Atenolol
State of Florida DOH Central Pharmacy
Atenolol Tablets, USP
FULL PRESCRIBING INFORMATION: CONTENTS*
- ATENOLOL DESCRIPTION
- CLINICAL PHARMACOLOGY
- ATENOLOL INDICATIONS AND USAGE
- ATENOLOL CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- ATENOLOL ADVERSE REACTIONS
- OVERDOSAGE
- ATENOLOL DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- Label Image for 25 mg
- Label Image for 50 mg
- Label Image for 100 mg
FULL PRESCRIBING INFORMATION
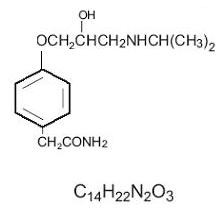
ATENOLOL DESCRIPTION
1
CLINICAL PHARMACOLOGY
12
Pharmacokinetics and Metabolism
2 DOSAGE AND ADMINISTRATION
Pharmacodynamics
121
Atenolol Geriatric Pharmacology
ATENOLOL INDICATIONS AND USAGE
Hypertension
Angina Pectoris Due to Coronary Atherosclerosis
Acute Myocardial Infarction
DOSAGE AND ADMINISTRATION, CONTRAINDICATIONS WARNINGS
ATENOLOL CONTRAINDICATIONS
WARNINGS
WARNINGS
Cardiac Failure
In Patients Without a History of Cardiac Failure
DOSAGE AND ADMNISTRATION
Concomitant Use of Calcium Channel Blockers
PRECAUTIONS
Bronchospastic Diseases
PATIENTS WITH BRONCHOSPASTIC DISEASE SHOULD, IN GENERAL, NOT RECEIVE BETA-BLOCKERS. Because of its relative beta1 selectivity, however, atenolol may be used with caution in patients with bronchospastic disease who do not respond to, or cannot tolerate, other antihypertensive treatment. Since beta1 selectivity is not absolute, the lowest possible dose of atenolol should be used with therapy initiated at 50 mg and a beta2-stimulating agent (bronchodilator) should be made available. If dosage must be increased, dividing the dose should be considered in order to achieve lower peak blood levels.
Anesthesia and Major Surgery
OVERDOSAGE
Diabetes and Hypoglycemia
Thyrotoxicosis
DOSAGE AND ADMINISTRATION
Untreated Pheochromocytoma
Pregnancy and Fetal Injury
PRECAUTIONS, Nursing Mothers
PRECAUTIONS
General
Impaired Renal Function
DOSAGE AND ADMINISTRATIONDrug Interactions
WARNINGS
Carcinogenesis, Mutagenesis, Impairment of Fertility
in vivoS typhimurium
Animal Toxicology
Usage in Pregnancy
WARNINGS - Pregnancy and Fetal Injury
Nursing Mothers
WARNINGS, Pregnancy and Fetal Injury
Pediatric Use
Geriatric Use
Hypertension and Angina Pectoris Due to Coronary Atherosclerosis
Acute Myocardial Infarction
CLINICAL PHARMACOLOGY INDICATIONS AND USAGE
ATENOLOL ADVERSE REACTIONS
| |
Volunteered (U.S. Studies) |
Total - Volunteered and Elicited (Foreign+U.S. Studies) |
||
| Atenolol (n=164) % |
Placebo (n=206) % |
Atenolol (n=399) % |
Placebo (n=407) % |
|
| CARDIOVASCULAR |
|
|
|
|
| Bradycardia |
3 |
0 |
3 |
0 |
| Cold Extremities |
0 |
0.5 |
12 |
5 |
| Postural Hypotension |
2 |
1 |
4 |
5 |
| Leg Pain |
0 |
0.5 |
3 |
1 |
| CENTRAL NERVOUS SYSTEM/NEUROMUSCULAR |
|
|
|
|
| Dizziness |
4 |
1 |
13 |
6 |
| Vertigo |
2 |
0.5 |
2 |
0.2 |
| Lightheadedness |
1 |
0 |
3 |
0.7 |
| Tiredness |
0.6 |
0.5 |
26 |
13 |
| Fatigue |
3 |
1 |
6 |
5 |
| Lethargy |
1 |
0 |
3 |
0.7 |
| Drowsiness |
0.6 |
0 |
2 |
0.5 |
| Depression |
0.6 |
0.5 |
12 |
9 |
| Dreaming |
0 |
0 |
3 |
1 |
| GASTROINTESTINAL |
|
|
|
|
| Diarrhea |
2 |
0 |
3 |
2 |
| Nausea |
4 |
1 |
3 |
1 |
| RESPIRATORY (see
WARNINGS
) |
|
|
|
|
| Wheeziness |
0 |
0 |
3 |
3 |
| Dyspnea |
0.6 |
1 |
6 |
4 |
Acute Myocardial Infarction
| |
Conventional Therapy Plus Atenolol (n=244) |
