Carbidopa, levodopa and entacapone
Carbidopa, Levodopa and Entacapone Tablets
FULL PRESCRIBING INFORMATION: CONTENTS*
- CARBIDOPA, LEVODOPA AND ENTACAPONE DESCRIPTION
- CLINICAL PHARMACOLOGY
- INDICATIONS
- CARBIDOPA, LEVODOPA AND ENTACAPONE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- General
- Hypotension/Syncope
- Diarrhea and Colitis
- Hallucinations
- Dyskinesia
- Other Events Reported With Dopaminergic Therapy
- Renal Toxicity
- Hepatic Impairment
- Biliary Obstruction
- Information for Patients
- Laboratory Tests
- Drug Interactions
- Carcinogenesis
- Mutagenesis
- Impairment of Fertility
- Pregnancy
- Nursing Women
- Pediatric Use
- CARBIDOPA, LEVODOPA AND ENTACAPONE ADVERSE REACTIONS
- DRUG ABUSE AND DEPENDENCE
- OVERDOSAGE
- CARBIDOPA, LEVODOPA AND ENTACAPONE DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- PRINCIPAL DISPLAY PANEL-Label-12.5 mg/50 mg/200 mg
- PRINCIPAL DISPLAY PANEL-Label-18.75 mg/75 mg/200 mg
- PRINCIPAL DISPLAY PANEL-Label-25 mg/100 mg/200 mg
- PRINCIPAL DISPLAY PANEL-Label-31.25 mg/125 mg/200 mg
- PRINCIPAL DISPLAY PANEL-Label-37.5 mg/150 mg/200 mg
- PRINCIPAL DISPLAY PANEL-Label-50 mg/200 mg/200 mg
FULL PRESCRIBING INFORMATION
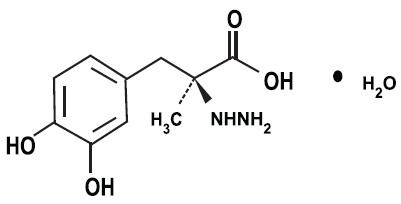
CARBIDOPA, LEVODOPA AND ENTACAPONE DESCRIPTION
1014242
9114

O141535
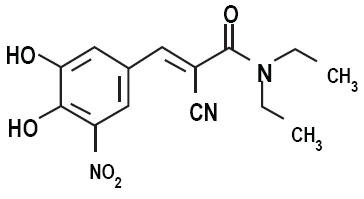
Carbidopa, levodopa and entacapone is supplied as tablets in six strengths:
Carbidopa, levodopa and entacapone tablets 12.5 mg/50 mg/200 mg, containing 12.5 mg of carbidopa, 50 mg of levodopa and 200 mg of entacapone; carbidopa, levodopa and entacapone tablets 18.75 mg/75 mg/200 mg, containing 18.75 mg of carbidopa, 75 mg of levodopa and 200 mg of entacapone; carbidopa, levodopa and entacapone tablets 25 mg/100 mg/200 mg, containing 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone; carbidopa, levodopa and entacapone tablets 31.25 mg/125 mg/200 mg, containing 31.25 mg of carbidopa, 125 mg of levodopa and 200 mg of entacapone; carbidopa, levodopa and entacapone tablets 37.5 mg/150 mg/200 mg, containing 37.5 mg of carbidopa, 150 mg of levodopa and 200 mg of entacapone; carbidopa, levodopa and entacapone tablets 50 mg/200 mg/200 mg, containing 50 mg of carbidopa, 200 mg of levodopa and 200 mg of entacapone.
The inactive ingredients of the carbidopa, levodopa and entacapone tablets are corn starch, croscarmellose sodium, glycerol 85%, hypromellose, magnesium stearate, mannitol, polysorbate 80, povidone, sucrose, red iron oxide, and titanium dioxide. Carbidopa, levodopa and entacapone tablets 12.5 mg/50 mg/200 mg, 25 mg/100 mg/200 mg and 37.5 mg/150 mg/200 mg also contain yellow iron oxide.
CLINICAL PHARMACOLOGY
Mechanism of Action
Levodopa
Current evidence indicates that symptoms of Parkinson's disease are related to depletion of dopamine in the corpus striatum. Administration of dopamine is ineffective in the treatment of Parkinson's disease apparently because it does not cross the blood-brain barrier. However, levodopa, the metabolic precursor of dopamine, does cross the blood-brain barrier, and presumably is converted to dopamine in the brain. This is thought to be the mechanism whereby levodopa relieves symptoms of Parkinson's disease.
Carbidopa
When levodopa is administered orally it is rapidly decarboxylated to dopamine in extracerebral tissues so that only a small portion of a given dose is transported unchanged to the central nervous system. Carbidopa inhibits the decarboxylation of peripheral levodopa, making more levodopa available for transport to the brain. When coadministered with levodopa, carbidopa increases plasma levels of levodopa and reduces the amount of levodopa required to produce a given response by about 75 %. Carbidopa prolongs the plasma half-life of levodopa from 50 minutes to 1.5 hours and decreases plasma and urinary dopamine and its major metabolite, homovanillic acid. The Tmax of levodopa, however, was unaffected by the coadministration.
Entacapone
Entacapone is a selective and reversible inhibitor of catechol-O-methyltransferase (COMT).
In mammals, COMT is distributed throughout various organs with the highest activities in the liver and kidney. COMT also occurs in neuronal tissues, especially in glial cells. COMT catalyzes the transfer of the methyl group of S-adenosyl-L-methionine to the phenolic group of substrates that contain a catechol structure. Physiological substrates of COMT include DOPA, catecholamines (dopamine, norepinephrine, and epinephrine) and their hydroxylated metabolites. The function of COMT is the elimination of biologically active catechols and some other hydroxylated metabolites. When decarboxylation of levodopa is prevented by carbidopa, COMT becomes the major metabolizing enzyme for levodopa, catalyzing its metabolism to 3-methoxy-4-hydroxy-L-phenylalanine (3-OMD).
When entacapone is given in conjunction with levodopa and carbidopa, plasma levels of levodopa are greater and more sustained than after administration of levodopa and carbidopa alone. It is believed that at a given frequency of levodopa administration, these more sustained plasma levels of levodopa result in more constant dopaminergic stimulation in the brain, leading to greater effects on the signs and symptoms of Parkinson’s disease. The higher levodopa levels may also lead to increased levodopa adverse effects, sometimes requiring a decrease in the dose of levodopa.
When 200 mg entacapone is coadministered with levodopa/carbidopa, it increases levodopa plasma exposure (AUC) by 35% to 40% and prolongs its elimination half-life in Parkinson’s disease patients from 1.3 hours to 2.4 hours. Plasma levels of the major COMT-mediated dopamine metabolite, 3-methoxy-4-hydroxy-L-phenylalanine (3-OMD), are also markedly decreased proportionally with increasing dose of entacapone.
Pharmacokinetics
The pharmacokinetics of carbidopa, levodopa and entacapone tablets have been studied in healthy subjects (age 45 years old to 75 years old). Overall, following administration of corresponding doses of levodopa, carbidopa and entacapone as carbidopa, levodopa and entacapone tablets or as carbidopa/levodopa product plus Comtan®* (entacapone) tablets, the mean plasma concentrations of levodopa, carbidopa, and entacapone are comparable.
Absorption/Distribution:
Both levodopa and entacapone are rapidly absorbed and eliminated, and their distribution volume is moderately small. Carbidopa is absorbed and eliminated slightly more slowly compared with levodopa and entacapone. There are substantial inter- and intra-individual variations in the absorption of levodopa, carbidopa and entacapone, particularly concerning its Cmax.
The food-effect on the carbidopa, levodopa and entacapone tablet has not been evaluated.
