Carbamazepine
FULL PRESCRIBING INFORMATION: CONTENTS*
- BOXED WARNING
- CARBAMAZEPINE DESCRIPTION
- CLINICAL PHARMACOLOGY
- PHARMACOKINETICS
- INDICATIONS & USAGE
- CARBAMAZEPINE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- INFORMATION FOR PATIENTS
- LABORATORY TESTS
- DRUG INTERACTIONS
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- PREGNANCY
- LABOR & DELIVERY
- NURSING MOTHERS
- PEDIATRIC USE
- GERIATRIC USE
- CARBAMAZEPINE ADVERSE REACTIONS
- DRUG ABUSE AND DEPENDENCE
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- INACTIVE INGREDIENT
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
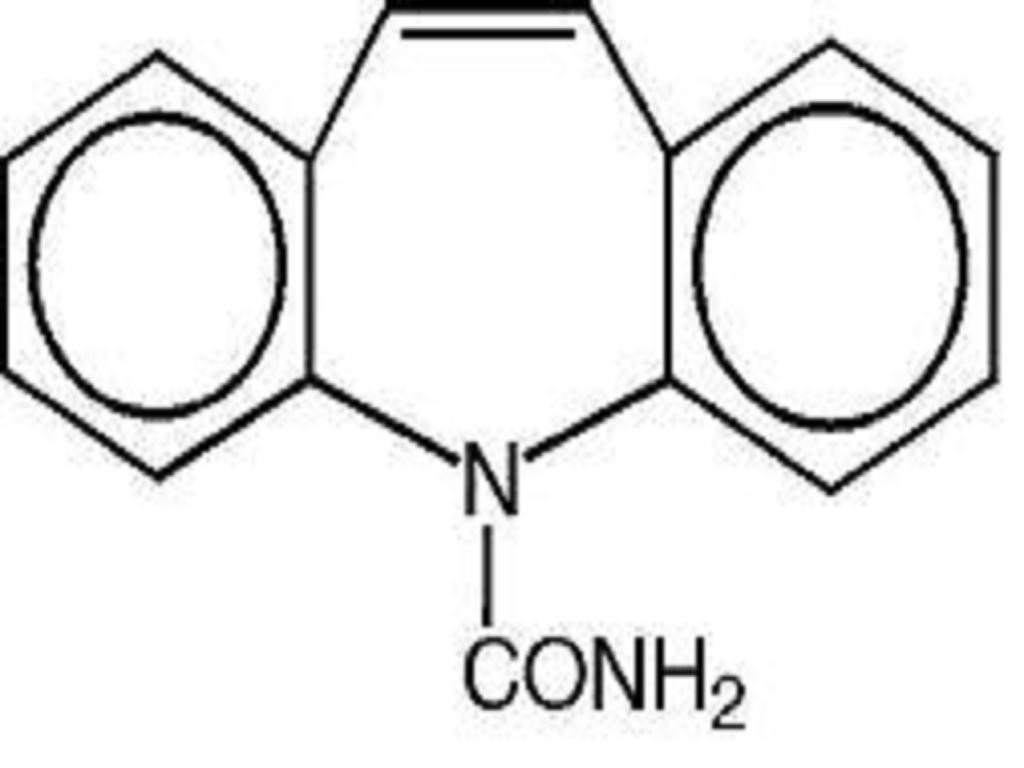
CARBAMAZEPINE DESCRIPTION
CLINICAL PHARMACOLOGY
Mechanism of Action
PHARMACOKINETICS
INDICATIONS & USAGE
EpilepsyTrigeminal Neuralgia
CARBAMAZEPINE CONTRAINDICATIONS
WARNINGS
Serious Dermatologic ReactionsSJS/TEN and HLA-B*1502 Allele
Aplastic Anemia and Agranulocytosis
Suicidal Behavior and Ideation
General
Usage in Pregnancy
PRECAUTIONS
GeneralINFORMATION FOR PATIENTS
LABORATORY TESTS
DRUG INTERACTIONS
Agents That May Affect Carbamazepine Plasma Levels
Effect of Carbamazepine on Plasma Levels of Concomitant Agents
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
PREGNANCY
Teratogenic EffectsPregnancy category D
LABOR & DELIVERY
NURSING MOTHERS
PEDIATRIC USE
GERIATRIC USE
CARBAMAZEPINE ADVERSE REACTIONS
DRUG ABUSE AND DEPENDENCE
OVERDOSAGE
Acute ToxicitySigns and Symptoms
Treatment
DOSAGE & ADMINISTRATION
Epilepsy
Adults and Children Over 12 Years of Age
Children 6 to 12 Years of Age
Children Under 6 Years of Age
Combination Therapy
Trigeminal Neuralgia
*
*
HOW SUPPLIED
INACTIVE INGREDIENT
COLLOIDAL SILICON DIOXIDECROSCARMELLOSE SODIUM
ETHYLCELLULOSE
GLYCERIN
LACTOSE MONOHYDRATE
MAGNESIUM STEARATE
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION

CarbamazepineCarbamazepine TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!