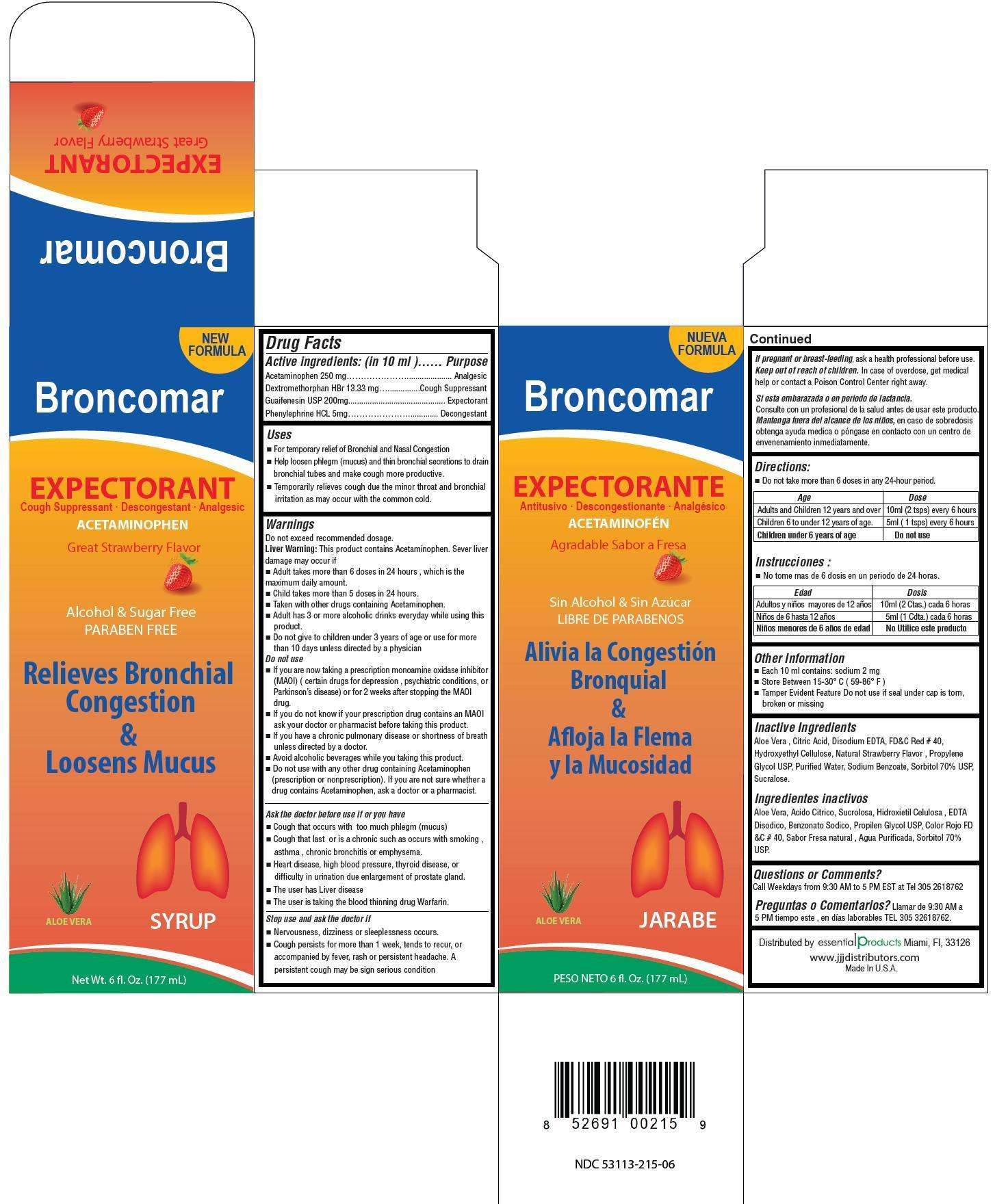
Broncomar
FULL PRESCRIBING INFORMATION
Active ingredient
Acvtive ingredients: (in 10 ml) Purpose
Acetaminophen 200 mg ............................. Analgesic
Dextromethorphan HBr 13.33 mg ................... Cough Suppressant
Guaifenesin 200 mg ................................... Expectorant
Phenylephrine HCl 5 mg ............................. Decongestant
Purpose
Purpose
Analgesic
Cough Suppressant
Expectorant
Decongestant
Uses
Uses
- For temporary relief of bronchial and nasal congestion
- Help loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes and make cough more productive.
- Teporarily relieves cough due to minor throat and bronchial irritation as may occur with the common cold
Warnings
Do not exceed recommended dosage
Liver warning: This product contains Acetaminophen. Sever liver damage may occur if
- Adult takes more than 6 doses in 24 hours, which is the maximum daily amount
- Child takes more than 5 doses in 24 hours
- Taken with other drugs containing Acetaminophen
- Adult has 3 or more alcoholic drinks everyday while using this product
- Do not give to children under 3 years of age or use for more than 10 days unless directed by a physician
Do not use
- If you are now taking a prescription monoamine oxidase inhibitor (MAOI)( certain drugs for depression , psychiatric conditions, or Parkinson’s disease) or for 2 weeks after stopping the MAOI drug.
- if you do not know if your prescription drug contains on MAOI ask your doctor or pharmacist before taking this product
- if you have a chronic pulmonary disease or shortness of breath unless directed by a doctor
- avoid alcoholic beverages while you are taking this product
- do not use with any other drug containing Acetaminophen (prescirption or nonprescription). If you are not sure whether a drug contains Acetaminophen, ask a doctor or a pharmacist.
Ask the doctor before use if you have
- cough that occurs with too much phlegm (mucus)
- cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis or emphysema
- heart disease, high blood pressure, thyroid disease, or difficulty in urination due to the enlargement of prostate gland.
- The user has liver disease
- The user is taking the blood thinning drug Warfarin.
Stop use and ask the doctor if
- Nervoisness, dizziness or sleeplessness occurs.
- Cough persists for more than 1 week, tends to recur, or accompanied by fever, rash or persistent headache. A persistent cough may be sign of a serious condition
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- Do not take more than 6 doses in any 24-hour period
Age Dose
Adults and children 12 years and over 10 mL (2 tsps) every 6 hours
Children 6 to under 12 years of age. 5 mL (1 tsps) every 6 hours
Children under 6 years of age Do not use
Inactive ingredients
Aloe vera, citric acid, disodium EDTA, FD&C Red #40, hydroxyethyl cellulose, natural strawberry flavor, propylene glycol, water, sodium benzoate, sorbitol 70%, sucralose
BroncomarAcetaminophen, Dextromethorphan HBr, Guaifenesin, Phenylephrine HCl LIQUID
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||