Levodopa
|
Tablet Strength
|
AUC0-
∞
(nanogram·h/mL) |
Cmax
(nanogram/mL) |
Tmax
(h) |
| 12.5 mg/50 mg/200 mg
|
1,040 ± 314 |
470 ± 154 |
1.1 ± 0.5 |
| 25 mg/100 mg/200 mg |
2,910 ± 715 |
975 ± 247 |
1.4 ± 0.6 |
| 37.5 mg/150 mg/200 mg |
3,770 ± 1,120 |
1,270 ± 329 |
1.5 ± 0.9 |
| 50 mg/200 mg/200 mg |
6,115 ± 1,536 |
1,859 ± 455 |
1.76 ± 0.7 |
Since levodopa competes with certain amino acids for transport across the gut wall, the absorption of levodopa may be impaired in some patients on a high protein diet. Meals rich in large neutral amino acids may delay and reduce the absorption of levodopa (see PRECAUTIONS).
Levodopa is bound to plasma protein only to a minor extent (about 10% to 30%).
Carbidopa
Following administration of carbidopa, levodopa and entacapone tablet as a single dose to healthy male and female subjects, the peak concentration of carbidopa was reached within 2.5 hours to 3.4 hours on average. The mean Cmax ranged from about 40 nanogram/mL to 225 nanogram/mL and the mean AUC from 170 nanogram•h/mL to 1,200 nanogram•h/mL, with different carbidopa, levodopa and entacapone tablet strengths providing 12.5 mg, 25 mg, 37.5 mg or 50 mg of carbidopa.
Carbidopa is approximately 36% bound to plasma protein.
Entacapone
max
Metabolism and Elimination:
Levodopa
The elimination half-life of levodopa, the active moiety of antiparkinsonian activity, was
1.7 hours (range 1.1 hours to 3.2 hours).
Levodopa is extensively metabolized to various metabolites. Two major pathways are decarboxylation by dopa decarboxylase (DDC) and O-methylation by catechol-O-methyltransferase (COMT).
Carbidopa
The elimination half-life of carbidopa was on average 1.6 hours to 2 hours (range 0.7 hours to 4.0 hours).
Carbidopa is metabolized to two main metabolites (α-methyl-3-methoxy-4-hydroxyphenylpropionic acid and α-methyl-3,4-dihydroxyphenylpropionic acid). These 2 metabolites are primarily eliminated in the urine unchanged or as glucuronide conjugates. Unchanged carbidopa accounts for 30% of the total urinary excretion.
Entacapone
The elimination half-life of entacapone was on average 0.8 hour to 1 hour (0.3 hour to 4.5 hours).
Entacapone is almost completely metabolized prior to excretion with only a very small amount (0.2% of dose) found unchanged in urine. The main metabolic pathway is isomerization to the cis-isomer, the only active metabolite. Entacapone and the cis-isomer are eliminated in the urine as glucuronide conjugates. The glucuronides account for 95% of all urinary metabolites (70% as parent and 25% as cis-isomer glucuronides). The glucuronide conjugate of the cis-isomer is inactive. After oral administration of a 14C-labeled dose of entacapone, 10% of labeled parent and metabolite is excreted in urine and 90% in feces.
Special Populations:
Hepatic Impairment :
Carbidopa, levodopa and entacapone
While there are no studies on the pharmacokinetics of carbidopa and levodopa in patients with hepatic impairment, carbidopa, levodopa and entacapone tablets should be administered cautiously to patients with biliary obstruction or hepatic disease since biliary excretion appears to be the major route of excretion of entacapone and hepatic impairment had a significant effect on the pharmacokinetics of entacapone when 200 mg entacapone was administered alone.
Entacapone
Hepatic impairment had a significant effect on the pharmacokinetics of entacapone when 200 mg entacapone was administered alone. A single 200 mg dose of entacapone, without levodopa/dopa decarboxylase inhibitor coadministration, showed approximately two-fold higher AUC and Cmax values in patients with a history of alcoholism and hepatic impairment (n=10) compared to normal subjects (n=10). All patients had biopsy-proven liver cirrhosis caused by alcohol. According to Child-Pugh grading 7 patients with liver disease had mild hepatic impairment and 3 patients had moderate hepatic impairment. As only about 10% of the entacapone dose is excreted in urine, as parent compound and conjugated glucuronide, biliary excretion appears to be the major route of excretion of this drug. Consequently, carbidopa, levodopa and entacapone tablets should be administered with care to patients with biliary obstruction or hepatic disease.
Renal Impairment :
Carbidopa, levodopa and entacapone
Carbidopa, levodopa and entacapone tablets should be administered cautiously to patients with severe renal disease. There are no studies on the pharmacokinetics of levodopa and carbidopa in patients with renal impairment.
Entacapone
No important effects of renal function on the pharmacokinetics of entacapone were found. The pharmacokinetics of entacapone have been investigated after a single 200 mg entacapone dose, without levodopa/dopa decarboxylase inhibitor coadministration, in a specific renal impairment study. There were three groups: normal subjects (n=7; creatinine clearance greater than 1.12 mL/sec/1.73 m2), moderate impairment (n=10; creatinine clearance ranging from 0.60 mL/sec/1.73 m2 to 0.89 mL/sec/1.73 m2), and severe impairment (n=7; creatinine clearance ranging from 0.20 mL/sec/1.73 m2 to 0.44 mL/sec/1.73 m2).
Concurrent Diseases:
Carbidopa, levodopa and entacapone tablets should be administered cautiously to patients with biliary obstruction, hepatic disease, severe cardiovascular or pulmonary disease, bronchial asthma, renal, or endocrine disease.
Elderly :
Carbidopa, levodopa and entacapone tablets have not been studied in Parkinson’s disease patients or in healthy volunteers older than 75 years old. In the pharmacokinetics studies conducted in healthy volunteers following single dose of carbidopa/levodopa/entacapone (as carbidopa, levodopa and entacapone tablets or as separate carbidopa/levodopa and Comtan* tablets):
Levodopa
The AUC of levodopa is significantly (on average 10% to 20%) higher in elderly (60 years to 75 years) than younger subjects (45 years to 60 years). There is no significant difference in the Cmax of levodopa between younger (45 years to 60 years) and elderly subjects (60 years to 75 years).
Carbidopa
There is no significant difference in the Cmax and AUC of carbidopa, between younger (45 years to 60 years) and elderly subjects (60 years to 75 years).
Entacapone
The AUC of entacapone is significantly (on average, 15%) higher in elderly (60 years to 75 years) than younger subjects (45 years to 60 years). There is no significant difference in the Cmax of entacapone between younger (45 years to 60 years) and elderly subjects (60 years to 75 years).
Gender:
The bioavailability of levodopa is significantly higher in females when given with or without carbidopa and/or entacapone. Following a single dose of carbidopa, levodopa and entacapone together, either as carbidopa, levodopa and entacapone tablets or as separate carbidopa/levodopa and Comtan* tablets in healthy volunteers (age range 45 years to 74 years):
Levodopa
The plasma exposure (AUC and Cmax) of levodopa is significantly higher in females than males (on average, 40% for AUC and 30% for Cmax). These differences are primarily explained by body weight. Other published literature showed significant gender effect (higher concentrations in females) even after correction for body weight.
Carbidopa:
There is no gender difference in the pharmacokinetics of carbidopa.
Entacapone:
Drug Interactions: See PRECAUTIONS, Drug Interactions
Clinical Studies
Each carbidopa, levodopa and entacapone tablet, provided in six single-dose strengths, contains carbidopa and levodopa in ratio 1:4 and a 200 mg dose of entacapone. Four carbidopa, levodopa and entacapone tablet strengths 12.5 mg/50 mg/200 mg, 25 mg/100 mg/200 mg, 37.5 mg/150 mg/200 mg and 50 mg/200 mg/200 mg have been shown to be bioequivalent to the corresponding doses of standard-release carbidopa/levodopa 25 mg/100 mg tablets and Comtan* 200 mg tablets.
The effectiveness of entacapone as an adjunct to levodopa in the treatment of Parkinson’s disease was established in three 24-week multicenter, randomized, double-blind placebo-controlled trials in patients with Parkinson’s disease. In two of these trials, the patients’ disease was “fluctuating”, i.e., was characterized by documented periods of “On” (periods of relatively good functioning) and “Off” (periods of relatively poor functioning), despite optimum levodopa therapy. There was also a withdrawal period following 6 months of treatment. In the third trial patients were not required to have been experiencing fluctuations. Prior to the controlled part of these trials, patients were stabilized on levodopa for 2 weeks to 4 weeks.
