Contents Under Pressure: Do not puncture. Do not use or store near heat or open flame. Exposure to temperatures above 120°F may cause bursting. Never throw container into fire or incinerator.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Not for Acute Symptoms
Advise patients that ASMANEX HFA is not indicated to relieve acute asthma symptoms, and extra doses should not be used for that purpose. ASMANEX HFA is not a bronchodilator and should not be used to treat status asthmaticus or to relieve acute asthma symptoms. Acute asthma symptoms should be treated with an inhaled, short-acting beta2-agonist such as albuterol. Prescribe the patient with such medication and instruct the patient in how it should be used [see Warnings and Precautions (5.1)].
Instruct patients to seek medical attention immediately if they experience any of the following:
- If their symptoms worsen
- Significant decrease in lung function as outlined by the physician
- If they need more inhalations of a short-acting beta2-agonist than usual
Advise patients not to increase the dose or frequency of ASMANEX HFA. The daily dosage of ASMANEX HFA should not exceed two inhalations twice daily. If they miss a dose, instruct patients to take their next dose at the same time they normally do.
Advise patients not to stop or reduce ASMANEX HFA therapy without physician/provider guidance since symptoms may recur after discontinuation.
Local Effects
Advise patients that localized infections with Candida albicans occurred in the mouth and pharynx in some patients. If oropharyngeal candidiasis develops, it should be treated with appropriate local or systemic (i.e., oral) antifungal therapy while still continuing with ASMANEX HFA therapy, but at times therapy with ASMANEX HFA may need to be temporarily interrupted under close medical supervision. Advise patients to rinse the mouth after inhalation of ASMANEX HFA [see Warnings and Precautions (5.2)].
Immunosuppression
Warn patients who are on immunosuppressant doses of corticosteroids to avoid exposure to chickenpox or measles and, if exposed, to consult their physician without delay. Inform patients of potential worsening of existing tuberculosis, fungal, bacterial, viral, or parasitic infections, or ocular herpes simplex [see Warnings and Precautions (5.3)].
Hypercorticism and Adrenal Suppression
Advise patients that ASMANEX HFA may cause systemic corticosteroid effects of hypercorticism and adrenal suppression. Additionally, instruct patients that deaths due to adrenal insufficiency have occurred during and after transfer from systemic corticosteroids. Patients should taper slowly from systemic corticosteroids if transferring to ASMANEX HFA [see Warnings and Precautions (5.4 and 5.5)].
Reduction in Bone Mineral Density
Advise patients who are at an increased risk for decreased BMD that the use of corticosteroids may pose an additional risk and should be monitored and, where appropriate, be treated for this condition [see Warnings and Precautions (5.9)].
Reduced Growth Velocity
Inform patients that orally inhaled corticosteroids, including ASMANEX HFA, may cause a reduction in growth velocity when administered to pediatric patients. Physicians should closely follow the growth of pediatric patients taking corticosteroids by any route [see Warnings and Precautions (5.10)].
Glaucoma and Cataracts
Advise patients that long-term use of inhaled corticosteroids may increase the risk of some eye problems (glaucoma or cataracts); regular eye examinations should be considered [see Warnings and Precautions (5.11)].
Hypersensitivity Reactions Including Anaphylaxis
Advise patients that hypersensitivity reactions, such as urticaria, flushing, allergic dermatitis, bronchospasm, rash, pruritus, angioedema, and anaphylactic reaction, may occur after administration of ASMANEX HFA. Instruct patients to discontinue ASMANEX HFA if such reactions occur [see Warnings and Precautions (5.8)].
Use Daily for Best Effect
Advise patients to use ASMANEX HFA at regular intervals, since its effectiveness depends on regular use. Maximum benefit may not be achieved for 1 week or longer after starting treatment. If symptoms do not improve after 2 weeks of therapy or if the condition worsens, instruct patients to contact their physician.
Manufactured for: Merck Sharp & Dohme Corp., a subsidiary of
MERCK & CO., INC., Whitehouse Station, NJ 08889, USA
Manufactured by: 3M Health Care Ltd., Loughborough, United Kingdom.
For patent information: www.merck.com/product/patent/home.html
Copyright © 2014 Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc.
All rights reserved.
uspi-mk0887-ao-1404r000
Patient Information
ASMANEX® HFA (AZ-ma-neks)
(mometasone furoate)
Inhalation Aerosol
Read this Patient Information before you start using ASMANEX HFA and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
What is ASMANEX HFA?
