Cetirizine Hydrochloride
Caraco Pharmaceutical Laboratories, Ltd.
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient (in each tablet)
- Purpose
- Cetirizine Hydrochloride Uses
- Warnings
- Directions
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 5 mg 30 Tablets
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 5 mg 30 Tablets
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 10 mg 30 Tablets
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 10 mg 30 Tablets
FULL PRESCRIBING INFORMATION
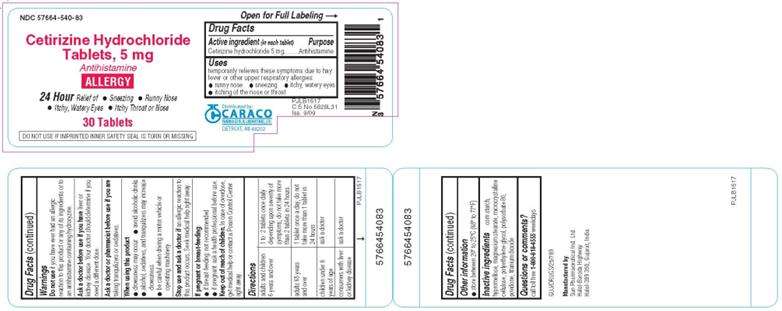
Drug Facts
Active ingredient (in each tablet)
For 5 mg:
Cetirizine hydrochloride 5 mg
For 10 mg:
Cetirizine hydrochloride 10 mg
Purpose
Antihistamine
Cetirizine Hydrochloride Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Warnings
Do not use if you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Ask a doctor or pharmacist before use if you are taking tranquilizers or sedatives.
When using this product
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery.
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
If pregnant or breast-feeding:
- If breast-feeding: not recommended
- if pregnant: ask a health professional before use.
Keep out of reach of children In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
For 5 mg:
| adults and children 6 years and over |
1 to 2 tablets once daily depending upon severity of symptoms; do not take more than 2 tablets in 24 hours. |
| adults 65 years and over |
1 tablet once a day; do not take more than 1 tablet in 24 hours |
| children under 6 years of age |
ask a doctor |
| consumers with liver or kidney disease |
ask a doctor |
For 10 mg:
| adults and children 6 years and over |
one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. |
| adults 65 years and over |
ask a doctor |
| children under 6 years of age |
ask a doctor |
| consumers with liver or kidney disease |
ask a doctor |
Other information
- store between 20° to 25°C (68° to 77°F)
Inactive ingredients corn starch, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, povidone, titanium dioxide
Questions or comments?
call toll free 1-800-818-4555 weekdays
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 5 mg 30 Tablets
NDC 57664-540-83
Cetirizine Hydrochloride Tablets, 5 mg
Antihistamine
ALLERGY
30 Tablets
Caraco Pharmaceutical Laboratories, Ltd.

PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 5 mg 30 Tablets
NDC 57664-540-83
Cetirizine Hydrochloride Tablets, 5 mg
Antihistamine
ALLERGY
30 Tablets
Caraco Pharmaceutical Laboratories, Ltd.
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 10 mg 30 Tablets
NDC 57664-541-83
Cetirizine Hydrochloride Tablets, 10 mg
Antihistamine
ALLERGY
30 Tablets
Caraco Pharmaceutical Laboratories, Ltd.

PACKAGE LABEL.PRINCIPAL DISPLAY PANEL - 10 mg 30 Tablets
NDC 57664-541-83
Cetirizine Hydrochloride Tablets, 10 mg
Antihistamine
ALLERGY
30 Tablets
Caraco Pharmaceutical Laboratories, Ltd.

Cetirizine HydrochlorideCetirizine Hydrochloride TABLET, FILM COATED
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Cetirizine HydrochlorideCetirizine Hydrochloride TABLET, FILM COATED
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||