There is limited experience of using entacapone in patients who do not experience fluctuations.
In the first two studies to be described, patients were randomized to receive placebo or entacapone 200 mg administered concomitantly with each dose of carbidopa-levodopa (up to 10 times daily, but averaging 4 doses to 6 doses per day). The formal double-blind portion of both trials was 6 months long. Patients recorded the time spent in the “On” and “Off” states in home diaries periodically throughout the duration of the trial. In one study, conducted in the Nordic countries, the primary outcome measure was the total mean time spent in the “On” state during an 18-hour diary recorded day (6 a.m. to midnight). In the other study, the primary outcome measure was the proportion of awake time spent over 24 hours in the “On” state.
In addition to the primary outcome measure, the amount of time spent in the “Off” state was evaluated, and patients were also evaluated by subparts of the Unified Parkinson’s Disease Rating Scale (UPDRS), a frequently used multi-item rating scale intended to assess mentation (Part I), activities of daily living (Part II), motor function (Part III), complications of therapy (Part IV), and disease staging (Part V and VI); an investigator’s and patient’s global assessment of clinical condition, a 7-point subjective scale designed to assess global functioning in Parkinson’s disease; and the change in daily carbidopa-levodopa dose.
In one of the studies, 171 patients were randomized in 16 centers in Finland, Norway, Sweden, and Denmark (Nordic study), all of whom received concomitant levodopa plus dopa-decarboxylase inhibitor (either carbidopa-levodopa or benserazide-levodopa). In the second trial, 205 patients were randomized in 17 centers in North America (US and Canada); all patients received concomitant carbidopa-levodopa.
|
Primary Measure from Home Diary (from an 18-hour Diary Day)
|
|||
| Baseline |
Change from Baseline at Month 6 |
p-value vs. placebo |
|
|
Hours of Awake Time “On”
|
|
||
| Placebo |
9.2 |
+0.1 |
– |
| Entacapone |
9.3 |
+1.5 |
<0.001 |
|
Duration of “On” Time After First AM Dose (Hrs)
|
|
||
| Placebo |
2.2 |
0.0 |
– |
| Entacapone |
2.1 |
+0.2 |
<0.05 |
|
Secondary Measures from Home Diary (from an 18-hour Diary Day)
|
|||
|
Hours of Awake Time “Off”
|
|
|
|
| Placebo |
5.3 |
0.0 |
– |
| Entacapone |
5.5 |
- 1.3 |
<0.001 |
Proportion of Awake Time “On” |
|||
| Placebo |
63.8 |
+0.6 |
– |
| Entacapone |
62.7 |
+9.3 |
<0.001 |
|
Levodopa Total Daily Dose (mg)
|
|||
| Placebo |
705 |
+14 |
– |
| Entacapone |
701 |
- 87 |
<0.001 |
|
Frequency of Levodopa Daily Intakes
|
|||
| Placebo |
6.1 |
+0.1 |
– |
| Entacapone |
6.2 |
- 0.4 |
<0.001 |
|
Other Secondary Measures
|
|||
| Baseline |
Change from Baseline at Month 6 |
p-value vs. placebo |
|
Investigator’s Global (overall) % Improved |
|
|
|
| Placebo |
– |
28 |
– |
| Entacapone |
– |
56 |
<0.01 |
Patient’s Global (overall) % Improved |
|||
| Placebo |
– |
22 |
– |
| Entacapone |
– |
39 |
N.S. |
|
UPDRS Total
|
|
|
|
| Placebo |
37.4 |
-1.1 |
– |
| Entacapone |
38.5 |
-4.8 |
<0.01 |
|
UPDRS Motor
|
|||
| Placebo |
24.6 |
-0.7 |
– |
| Entacapone |
25.5 |
-3.3 |
<0.05 |
|
UPDRS ADL
|
|||
| Placebo |
11.0 |
-0.4 |
– |
| Entacapone |
11.2 |
-1.8 |
<0.05 |
|
Primary Measure from Home Diary (for a 24-hour Diary Day)
|
|||
| Baseline |
Change from Baseline at Month 6 |
p-value vs. placebo |
|
|
Percent of Awake Time “On”
|
|
||
| Placebo |
60.8 |
+2.0 |
– |
| Entacapone |
60.0 |
+6.7 |
<0.05 |
|
Secondary Measures from Home Diary (for a 24-hour Diary Day)
|
|||
|
Hours of Awake Time “Off”
|
|
|
|
| Placebo |
6.6 |
- 0.3 |
– |
| Entacapone |
6.8 |
- 1.2 |
<0.01 |
|
Hours of Awake Time “On”
|
|||
| Placebo |
10.3 |
+ 0.4 |
– |
| Entacapone |
10.2 |
+ 1.0 |
N.S. |
|
Levodopa Total Daily Dose (mg)
|
|||
| Placebo |
758 |
+ 19 |
– |
| Entacapone |
804 |
- 93 |
<0.001 |
|
Frequency of Levodopa Daily Intakes
|
|||
| Placebo |
6.0 |
+ 0.2 |
– |
| Entacapone |
6.2 |
0.0 |
N.S. |
|
Other Secondary Measures
|
|||
| Baseline |
Change from Baseline at Month 6
|
p-value vs. placebo |
|
Investigator’s Global (overall) % Improved |
|||
| Placebo |
– |
21 |
– |
| Entacapone |
– |
34 |
<0.05 |
Patient’s Global (overall) % Improved |
|||
| Placebo |
– |
20 |
– |
| Entacapone |
– |
31 |
<0.05 |
UPDRS Total |
|||
| Placebo |
35.6 |
+2.8 |
– |
| Entacapone |
35.1 |
-0.6 |
<0.05 |
UPDRS Motor |
|||
| Placebo |
22.6 |
+1.2 |
– |
| Entacapone |
22.0 |
-0.9 |
<0.05 |
UPDRS ADL |
|||
| Placebo |
11.7 |
+1.1 |
– |
| Entacapone |
11.9 |
0.0 |
<0.05 |
Effects on “On” time did not differ by age, sex, weight, disease severity at baseline, levodopa dose and concurrent treatment with dopamine agonists or selegiline.
Withdrawal of entacapone: In the North American study, abrupt withdrawal of entacapone, without alteration of the dose of carbidopa-levodopa, resulted in a significant worsening of fluctuations, compared to placebo. In some cases, symptoms were slightly worse than at baseline, but returned to approximately baseline severity within two weeks following levodopa dose increase on average by 80 mg. In the Nordic study, similarly, a significant worsening of parkinsonian symptoms was observed after entacapone withdrawal, as assessed two weeks after drug withdrawal. At this phase, the symptoms were approximately at baseline severity following levodopa dose increase by about 50 mg.
|
Primary Measures
|
|||
| Baseline |
Change from Baseline at Month 6 |
p-value vs. placebo (LOCF) |
|
UPDRS ADL |
|
|
|
| Placebo |
12.0 |
+0.5 |
– |
| Entacapone |
12.4 |
-0.4 |
<0.05 |
UPDRS Motor |
|||
| Placebo |
24.1 |
+0.1 |
– |
| Entacapone |
24.9 |
-2.5 |
<0.05 |
Hours of Awake Time “On” (Home Diary) |
|||
| Placebo |
10.1 |
+0.5 |
– |
| Entacapone |
10.2 |
+1.1 |
N.S. |
|
Secondary Measures
|
|||
| |
Baseline |
Change from Baseline at Month 6 |
p-value vs. placebo |
UPDRS Total |
|||
| Placebo |
37.7 |
+0.6 |
– |
| Entacapone |
39.0 |
-3.4 |
<0.05 |
Percent of Awake Time “On” (Home Diary) |
|||
| Placebo |
59.8 |
+3.5 |
– |
| Entacapone |
62.0 |
+6.5 |
N.S. |
Hours of Awake Time “Off” (Home Diary) |
|||
| Placebo |
6.8 |
-0.6 |
– |
| Entacapone |
6.3 |
-1.2 |
0.07 |
Levodopa Total Daily Dose (mg) |
|||
| Placebo |
572 |
+4 |
– |
| Entacapone |
566 |
-35 |
N.S. |
Frequency of Levodopa Daily Intake |
|||
| Placebo |
5.6 |
+0.2 |
– |
| Entacapone |
5.4 |
0.0 |
<0.01 |
Global (overall) % Improved |
|||
| Placebo |
– |
34 |
– |
| Entacapone |
– |
38 |
N.S. |
INDICATIONS
Carbidopa, levodopa and entacapone tablets are indicated to treat patients with idiopathic Parkinson’s disease:
- To substitute (with equivalent strength of each of the three components) for immediate-release carbidopa/levodopa and entacapone previously administered as individual products.