ASMANEX HFA is an inhaled prescription medicine used as maintenance treatment for the prevention and control of asthma symptoms in people 12 years of age and older.
- ASMANEX HFA is not used to treat sudden severe symptoms of asthma.
- ASMANEX HFA should not be used as a rescue inhaler.
- It is not known if ASMANEX HFA is safe and effective in children less than 12 years of age.
Who should not use ASMANEX HFA?
Do not use ASMANEX HFA:
- to treat sudden severe symptoms of asthma
- if you are allergic to mometasone furoate or any of the ingredients in ASMANEX HFA. See the end of this Patient Information leaflet for a complete list of ingredients in ASMANEX HFA.
What should I tell my doctor before and during treatment with ASMANEX HFA?
Before you use ASMANEX HFA, tell your healthcare provider if you:
- have liver problems
- have osteoporosis
- have an immune system problem
- have eye problems such as increased pressure in the eye, glaucoma, or cataracts
- are allergic to any medicines
- are exposed to chickenpox or measles
- have or had tuberculosis (TB)
- have any other medical problems
- are pregnant or planning to become pregnant. It is not known if ASMANEX HFA may harm your unborn baby.
- are breastfeeding. It is not known if ASMANEX HFA passes into your breast milk and if it can harm your baby. You and your healthcare provider should decide if you will either take ASMANEX HFA or breastfeed.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
ASMANEX HFA may affect the way other medicines work, and other medicines may affect how ASMANEX HFA works.
Especially, tell your healthcare provider if you take antifungal medicines or anti-HIV medicines such as:
|
|
Ask your healthcare provider if you are not sure if any of your medicines are the kinds listed above.
Know the medicines you take. Keep a list and show it to your healthcare provider and pharmacist each time you get a new medicine.
How should I use ASMANEX HFA?
Read the step-by-step instructions for using ASMANEX HFA in the Instructions for Use.
- Use ASMANEX HFA exactly as prescribed. Do not use ASMANEX HFA more often than prescribed.
- You must use ASMANEX HFA regularly. It may take 1 week or longer after you start using ASMANEX HFA for your asthma symptoms to get better. Do not stop using ASMANEX HFA even if you are feeling better, unless your healthcare provider tells you to.
- Do not change or stop using ASMANEX HFA or other asthma medicines used to control or treat your breathing problems unless told to do so by your healthcare provider. Your healthcare provider will change your medicines as needed.
- ASMANEX HFA comes in 2 strengths. Your healthcare provider has prescribed the strength that is best for you. Pay attention to the differences between ASMANEX HFA and your other inhaled medicines, including their prescribed use and the way they look.
- Take ASMANEX HFA every day, with 2 puffs in the morning and 2 puffs in the evening.
- If you miss a dose of ASMANEX HFA, skip your missed dose and take your next dose at your regular time. Do not take ASMANEX HFA more often or use more puffs than you have been prescribed.
- If you take more ASMANEX HFA than your healthcare provider has prescribed, call your healthcare provider right away.
- ASMANEX HFA does not relieve sudden asthma symptoms. Always have a rescue inhaler with you to treat sudden symptoms. Use your rescue inhaler if you have breathing problems between doses of ASMANEX HFA. If you do not have a rescue inhaler, call your healthcare provider to have a rescue inhaler prescribed for you.
- Do not use the ASMANEX HFA canister or actuator with any other medicines. Do not use any other medicine canister or actuator with ASMANEX HFA.
- Rinse your mouth with water after each dose (2 puffs) of ASMANEX HFA. This will help to lessen the chance of getting a yeast infection (thrush) in your mouth and throat.
- Do not spray ASMANEX HFA in your eyes. If you accidentally get ASMANEX HFA in your eyes, rinse your eyes with water and if redness or irritation continues, call your healthcare provider.
-
Call your healthcare provider or get medical care right away if:
- your breathing problems worsen with ASMANEX HFA
- you need to use your rescue inhaler more often than usual
- your rescue inhaler does not work as well for you at relieving symptoms
- you need to use 4 or more inhalations of your rescue inhaler for 2 or more days in a row
- you use 1 whole canister of your rescue inhaler within 8 weeks
- your peak flow meter results decrease. Your healthcare provider will tell you the numbers that are right for you.
- you have asthma and your symptoms do not improve after using ASMANEX HFA regularly for 1 to 2 weeks
What are the possible side effects of ASMANEX HFA?
