Carbaphen Ped CH
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient (in each 1 mL)
- Purpose
- Carbaphen Ped CH Uses
- Warnings
- Directions
- Carbaphen Ped CH Other information
- Inactive ingredients
- Questions?
- Package/Label Principal Display Panel
FULL PRESCRIBING INFORMATION
Active ingredient (in each 1 mL)
Chlophedianol HCl 8 mg
Chlorpheniramine Maleate 1.25 mg
Phenylephrine HCl 2.5 mg
Purpose
Antitussive
Antihistamine
Nasal Decongestant
Carbaphen Ped CH Uses
Temporarily relieves:
- cough due to the common cold.
- runny nose and sneezing, itching of the nose or throat, and itchy, watery eyes due to hay fever or other upper respiratory allergies.
- nasal congestion due to the common cold.
Warnings
Do not use
- in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your child's prescription drug contains an MAOI, ask a doctor or pharmacist before giving this product.
Ask a doctor before use if you have
- asthma
- emphysema
- glaucoma
- diabetes
- excessive phlegm (mucus)
- breathing problems
- chronic bronchitis
- persistent or chronic cough
- cough associated with smoking
- trouble urinating due to enlarged prostate gland
- a sodium-restricted diet
- thyroid disorders
- heart disease
- high blood pressure
Ask a doctor or pharmacist before use if you are
- taking sedatives or tranquilizers
When using this product
- do not use more than directed
- excitability may occur, especially in children
- marked drowsiness may occur
- avoid alcoholic drinks
- be careful when driving a motor vehicle or operating machinery
- alcohol, sedatives, and tranquilizers may increase drowsiness
Stop use and ask a doctor if
- cough lasts more than 7 days, comes back, or occurs with fever, rash, headache that lasts, or sleeplessness. These could be signs of a serious condition.
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- use 5 mL dosing syringe
- do not exceed 4 doses in 24 hours
| Children 6 to 12 years of age: | 1 to 1.5 mL every 6 to 8 hours. |
| Children under 6 years of age: | Consult a doctor. |
Carbaphen Ped CH Other information
- store at room temperature
Inactive ingredients
banana flavor, benzoic acid, blueberry flavor, citric acid, ethyl alcohol, glycerin, methylparaben, natural masking agent, propylene glycol, propylparaben, sodium citrate, sorbital, sucralose, water, and xanthan gum.
Questions?
Call Gil Pharmaceuticals Corp. at 787-848-9114, Mon.-Fri. 8:00 am to 5:00 pm, EST.
Package/Label Principal Display Panel
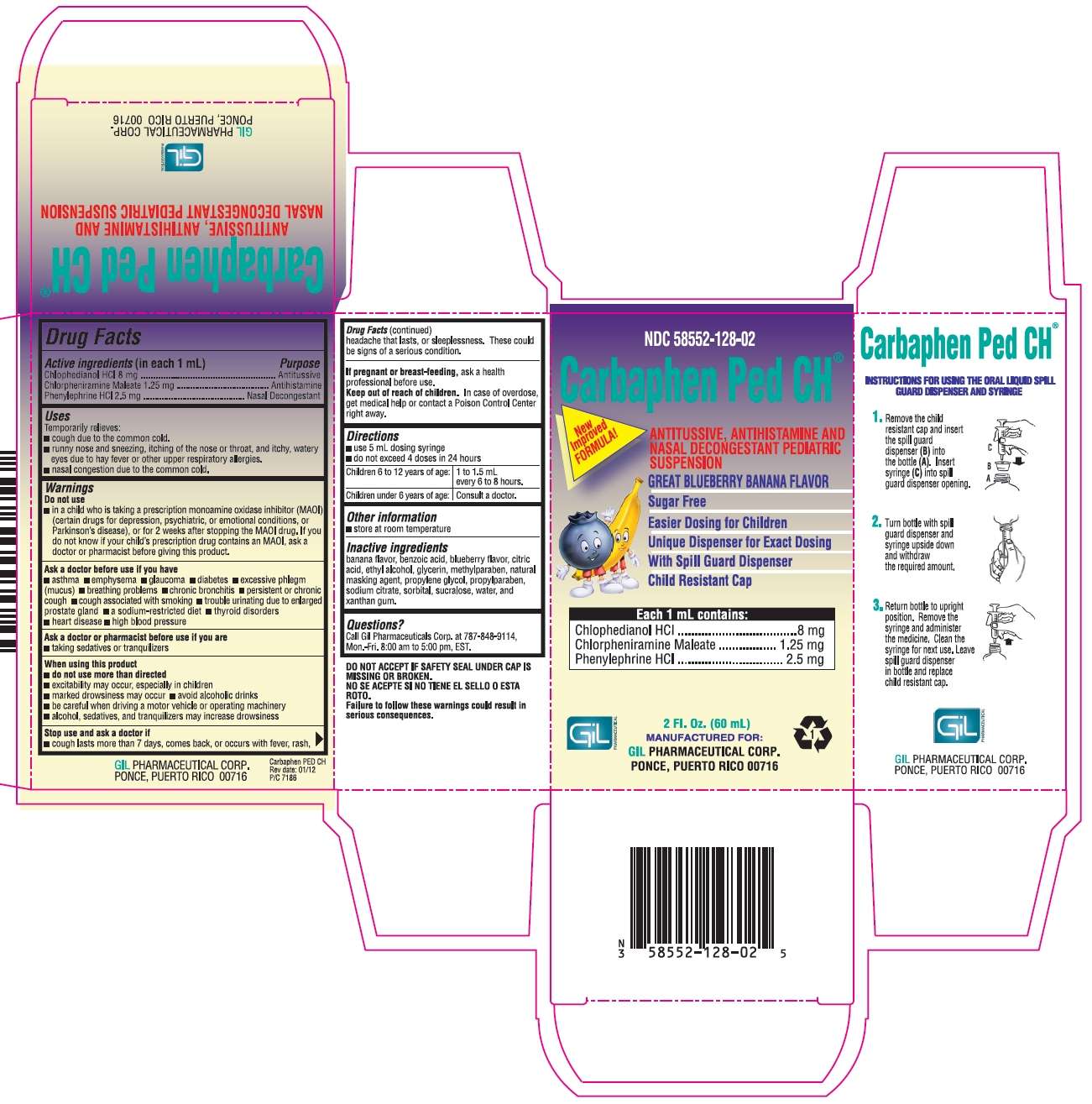
NDC 58552-128-02
Carbapen Ped CH®
New Improved FORMULA!
ANTITUSSIVE, ANTIHISTAMINE AND NASAL DECONGESTANT PEDIATRIC SUSPENSION
GREAT BLUEBERRY BANANA FLAVOR
Sugar Free
Easier Dosing for Children
Unique Dispenser for Exact Dosing
With Spill Guard Dispenser
Child Resistant Cap
Each 1 mL contains:
Chlophedianol HCl...............................8 mg
Chlorpheniramine Maleate...............1.25 mg
Phenylephrine HCl.............................2.5 mg
2 Fl. Oz. (60 mL)
MANUFACTURED FOR:
GIL PHARMACEUTICAL CORP.
PONCE, PUERTO RICO 00716
Carbaphen Ped CHChlophedianol HCL, Chlorpheniramine Maleate and Phenylephrine HCl SUSPENSION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||