- To replace immediate-release carbidopa/levodopa therapy (without entacapone) when patients experience the signs and symptoms of end-of-dose “wearing-off” (only for patients taking a total daily dose of levodopa of 600 mg or less and not experiencing dyskinesias, see DOSAGE AND ADMINISTRATION).
CARBIDOPA, LEVODOPA AND ENTACAPONE CONTRAINDICATIONS
Carbidopa, levodopa and entacapone tablets are contraindicated in patients who have demonstrated hypersensitivity to any component (carbidopa, levodopa, or entacapone) of the drug or its excipients.
Monoamine oxidase (MAO) and COMT are the two major enzyme systems involved in the metabolism of catecholamines. It is theoretically possible, therefore, that the combination of entacapone and a non-selective MAO inhibitor (e.g., phenelzine and tranylcypromine) would result in inhibition of the majority of the pathways responsible for normal catecholamine metabolism. As with carbidopa-levodopa, nonselective monoamine oxidase (MAO) inhibitors are contraindicated for use with carbidopa, levodopa and entacapone tablets. These inhibitors must be discontinued at least two weeks prior to initiating therapy with carbidopa, levodopa and entacapone tablets. Carbidopa, levodopa and entacapone tablets may be administered concomitantly with the manufacturer's recommended dose of MAO inhibitors with selectivity for MAO type B (e.g., selegiline HCl) (See PRECAUTIONS, Drug Interactions).
Carbidopa, levodopa and entacapone tablets are contraindicated in patients with narrow-angle glaucoma.
WARNINGS
The addition of carbidopa to levodopa reduces the peripheral effects (nausea, vomiting) due to decarboxylation of levodopa; however, carbidopa does not decrease the adverse reactions due to the central effects of levodopa. Because carbidopa as well as entacapone permits more levodopa to reach the brain and more dopamine to be formed, certain adverse CNS effects, e.g., dyskinesia (involuntary movements) may occur at lower dosages and sooner with levodopa preparations containing carbidopa and entacapone than with levodopa alone.
The occurrence of dyskinesias may require dosage reduction (see PRECAUTIONS, Dyskinesia).
Carbidopa, levodopa and entacapone tablets may cause mental disturbances. These reactions are thought to be due to increased brain dopamine following administration of levodopa. All patients should be observed carefully for the development of depression with concomitant suicidal tendencies. Patients with past or current psychoses should be treated with caution.
Carbidopa, levodopa and entacapone tablets should be administered cautiously to patients with severe cardiovascular or pulmonary disease, bronchial asthma, renal, hepatic or endocrine disease.
As with levodopa, care should be exercised in administering carbidopa, levodopa and entacapone tablets to patients with a history of myocardial infarction who have residual atrial, nodal, or ventricular arrhythmias. In such patients, cardiac function should be monitored carefully during the period of initial dosage adjustment, in a facility with provisions for intensive cardiac care.
Neuroleptic Malignant Syndrome (NMS)
Sporadic cases of a symptom complex resembling NMS have been reported in association with dose reductions or withdrawal of therapy with carbidopa-levodopa. Therefore, patients should be observed carefully when the dosage of carbidopa, levodopa and entacapone tablets is reduced abruptly or discontinued, especially if the patient is receiving neuroleptics. NMS is an uncommon but life-threatening syndrome characterized by fever or hyperthermia. Neurological findings, including muscle rigidity, involuntary movements, altered consciousness, mental status changes; other disturbances, such as autonomic dysfunction, tachycardia, tachypnea, sweating, hyper- or hypotension; laboratory findings, such as creatine phosphokinase elevation, leukocytosis, myoglobinuria, and increased serum myoglobin have been reported.
The early diagnosis of this condition is important for the appropriate management of these patients. Considering NMS as a possible diagnosis and ruling out other acute illnesses (e.g., pneumonia, systemic infection, etc.) is essential. This may be especially complex if the clinical presentation includes both serious medical illness and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system (CNS) pathology.
The management of NMS should include: 1) intensive symptomatic treatment and medical monitoring and 2) treatment of any concomitant serious medical problems for which specific treatments are available. Dopamine agonists, such as bromocriptine, and muscle relaxants, such as dantrolene, are often used in the treatment of NMS, however, their effectiveness has not been demonstrated in controlled studies.
Drugs Metabolized By Catechol-O-Methyltransferase (COMT)
When a single 400 mg dose of entacapone was given together with intravenous isoprenaline (isoproterenol) and epinephrine without coadministered levodopa/dopa decarboxylase inhibitor, the overall mean maximal changes in heart rate during infusion were about 50% and 80% higher than with placebo, for isoprenaline and epinephrine, respectively.
Therefore, drugs known to be metabolized by COMT, such as isoproterenol, epinephrine, norepinephrine, dopamine, dobutamine, alpha-methyldopa, apomorphine, isoetherine, and bitolterol should be administered with caution in patients receiving entacapone regardless of the route of administration (including inhalation), as their interaction may result in increased heart rates, possibly arrhythmias, and excessive changes in blood pressure.
Ventricular tachycardia was noted in one 32-year-old healthy male volunteer in an interaction study after epinephrine infusion and oral entacapone administration. Treatment with propranolol was required. A causal relationship to entacapone administration appears probable but cannot be attributed with certainty.
PRECAUTIONS
General
Hypotension/Syncope
In the large controlled trials of entacapone, approximately 1.2% and 0.8% of 200 mg entacapone and placebo patients treated also with levodopa/dopa decarboxylase inhibitor, respectively, reported at least one episode of syncope. Reports of syncope were generally more frequent in patients in both treatment groups who had an episode of documented hypotension (although the episodes of syncope, obtained by history, were themselves not documented with vital sign measurement).
Diarrhea and Colitis
In clinical trials of entacapone, diarrhea developed in 60 of 603 (10.0%) and 16 of 400 (4.0%) of patients treated with 200 mg of entacapone or placebo in combination with levodopa/dopa decarboxylase inhibitor, respectively. In patients treated with entacapone, diarrhea was generally mild to moderate in severity (8.6%) but was regarded as severe in 1.3%. Diarrhea resulted in withdrawal in 10 of 603 (1.7%) patients, 7 (1.2%) with mild and moderate diarrhea and 3 (0.5%) with severe diarrhea. Diarrhea generally resolved after discontinuation of entacapone. Two patients with diarrhea were hospitalized. Typically, diarrhea presents within 4 weeks to 12 weeks after entacapone is started, but it may appear as early as the first week and as late as many months after the initiation of treatment. Diarrhea may be associated with weight loss, dehydration, and hypokalemia.
Post-marketing experience has shown that diarrhea may be a sign of drug-induced microscopic colitis, primarily lymphocytic colitis. In these cases diarrhea has usually been moderate to severe, watery and non-bloody, at times associated with dehydration, abdominal pain, weight loss, and hypokalemia. In the majority of cases, diarrhea and other colitis-related symptoms resolved or significantly improved when entacapone treatment was stopped. In some patients with biopsy confirmed colitis, diarrhea had resolved or significantly improved after discontinuation of entacapone but recurred after retreatment with entacapone.