ASMANEX HFA can cause serious side effects, including
- Thrush in your mouth and throat. You may develop a yeast infection (Candida albicans) in your mouth or throat. Rinse your mouth with water after using ASMANEX HFA to help prevent an infection in your mouth or throat.
-
Immune system effects and a higher chance for infections.
Tell your healthcare provider about any signs of infection such as:- fever
- feeling tired
- pain
- nausea
- body aches
- vomiting
- chills
-
Adrenal insufficiency that can lead to death can happen when you stop taking oral corticosteroid medicines and start using inhaled corticosteroid medicines. Adrenal insufficiency can also happen in people who take higher doses of ASMANEX HFA than recommended over a long period of time. When your body is under stress such as from fever, trauma (such as a car accident), infection, or surgery, adrenal insufficiency can get worse. Symptoms of adrenal insufficiency include:
- feeling tired or exhausted (fatigue)
- lack of energy
- weakness
- nausea and vomiting
- low blood pressure (hypotension)
- dizziness or feeling faint
- Increased wheezing right after taking ASMANEX HFA. Always have a rescue inhaler with you to treat sudden wheezing.
-
Serious allergic reactions. Stop taking ASMANEX HFA and call your healthcare provider or get emergency medical care right away if you get any of the following symptoms of a serious allergic reaction:
- rash
- hives
- swelling, including swelling of the face, mouth, and tongue
- breathing problems
- Lower bone mineral density. This may be a problem for people who already have a higher chance for low bone density (osteoporosis).
- Slowed growth in children. A child's growth should be checked often.
- Eye problems including glaucoma and cataracts. You should have regular eye exams while using ASMANEX HFA.
The most common side effects of ASMANEX HFA include:
- inflammation of the nose and throat (nasopharyngitis)
- headache
- inflammation of the sinuses (sinusitis)
- bronchitis
- flu infection (influenza)
Other side effects:
Worsening asthma or sudden asthma attacks have been reported with the use of inhaled mometasone furoate.
Tell your healthcare provider about any side effect that bothers you or that does not go away.
These are not all the side effects with ASMANEX HFA. Ask your healthcare provider or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ASMANEX HFA?
- Store ASMANEX HFA at room temperature between 68°F to 77°F (20°C to 25°C).
- The contents of your ASMANEX HFA are under pressure. Do not puncture. Do not use or store near heat or open flame. Storage above 120°F may cause the canister to burst.
- Do not throw container into fire or incinerator.
- Keep ASMANEX HFA and all medicines out of the reach of children.
General Information about the safe and effective use of ASMANEX HFA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ASMANEX HFA for a condition for which it was not prescribed. Do not give your ASMANEX HFA to other people, even if they have the same condition that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about ASMANEX HFA. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about ASMANEX HFA that was written for healthcare professionals.
For more information or to report side effects, you may go to www.ASMANEX.com or call 1-877-888-4231.
What are the ingredients in ASMANEX HFA?
Active ingredient: mometasone furoate
Inactive ingredients: hydrofluoroalkane (HFA-227: 1,1,1,2,3,3,3-heptafluoropropane), ethanol and oleic acid
Instructions for Use
ASMANEX® HFA
(mometasone furoate)
Inhalation Aerosol
How to use your ASMANEX HFA:
Before using your ASMANEX HFA, read the complete instructions and use only as directed.
The parts of your ASMANEX HFA:
There are 2 main parts to your ASMANEX HFA inhaler: the metal canister that holds the medicine and the plastic actuator that sprays the medicine from the canister. The inhaler also has a cap that covers the mouthpiece of the actuator (see Figure 1). The cap from the mouthpiece must be removed before use. The inhaler contains "120" actuations (puffs).
The inhaler comes with a dose counter located on the plastic actuator (see Figure 1). The counter display will show the number of actuations (puffs) of medicine remaining. The first time you use ASMANEX HFA the dose counter will show "124" actuations remaining. Each time you press the canister, a puff of medicine is released and the counter will count down by 1. The counter will stop counting at 0.
- You should not remove the canister from the actuator because reinsertion may cause the counter to count down by 1 and discharge a puff.
- Use the ASMANEX HFA canister only with the actuator supplied with the product. Do not use parts of the ASMANEX HFA inhaler with parts from any other inhalation medicine.
Before using your ASMANEX HFA:
Remove the cap from the mouthpiece of the actuator (see Figure 2). Check the mouthpiece for objects before use. Make sure the canister is fully inserted into the actuator.
Priming your ASMANEX HFA Inhaler:
Before you use ASMANEX HFA for the first time, you must prime the inhaler.