Conventional Therapy Alone (n=233) |
||
| Bradycardia |
43 |
(18%) |
24 |
(10%) |
| Hypotension |
60 |
(25%) |
34 |
(15%) |
| Bronchospasm |
3 |
(1.2%) |
2 |
(0.9%) |
| Heart Failure |
46 |
(19%) |
56 |
(24%) |
| Heart Block |
11 |
(4.5%) |
10 |
(4.3%) |
| BBB + Major Axis Deviation |
16 |
(6.6%) |
28 |
(12%) |
| Supraventricular Tachycardia |
28 |
(11.5%) |
45 |
(19%) |
| Atrial Fibrillation |
12 |
(5%) |
29 |
(11%) |
| Atrial Flutter |
4 |
(1.6%) |
7 |
(3%) |
| Ventricular Tachycardia |
39 |
(16%) |
52 |
(22%) |
| Cardiac Reinfarction |
0 |
(0%) |
6 |
(2.6%) |
| Total Cardiac Arrests |
4 |
(1.6%) |
16 |
(6.9%) |
| Nonfatal Cardiac Arrests |
4 |
(1.6%) |
12 |
(5.1%) |
| Deaths |
7 |
(2.9%) |
16 |
(6.9%) |
| Cardiogenic Shock |
1 |
(0.4%) |
4 |
(1.7%) |
| Development of Ventricular Septal Defect |
0 |
(0%) |
2 |
(0.9%) |
| Development of Mitral Regurgitation |
0 |
(0%) |
2 |
(0.9%) |
| Renal Failure |
1 |
(0.4%) |
0 |
(0%) |
| Pulmonary Emboli |
3 |
(1.2%) |
0 |
(0%) |
| *Full dosage was 10 mg and some patients received less than 10 mg but more than 5 mg. |
||||
|
|
Reasons for Reduced Dosage
|
|||
| IV Atenolol Reduced Dose (< 5 mg)* |
Oral Partial Dose |
|||
| Hypotension/Bradycardia |
105 |
(1.3%) |
1168 |
(14.5%) |
| Cardiogenic Shock |
4 |
(.04%) |
35 |
(.44%) |
| Reinfarction |
0 |
(0%) |
5 |
(.06%) |
| Cardiac Arrest |
5 |
(.06%) |
28 |
(.34%) |
| Heart Block (> first degree) |
5 |
(.06%) |
143 |
(1.7%) |
| Cardiac Failure |
1 |
(.01%) |
233 |
(2.9%) |
| Arrhythmias |
3 |
(.04%) |
22 |
(.27%) |
| Bronchospasm |
1 |
(.01%) |
50 |
(.62%) |
POTENTIAL ADVERSE EFFECTS
Hematologic:
Allergic:
Central Nervous System:
Gastrointestinal:
Other:
Miscellaneous: DOSAGE AND ADMINISTRATION
OVERDOSAGE
2
ATENOLOL DOSAGE AND ADMINISTRATION
Hypertension
Angina Pectoris
Acute Myocardial Infarction
Elderly Patients or Patients with Renal Impairment
2
| Creatinine Clearance (mL/min/1.73 m2) |
Atenolol Elimination Half-Life (h) |
Maximum Dosage |
| 15-35 |
16-27 |
50 mg daily |
| <15 |
>27 |
25 mg daily |
Cessation of Therapy in Patients with Angina Pectoris
HOW SUPPLIED
Atenolol Tablets USP, 25 mg
Atenolol Tablets USP, 50 mg
Atenolol Tablets USP, 100 mg
They are supplied by State of Florida DOH Central Pharmacy as follows:
| NDC | Strength | Quantity/Form | Color | Source Prod. Code |
| 53808-0658-1 | 25 mg | 30 Tablets in a Blister Pack | White to Off-white | 65862-168 |
| 53808-0900-1 | 50 mg | 30 Tablets in a Blister Pack | White to Off-white | 65862-169 |
| 53808-0660-1 | 100 mg | 30 Tablets in a Blister Pack | White to Off-white | 65862-170 |
Store at
Aurobindo Pharma USA, Inc.
Aurobindo Pharma Limited
This Product was Repackaged By:
State of Florida DOH Central Pharmacy
104-2 Hamilton Park Drive
Tallahassee, FL 32304
United States
Label Image for 25 mg
NDC 53808-0658-1
Atenolol Tablets, USP
25 mg
Rx only 30 Tablets
AUROBINDO
_97ec87b2.jpg)
Label Image for 50 mg
NDC 53808-0900-1
Atenolol Tablets, USP
50 mg
Rx only 30 Tablets
AUROBINDO
_97ec87b2.jpg)
Label Image for 100 mg
NDC 53808-0660-1
Atenolol Tablets, USP
100 mg
Rx only 30 Tablets
AUROBINDO
_97ec87b2.jpg)
AtenololAtenolol TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
AtenololAtenolol TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
AtenololAtenolol TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!