If prolonged diarrhea is suspected to be related to carbidopa, levodopa and entacapone tablets, the drug should be discontinued and appropriate medical therapy considered. If the cause of prolonged diarrhea remains unclear or continues after stopping entacapone, then further diagnostic investigations including colonoscopy and biopsies should be considered.
Hallucinations
Dyskinesia
Other Events Reported With Dopaminergic Therapy
The events listed below are rare events known to be associated with the use of drugs that increase dopaminergic activity, although they are most often associated with the use of direct dopamine agonists.
Rhabdomyolysis: Cases of severe rhabdomyolysis have been reported with entacapone when used in combination with levodopa. The complicated nature of these cases makes it impossible to determine what role, if any, entacapone played in their pathogenesis. Severe prolonged motor activity including dyskinesia may account for rhabdomyolysis. One case, however, included fever and alteration of consciousness. It is therefore possible that the rhabdomyolysis may be a result of the syndrome described in Hyperpyrexia and Confusion (see PRECAUTIONS, Other Events Reported With Dopaminergic Therapy).
Hyperpyrexia and Confusion: Cases of a symptom complex resembling the neuroleptic malignant syndrome characterized by elevated temperature, muscular rigidity, altered consciousness, and elevated CPK have been reported in association with the rapid dose reduction or withdrawal of other dopaminergic drugs. No cases have been reported following the abrupt withdrawal or dose reduction of entacapone treatment during clinical studies.
Prescribers should exercise caution when discontinuing carbidopa, levodopa and entacapone combination treatment. When considered necessary, withdrawal should proceed slowly. If a decision is made to discontinue treatment with carbidopa, levodopa and entacapone tablets, recommendations include monitoring the patient closely and adjusting other dopaminergic treatments as needed. This syndrome should be considered in the differential diagnosis for any patient who develops a high fever or severe rigidity. Tapering entacapone has not been systematically evaluated.
Fibrotic Complications: Cases of retroperitoneal fibrosis, pulmonary infiltrates, pleural effusion, and pleural thickening have been reported in some patients treated with ergot derived dopaminergic agents. These complications may resolve when the drug is discontinued, but complete resolution does not always occur. Although these adverse events are believed to be related to the ergoline structure of these compounds, whether other, nonergot derived drugs (e.g., entacapone, levodopa) that increase dopaminergic activity can cause them is unknown. It should be noted that the expected incidence of fibrotic complications is so low that even if entacapone caused these complications at rates similar to those attributable to other dopaminergic therapies, it is unlikely that it would have been detected in a cohort of the size exposed to entacapone. Four cases of pulmonary fibrosis were reported during clinical development of entacapone; three of these patients were also treated with pergolide and one with bromocriptine. The duration of treatment with entacapone ranged from 7 months to 17 months.
Melanoma: Epidemiological studies have shown that patients with Parkinson’s disease have a higher risk (2- to approximately 6-fold higher) of developing melanoma than the general population. Whether the increased risk observed was due to Parkinson’s disease or other factors, such as drugs used to treat Parkinson’s disease, is unclear.
For the reasons stated above, patients and providers are advised to monitor for melanomas frequently and on a regular basis when using carbidopa, levodopa and entacapone tablets for any indication. Ideally, periodic skin examination should be performed by appropriately qualified individuals (e.g., dermatologists).
Renal Toxicity
Hepatic Impairment
Patients with hepatic impairment should be treated with caution. The AUC and Cmax of entacapone approximately doubled in patients with documented liver disease compared to controls. (See CLINICAL PHARMACOLOGY, Pharmacokinetics, and DOSAGE AND ADMINISTRATION).
Biliary Obstruction
Information for Patients
The patient should be instructed to take carbidopa, levodopa and entacapone tablets only as prescribed. The patient should be informed that carbidopa, levodopa and entacapone tablets are a standard-release formulation of carbidopa-levodopa combined with entacapone that is designed to begin release of ingredients within 30 minutes after ingestion. It is important that carbidopa, levodopa and entacapone tablets be taken at regular intervals according to the schedule outlined by the physician. The patient should be cautioned not to change the prescribed dosage regimen and not to add any additional antiparkinsonian medications, including other carbidopa-levodopa preparations, without first consulting the physician.
Patients should be advised that sometimes a “wearing-off” effect may occur at the end of the dosing interval. The physician should be notified for possible treatment adjustments if such response poses a problem to patient's everyday life.
Patients should be advised that occasionally, dark color (red, brown, or black) may appear in saliva, urine, or sweat after ingestion of carbidopa, levodopa and entacapone tablets. Although the color appears to be clinically insignificant, garments may become discolored.
The patient should be advised that a change in diet to foods that are high in protein may delay the absorption of levodopa and may reduce the amount taken up in the circulation. Excessive acidity also delays stomach emptying, thus delaying the absorption of levodopa. Iron salts (such as in multi-vitamin tablets) may also reduce the amount of levodopa available to the body. The above factors may reduce the clinical effectiveness of the levodopa, carbidopa-levodopa and carbidopa, levodopa and entacapone tablets therapy.
NOTE: The suggested advice to patients being treated with carbidopa, levodopa and entacapone tablets is intended to aid in the safe and effective use of this medication. It is not a disclosure of all possible adverse or intended effects.
Patients should be informed that hallucinations can occur.
Patients should be advised that they may develop postural (orthostatic) hypotension with or without symptoms such as dizziness, nausea, syncope, and sweating. Hypotension may occur more frequently during initial therapy or when total daily levodopa dosage is increased. Accordingly, patients should be cautioned against rising rapidly after sitting or lying down, especially if they have been doing so for prolonged periods, and especially at the initiation of treatment with carbidopa, levodopa and entacapone tablets.
Patients should be advised that they should neither drive a car nor operate other complex machinery until they have gained sufficient experience on carbidopa, levodopa and entacapone tablets to gauge whether or not it affects their mental and/or motor performance adversely. Because of the possible additive sedative effects, caution should be used when patients are taking other CNS depressants in combination with carbidopa, levodopa and entacapone tablets.
Patients should be informed that nausea may occur, especially at the initiation of treatment with carbidopa, levodopa and entacapone tablets.
Patients should be informed that diarrhea may occur with carbidopa, levodopa and entacapone tablets and it may have a delayed onset. Sometimes prolonged diarrhea may be caused by colitis (inflammation of the large intestine). Patients with diarrhea should drink fluids to maintain adequate hydration and monitor for weight loss. If diarrhea associated with carbidopa, levodopa and entacapone tablets is prolonged, discontinuing the drug is expected to lead to resolution, if diarrhea continues after stopping carbidopa, levodopa and entacapone tablets, further diagnostic investigations may be needed.
Patients should be advised of the possibility of an increase in dyskinesia.
Carbidopa-levodopa combination and entacapone are known to affect embryo-fetal development in the rabbit and in the rat, respectively. Accordingly, patients should be advised to notify their physicians if they become pregnant or intend to become pregnant during therapy (see PRECAUTIONS, Pregnancy).
Carbidopa and entacapone are known to be excreted into maternal milk in rats. Because of the possibility that carbidopa, levodopa and entacapone may be excreted into human maternal milk, patients should be advised to notify their physicians if they intend to breast-feed or are breast-feeding an infant.
Laboratory Tests
Abnormalities in laboratory tests may include elevations of liver function tests such as alkaline phosphatase, SGOT (AST), SGPT (ALT), lactic dehydrogenase, and bilirubin. Abnormalities in blood urea nitrogen and positive Coombs’ test have also been reported. Commonly, levels of blood urea nitrogen, creatinine, and uric acid are lower during administration of carbidopa, levodopa and entacapone tablets than with levodopa.
Carbidopa, levodopa and entacapone tablets may cause a false-positive reaction for urinary ketone bodies when a test tape is used for determination of ketonuria. This reaction will not be altered by boiling the urine specimen. False-negative tests may result with the use of glucose-oxidase methods of testing for glucosuria.
Cases of falsely diagnosed pheochromocytoma in patients on carbidopa-levodopa therapy have been reported very rarely. Caution should be exercised when interpreting the plasma and urine levels of catecholamines and their metabolites in patients on carbidopa-levodopa therapy.