-
1. To prime the inhaler, hold it in the upright position away from your face, and press down firmly and fully on the top of the canister until it stops moving in the actuator. Do this 4 times to release a total of 4 actuations (puffs) into the air. -
2. Shake the inhaler well before each of the priming actuations. After priming 4 times, the dose counter should read "120". -
3. If you do not use your ASMANEX HFA for more than 5 days, you will need to prime it again before use.
Using your ASMANEX HFA:
-
4. Remove the cap from the mouthpiece of the actuator (see Figure 3). Check the mouthpiece for objects before use. Make sure the canister is fully inserted into the actuator. -
5. Shake the inhaler well before each use. -
6. Breathe out as fully as you comfortably can through your mouth. Push out as much air from your lungs as possible. Hold the inhaler in the upright position and place the mouthpiece into your mouth (see Figure 4). Close your lips around the mouthpiece.
|
|
|
| Figure 3 | Figure 4 |
-
7. Take a deep breath (inhale) in slowly through your mouth. While doing this, press down firmly and fully on the top of the canister until it stops moving in the actuator. Take your finger off the canister. -
8. When you have finished breathing in, hold your breath as long as you comfortably can, up to 10 seconds. Then remove the inhaler from your mouth and breathe out through your nose, while keeping your lips closed. -
9. Wait at least 30 seconds to take your second puff of ASMANEX HFA. -
10. Shake the inhaler well again and repeat steps 6 through 8 to take your second puff of ASMANEX HFA.
After using your ASMANEX HFA inhaler:
-
11. Replace the cap over the mouthpiece right away after use (see Figure 5).
-
12. After you finish taking ASMANEX HFA (2 puffs), rinse your mouth with water.
Reading the counter:
- The dose counter identifies the number of inhalations (puffs) left in your inhaler (see Figure 6).
- The counter will count down each time you release a puff of medicine (either when preparing your ASMANEX HFA inhaler for use or when using the medicine).
When to replace your ASMANEX HFA:
- It is important that you pay attention to the number of inhalations (puffs) left in your ASMANEX HFA inhaler by reading the counter.
- When the counter reads "20", you should refill your prescription or ask your healthcare provider if you need a new prescription for ASMANEX HFA.
- Throw away ASMANEX HFA after the counter reaches "0", indicating that you have used the number of actuations on the product label and box. Your inhaler may not feel empty and it may continue to operate, but you will not get the right amount of medicine if you keep using it.
- Never try to change the numbers on the counter or remove the counter from the actuator.
- Do not use the inhaler after the expiration date.
How to clean your ASMANEX HFA:
The mouthpiece should be cleaned using a dry wipe after every 7 days of use.
Routine cleaning instructions:
- Remove the cap off the mouthpiece. Wipe the inside and outside surfaces of the actuator mouthpiece with a clean, dry, lint-free tissue or cloth. Put the cap back on the mouthpiece after cleaning.
- Do not attempt to unblock the actuator with a sharp object, such as a pin.
- Do not wash or put any parts of your inhaler in water.
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Manufactured for: Merck Sharp & Dohme Corp., a subsidiary of
MERCK & CO., INC., Whitehouse Station, NJ 08889, USA
Manufactured by: 3M Health Care Ltd., Loughborough, United Kingdom.
For patent information: www.merck.com/product/patent/home.html
The trademarks depicted herein are owned by their respective companies.
Copyright © 2014 Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc.
All rights reserved.
Revised: 04/2014
usppi-mk0887-ao-1404r000
PRINCIPAL DISPLAY PANEL - 100 mcg Canister Carton
NDC 0085-4333-01
Asmanex
®
HFA
(mometasone furoate)
Inhalation Aerosol
100 mcg per actuation
For oral inhalation only
Asmanex HFA canister to be used
with Asmanex HFA actuator only.
SHAKE WELL BEFORE USING.
Rx only
120 Metered Actuations
Net Wt. 13g

PRINCIPAL DISPLAY PANEL - 200 mcg Canister Carton
NDC 0085-4334-01
Asmanex
®
HFA
(mometasone furoate)
Inhalation Aerosol
200 mcg per actuation
For oral inhalation only
Asmanex HFA canister to be used
with Asmanex HFA actuator only.
SHAKE WELL BEFORE USING.
Rx only
120 Metered Actuations
Net Wt. 13g

ASMANEX HFAMOMETASONE FUROATE AEROSOL
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
ASMANEX HFAMOMETASONE FUROATE AEROSOL
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||