Entacapone is a chelator of iron. The impact of entacapone on the body’s iron stores is unknown; however, a tendency towards decreasing serum iron concentrations was noted in clinical trials. In a controlled clinical study serum ferritin levels (as marker of iron deficiency and subclinical anemia) were not changed with entacapone compared to placebo after one year of treatment and there was no difference in rates of anemia or decreased hemoglobin levels.
Drug Interactions
Caution should be exercised when the following drugs are administered concomitantly with carbidopa, levodopa and entacapone tablets.
Anti-hypertensive agents: Symptomatic postural hypotension has occurred when carbidopa-levodopa was added to the treatment of patients receiving antihypertensive drugs. Therefore, when therapy with carbidopa, levodopa and entacapone tablets is started, dosage adjustment of the antihypertensive drug may be required.
MAO inhibitors: For patients receiving nonselective MAO inhibitors, see CONTRAINDICATIONS. Concomitant therapy with selegiline and carbidopa-levodopa may be associated with severe orthostatic hypotension not attributable to carbidopa-levodopa alone.
Tricyclic antidepressants: There have been rare reports of adverse reactions, including hypertension and dyskinesia, resulting from the concomitant use of tricyclic antidepressants and carbidopa-levodopa.
Dopamine D2 receptor antagonists (e.g., phenothiazines, butyrophenones, risperidone) and isoniazid: Dopamine D2 receptor antagonists (e.g., phenothiazines, butyrophenones, risperidone) and isoniazid may reduce the therapeutic effects of levodopa.
Phenytoin and papaverine: The beneficial effects of levodopa in Parkinson's disease have been reported to be reversed by phenytoin and papaverine. Patients taking these drugs with carbidopa-levodopa should be carefully observed for loss of therapeutic response.
Iron salts: Iron salts may reduce the bioavailability of levodopa, carbidopa and entacapone. The clinical relevance is unclear.
Metoclopramide: Although metoclopramide may increase the bioavailability of levodopa by increasing gastric emptying, metoclopramide may also adversely affect disease control by its dopamine receptor antagonistic properties.
Drugs known to interfere with biliary excretion, glucuronidation, and intestinal beta-glucuronidase (probenecid, cholestyramine, erythromycin, rifampicin, ampicillin and chloramphenicol): As most entacapone excretion is via the bile, caution should be exercised when drugs known to interfere with biliary excretion, glucuronidation, and intestinal beta-glucuronidase are given concurrently with entacapone. These include probenecid, cholestyramine, and some antibiotics (e.g., erythromycin, rifampicin, ampicillin and chloramphenicol).
Pyridoxine: Carbidopa, levodopa and entacapone tablets can be given to patients receiving supplemental pyridoxine. Oral coadministration of 10 mg to 25 mg of pyridoxine hydrochloride (vitamin B6) with levodopa may reverse the effects of levodopa by increasing the rate of aromatic amino acid decarboxylation. Carbidopa inhibits this action of pyridoxine; therefore, carbidopa, levodopa and entacapone tablets can be given to patients receiving supplemental pyridoxine.
Effect of levodopa and carbidopa in carbidopa, levodopa and entacapone tablets on the metabolism of other drugs: Inhibition or induction effect of levodopa and carbidopa has not been investigated.
Effect of entacapone in carbidopa, levodopa and entacapone tablets on the metabolism of other drugs: Entacapone is unlikely to inhibit the metabolism of other drugs that are metabolized by major P450s including CYP1A2, CYP2A6, CYP2C9, CYP2C19, CYP2D6, CYP2E1 and CYP3A. In vitro studies of human CYP enzymes showed that entacapone inhibited the CYP enzymes 1A2, 2A6, 2C9, 2C19, 2D6, 2E1 and 3A only at very high concentrations (IC50 from 200 microM to over 1,000 microM; an oral 200 mg dose achieves a highest level of approximately 5 microM in people); these enzymes would therefore not be expected to be inhibited in clinical use. However, no information is available regarding the induction effect from entacapone.
Drugs that are highly protein bound (such as warfarin, salicylic acid, phenylbutazone, and diazepam):
Levodopa
Levodopa is bound to plasma protein only to a minor extent (about 10% to 30%).
Carbidopa
Carbidopa is approximately 36% bound to plasma protein.
Entacapone
Entacapone is highly protein bound (98%). In vitro studies have shown no binding displacement between entacapone and other highly bound drugs, such as warfarin, salicylic acid, phenylbutazone, and diazepam.
Hormone Levels
Carcinogenesis
In a two-year bioassay of carbidopa-levodopa, no evidence of carcinogenicity was found in rats receiving doses of approximately two times the maximum daily human dose of carbidopa and four times the maximum daily human dose of levodopa.
2Mutagenesis
Carbidopa was positive in the Ames test in the presence and absence of metabolic activation, was mutagenic in the in vitro mouse lymphoma/thymidine kinase assay in the absence of metabolic activation, and was negative in the in vivo mouse micronucleus test.
in vitroin vivoImpairment of Fertility
In reproduction studies with carbidopa-levodopa, no effects on fertility were found in rats receiving doses of approximately two times the maximum daily human dose of carbidopa and four times the maximum daily human dose of levodopa.
Pregnancy
Pregnancy Category C
Carbidopa-levodopa caused both visceral and skeletal malformations in rabbits at all doses and ratios of carbidopa-levodopa tested, which ranged from 10 times/5 times the maximum recommended human dose of carbidopa-levodopa to 20 times/10 times the maximum recommended human dose of carbidopa-levodopa. There was a decrease in the number of live pups delivered by rats receiving approximately two times the maximum recommended human dose of carbidopa and approximately five times the maximum recommended human dose of levodopa during organogenesis. No teratogenic effects were observed in mice receiving up to 20 times the maximum recommended human dose of carbidopa-levodopa.
It has been reported from individual cases that levodopa crosses the human placental barrier, enters the fetus, and is metabolized. Carbidopa concentrations in fetal tissue appeared to be minimal.
In embryo-fetal development studies, entacapone was administered to pregnant animals throughout organogenesis at doses of up to 1,000 mg/kg/day in rats and 300 mg/kg/day in rabbits. Increased incidences of fetal variations were evident in litters from rats treated with the highest dose, in the absence of overt signs of maternal toxicity. The maternal plasma drug exposure (AUC) associated with this dose was approximately 34 times the estimated plasma exposure in humans receiving the maximum recommended daily dose (MRDD) of 1,600 mg. Increased frequencies of abortions and late/total resorptions and decreased fetal weights were observed in the litters of rabbits treated with maternotoxic doses of 100 mg/kg/day (plasma AUCs 0.4 times those in humans receiving the MRDD) or greater. There was no evidence of teratogenicity in these studies.
However, when entacapone was administered to female rats prior to mating and during early gestation, an increased incidence of fetal eye anomalies (macrophthalmia, microphthalmia, anophthalmia) was observed in the litters of dams treated with doses of 160 mg/kg/day (plasma AUCs seven times those in humans receiving the MRDD) or greater, in the absence of maternotoxicity. Administration of up to 700 mg/kg/day (plasma AUCs 28 times those in humans receiving the MRDD) to female rats during the latter part of gestation and throughout lactation, produced no evidence of developmental impairment in the offspring.
There is no experience from clinical studies regarding the use of carbidopa, levodopa and entacapone tablets in pregnant women. Therefore, carbidopa, levodopa and entacapone tablets should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Women
Pediatric Use
CARBIDOPA, LEVODOPA AND ENTACAPONE ADVERSE REACTIONS
Carbidopa-levodopa
Body as a Whole
Cardiovascular
Gastrointestinal
Hematologic
Hypersensitivity
Musculoskeletal
Nervous System/PsychiatricWARNINGS
Respiratory
Skin:
Urogenital
Laboratory Tests:
Other adverse reactions that have been reported with levodopa alone and with various carbidopa-levodopa formulations, and may occur with carbidopa, levodopa and entacapone tablets are:
Body as a Whole:
Cardiovascular
Gastrointestinal:
Metabolic:
Musculoskeletal:
Nervous System/Psychiatric:
Respiratory
Skin: CONTRAINDICATIONS.
Special Senses:
Urogenital:
Miscellaneous
Laboratory Tests:
Entacapone
The most commonly observed adverse events (greater than 5%) in the double-blind, placebo-controlled trials of entacapone (N=1,003) associated with the use of entacapone alone and not seen at an equivalent frequency among the placebo-treated patients were: dyskinesia/hyperkinesia, nausea, urine discoloration, diarrhea, and abdominal pain.
Approximately 14% of the 603 patients given entacapone in the double-blind, placebo-controlled trials discontinued treatment due to adverse events compared to 9% of the 400 patients who received placebo. The most frequent causes of discontinuation in decreasing order are: psychiatric reasons (2% vs. 1%), diarrhea (2% vs. 0%), dyskinesia/hyperkinesia (2% vs. 1%), nausea (2% vs. 1%), abdominal pain (1% vs. 0%), and aggravation of Parkinson’s disease symptoms (1% vs. 1%).
Adverse Event Incidence in Controlled Clinical Studies of Entacapone
|
SYSTEM ORGAN CLASS
Preferred Term |
Entacapone (n = 603) % of patients |
Placebo (n = 400) % of patients |
|
SKIN AND APPENDAGES DISORDERS
|
||
| Sweating Increased |
2 |
1 |
|
MUSCULOSKELETAL SYSTEM DISORDERS
|
||
| Back Pain |
2 |
1 |
|
CENTRAL AND PERIPHERAL NERVOUS SYSTEM DISORDERS
|
||
| Dyskinesia |
25 |
15 |
| Hyperkinesia |
10 |
5 |
| Hypokinesia |
9 |
8 |
| Dizziness |
8 |
6 |
|
SPECIAL SENSES, OTHER DISORDERS
|
||
| Taste Perversion |
1 |
0 |
|
PSYCHIATRIC DISORDERS
|
||
| Anxiety |
2 |
1 |
| Somnolence |
2 |
0 |
| Agitation |
1 |
0 |
|
GASTROINTESTINAL SYSTEM DISORDERS
|
||
| Nausea |
14 |
8 |
| Diarrhea |
10 |
4 |
| Abdominal Pain |
8 |
4 |
| Constipation |
6 |
4 |
| Vomiting |
4 |
1 |
| Mouth Dry |
3 |
0 |
| Dyspepsia |
2 |
1 |
| Flatulence |
2 |
0 |
| Gastritis |
1 |
0 |
| Gastrointestinal Disorders NOS |
1 |
0 |
|
RESPIRATORY SYSTEM DISORDERS
|
||
| Dyspnea |
3 |
1 |
|
PLATELET, BLEEDING AND CLOTTING DISORDERS
|
||
| Purpura |
2 |
1 |
|
URINARY SYSTEM DISORDERS
|
||
| Urine Discoloration |
10 |
0 |
|
BODY AS A WHOLE - GENERAL DISORDERS
|
||
| Back Pain |
4 |
2 |
| Fatigue |
6 |
4 |
| Asthenia |
2 |
1 |
|
RESISTANCE MECHANISM DISORDERS
|
||
| Infection Bacterial |
1 |
0 |
The prescriber should be aware that these figures cannot be used to predict the incidence of adverse events in the course of usual medical practice where patient characteristics and other factors differ from those that prevailed in the clinical studies. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses, and investigators. The cited figures do, however, provide the prescriber with some basis for estimating the relative contribution of drug and nondrug factors to the adverse events observed in the population studied.
Effects of Gender and Age on Adverse Reactions
DRUG ABUSE AND DEPENDENCE
Controlled substance class- Carbidopa, levodopa and entacapone is not a controlled substance.
OVERDOSAGE
Management of acute overdosage with carbidopa, levodopa and entacapone is the same as management of acute overdosage with levodopa and entacapone. Pyridoxine is not effective in reversing the actions of carbidopa, levodopa and entacapone tablets.
Hospitalization is advised, and general supportive measures should be employed, along with immediate gastric lavage and repeated doses of charcoal over time. This may hasten the elimination of entacapone in particular, by decreasing its absorption/reabsorption from the GI tract. Intravenous fluids should be administered judiciously and an adequate airway maintained.
The adequacy of the respiratory, circulatory and renal systems should be carefully monitored and appropriate supportive measures employed. Electrocardiographic monitoring should be instituted and the patient carefully observed for the development of arrhythmias; if required, appropriate antiarrhythmic therapy should be given. The possibility that the patient may have taken other drugs, increasing the risk of drug interactions (especially catechol-structured drugs) should be taken into consideration. To date, no experience has been reported with dialysis; hence, its value in overdosage is not known. Hemodialysis or hemoperfusion is unlikely to reduce entacapone levels due to its high binding to plasma proteins.
There are very few cases of overdosage with levodopa reported in the published literature. Based on the limited available information, the acute symptoms of levodopa/dopa decarboxylase inhibitor overdosage can be expected to arise from dopaminergic overstimulation. Doses of a few grams may result in CNS disturbances, with an increasing likelihood of cardiovascular disturbance (e.g., hypotension, tachycardia) and more severe psychiatric problems at higher doses. An isolated report of rhabdomyolysis and another of transient renal insufficiency suggest that levodopa overdosage may give rise to systemic complications, secondary to dopaminergic overstimulation.
There have been no reported cases of either accidental or intentional overdose with entacapone tablets. However, COMT inhibition by entacapone treatment is dose-dependent. A massive overdose of entacapone may theoretically produce a 100% inhibition of the COMT enzyme in people, thereby preventing the O-methylation of endogenous and exogenous catechols.
The highest single dose of entacapone administered to humans was 800 mg, resulting in a plasma concentration of 14.1 microgram/mL. The highest daily dose given to humans was 2,400 mg, administered in one study as 400 mg six times daily with carbidopa-levodopa for 14 days in 15 Parkinson’s disease patients, and in another study as 800 mg t.i.d. for 7 days in 8 healthy volunteers. At this daily dose, the peak plasma concentrations of entacapone averaged 2.0 microgram/mL (at 45 min., compared to 1.0 microgram/mL and 1.2 microgram/mL with 200 mg entacapone at 45 min.). Abdominal pain and loose stools were the most commonly observed adverse events during this study. Daily doses as high as 2,000 mg entacapone have been administered as 200 mg 10 times daily with carbidopa-levodopa or benserazide-levodopa for at least 1 year in 10 patients, for at least 2 years in 8 patients and for at least 3 years in 7 patients. Overall, however, clinical experience with daily doses above 1,600 mg is limited.
CARBIDOPA, LEVODOPA AND ENTACAPONE DOSAGE AND ADMINISTRATION
Individual tablets should not be fractionated and only one tablet should be administered at each dosing interval.
Generally speaking, carbidopa, levodopa and entacapone tablets should be used as a substitute for patients already stabilized on equivalent doses of carbidopa-levodopa and entacapone. However, some patients who have been stabilized on a given dose of carbidopa-levodopa may be treated with carbidopa, levodopa and entacapone tablets if a decision has been made to add entacapone (see below).
The optimum daily dosage of carbidopa, levodopa and entacapone tablets must be determined by careful titration in each patient. Carbidopa, levodopa and entacapone tablets are available in six strengths, each in a 1:4 ratio of carbidopa to levodopa and combined with 200 mg of entacapone in a standard release formulation (carbidopa, levodopa and entacapone tablets 12.5 mg/50 mg/200 mg containing 12.5 mg of carbidopa, 50 mg of levodopa and 200 mg of entacapone; carbidopa, levodopa and entacapone tablets 18.75 mg/75 mg/200 mg, containing 18.75 mg of carbidopa, 75 mg of levodopa and 200 mg of entacapone; carbidopa, levodopa and entacapone tablets 25 mg/100 mg/200 mg containing 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone; carbidopa, levodopa and entacapone tablets 31.25 mg/125 mg/200 mg, containing 31.25 mg of carbidopa, 125 mg of levodopa and 200 mg of entacapone; carbidopa, levodopa and entacapone tablets 37.5 mg/150 mg/200 mg containing 37.5 mg of carbidopa, 150 mg of levodopa and 200 mg of entacapone; and carbidopa, levodopa and entacapone tablets 50 mg/200 mg/200 mg containing 50 mg of carbidopa, 200 mg of levodopa and 200 mg of entacapone).
Therapy should be individualized and adjusted according to the desired therapeutic response.
Studies show that peripheral dopa decarboxylase is saturated by carbidopa at approximately 70 mg to 100 mg a day. Patients receiving less than this amount of carbidopa are more likely to experience nausea and vomiting.
Clinical experience with daily doses above 1,600 mg of entacapone is limited. It is recommended that no more than one carbidopa, levodopa and entacapone tablet be taken at each dosing administration.
Thus the maximum recommended daily dose of carbidopa, levodopa and entacapone tablet 12.5 mg/50 mg/200 mg, 18.75 mg/75 mg/200 mg, 25 mg/100 mg/200 mg, 31.25 mg/125 mg/200 mg, and 37.5 mg/150 mg/200 mg defined by the maximum daily dose of entacapone, is eight tablets per day. Because there is limited experience with total daily doses of carbidopa greater than 300 mg, the maximum recommended daily dose of carbidopa, levodopa and entacapone tablets 50 mg/200 mg/200 mg is six tablets per day.
How to transfer patients taking carbidopa-levodopa preparations and Comtan®* (entacapone) tablets to carbidopa, levodopa and entacapone tablets
There is no experience in transferring patients currently treated with formulations of carbidopa-levodopa other than immediate-release carbidopa-levodopa with a 1:4 ratio (controlled-release formulations, or standard-release presentations with a 1:10 ratio of carbidopa-levodopa) and entacapone to carbidopa, levodopa and entacapone tablets.
Patients who are currently treated with Comtan* 200 mg tablet with each dose of standard-release carbidopa-levodopa, can be directly switched to the corresponding strength of carbidopa, levodopa and entacapone tablet containing the same amounts of levodopa and carbidopa. For example, patients receiving one tablet of standard-release carbidopa-levodopa 25 mg/100 mg and one tablet of Comtan* 200 mg at each administration can be switched to a single carbidopa, levodopa and entacapone tablet 25 mg/100 mg/200 mg (containing 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone).
How to transfer patients not currently treated with Comtan®* (entacapone) tablets from carbidopa-levodopa to carbidopa, levodopa and entacapone tablets
In patients with Parkinson’s disease who experience the signs and symptoms of end-of-dose “wearing-off” on their current standard-release carbidopa-levodopa treatment, clinical experience shows that patients with a history of moderate or severe dyskinesias or taking more than 600 mg of levodopa per day are likely to require a reduction in daily levodopa dose when entacapone is added to their treatment. Since dose adjustment of the individual components is impossible with fixed-dose products, it is recommended that patients first be titrated individually with a carbidopa-levodopa product (ratio 1:4) and an entacapone product, and then transferred to a corresponding dose of carbidopa, levodopa and entacapone tablet once the patient’s status has stabilized.
In patients who take a total daily levodopa dose up to 600 mg, and who do not have dyskinesias, an attempt can be made to transfer to the corresponding daily dose of carbidopa, levodopa and entacapone tablet. Even in these patients, a reduction of carbidopa-levodopa or entacapone may be necessary however, the provider is reminded that this may not be possible with carbidopa, levodopa and entacapone tablet. Since entacapone prolongs and enhances the effects of levodopa, therapy should be individualized and adjusted if necessary according to the desired therapeutic response.
Maintenance of Carbidopa, Levodopa and Entacapone Tablets Treatment
Therapy should be individualized and adjusted for each patient according to the desired therapeutic response.
When less levodopa is required, the total daily dosage of carbidopa-levodopa should be reduced by either decreasing the strength of carbidopa, levodopa and entacapone tablets at each administration or by decreasing the frequency of administration by extending the time between doses.
When more levodopa is required, the next higher strength of carbidopa, levodopa and entacapone tablet should be taken and/or the frequency of doses should be increased, up to a maximum of 8 times daily of carbidopa, levodopa and entacapone tablets 12.5 mg/50 mg/200 mg, 18.75 mg/75 mg/200 mg, 25 mg/100 mg/200 mg, 31.25 mg/125 mg/200 mg, and 37.5 mg/150 mg/200 mg, and maximum of 6 times daily of carbidopa, levodopa and entacapone tablets 50 mg/200 mg/200 mg.
Addition of Other Antiparkinsonian Medications
Standard drugs for Parkinson's disease may be used concomitantly while carbidopa, levodopa and entacapone tablets are being administered, although dosage adjustments may be required.
Interruption of Therapy
Sporadic cases of a symptom complex resembling Neuroleptic Malignant Syndrome (NMS) have been associated with dose reductions and withdrawal of levodopa preparations. Patients should be observed carefully if abrupt reduction or discontinuation of carbidopa, levodopa and entacapone tablets is required, especially if the patient is receiving neuroleptics. (See WARNINGS.)
If general anesthesia is required, carbidopa, levodopa and entacapone tablet may be continued as long as the patient is permitted to take fluids and medication by mouth. If therapy is interrupted temporarily, the patient should be observed for symptoms resembling NMS, and the usual daily dosage may be administered as soon as the patient is able to take oral medication.
Special Populations
Patients With Impaired Hepatic Function:
maxCLINICAL PHARMACOLOGY, Pharmacokinetics of EntacaponeHOW SUPPLIED
Carbidopa, levodopa and entacapone tablets are supplied as film-coated tablets for oral administration in the following six strengths:
47335‑001‑88
Caraco Pharmaceutical Laboratories, Ltd.
PRINCIPAL DISPLAY PANEL-Label-12.5 mg/50 mg/200 mg
NDC 47335-001-88
Carbidopa, Levodopa and Entacapone Tablets
Health Care Provider: Do not combine lower strength tablets to
achieve a higher strength tablet due to risk of entacapone overdose.

PRINCIPAL DISPLAY PANEL-Label-18.75 mg/75 mg/200 mg
NDC 47335-002-88
Carbidopa, Levodopa and Entacapone Tablets
Health Care Provider: Do not combine lower strength tablets to
achieve a higher strength tablet due to risk of entacapone overdose.

PRINCIPAL DISPLAY PANEL-Label-25 mg/100 mg/200 mg
NDC 47335-003-88
Carbidopa, Levodopa and Entacapone Tablets
Health Care Provider: Do not combine lower strength tablets to
achieve a higher strength tablet due to risk of entacapone overdose.

PRINCIPAL DISPLAY PANEL-Label-31.25 mg/125 mg/200 mg
NDC 47335-004-88
Carbidopa, Levodopa and Entacapone Tablets
Health Care Provider: Do not combine lower strength tablets to
achieve a higher strength tablet due to risk of entacapone overdose.

PRINCIPAL DISPLAY PANEL-Label-37.5 mg/150 mg/200 mg
NDC 47335-005-88
Carbidopa, Levodopa and Entacapone Tablets
Health Care Provider: Do not combine lower strength tablets to
achieve a higher strength tablet due to risk of entacapone overdose.

PRINCIPAL DISPLAY PANEL-Label-50 mg/200 mg/200 mg
NDC 47335-006-88
Carbidopa, Levodopa and Entacapone Tablets
Health Care Provider: Do not combine lower strength tablets to
achieve a higher strength tablet due to risk of entacapone overdose.

Carbidopa, levodopa and entacaponeCarbidopa, levodopa and entacapone TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Carbidopa, levodopa and entacaponeCarbidopa, levodopa and entacapone TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Carbidopa, levodopa and entacaponeCarbidopa, levodopa and entacapone TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Carbidopa, levodopa and entacaponeCarbidopa, levodopa and entacapone TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Carbidopa, levodopa and entacaponeCarbidopa, levodopa and entacapone TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Carbidopa, levodopa and entacaponeCarbidopa, levodopa and entacapone TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||