Chloroquine Phosphate
Global Pharmaceuticals, Division of Impax Laboratories, Inc.
CHLOROQUINE PHOSPHATE TABLETS, USP250 mg
FULL PRESCRIBING INFORMATION: CONTENTS*
- WARNING
- CHLOROQUINE PHOSPHATE DESCRIPTION
- CLINICAL PHARMACOLOGY
- CHLOROQUINE PHOSPHATE INDICATIONS AND USAGE
- CHLOROQUINE PHOSPHATE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- CHLOROQUINE PHOSPHATE ADVERSE REACTIONS
- OVERDOSAGE
- CHLOROQUINE PHOSPHATE DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- PRINCIPAL DISPLAY PANEL - 250 mg Tablet Bottle Label
FULL PRESCRIBING INFORMATION
Rx Only
FOR USE IN MALARIA AND EXTRAINTESTINAL AMEBIASIS ONLY
WARNING
PHYSICIANS SHOULD FAMILIARIZE THEMSELVES WITH THE COMPLETE CONTENTS OF THIS OUTSERT BEFORE PRESCRIBING CHLOROQUINE PHOSPHATE
CHLOROQUINE PHOSPHATE DESCRIPTION
Chloroquine phosphate, USP, is a 4-aminoquinoline compound for oral administration. It is a white, odorless, bitter tasting, crystalline substance, freely soluble in water.
Chloroquine phosphate is an antimalarial and amebicidal drug.
Chemically, it is 7-Chloro-4[[4-(diethylamino)-1-methylbutyl]amino] quinoline phosphate (1:2). Its molecular formula is C18H32ClN3O8P2 and its structural formula is:
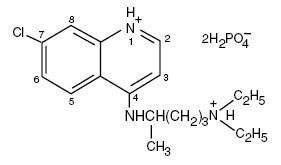
The molecular weight is 515.86.
Inactive Ingredients: Citric Acid, Anhydrous, Hydrogenated Vegetable Oil, Lactose Monohydrate, Magnesium Stearate, Corn Starch, Pregelatinized Starch, Starch.
CLINICAL PHARMACOLOGY
Chloroquine phosphate has been found to be highly active against the erythrocytic forms of Plasmodium vivax and Plasmodium malariae and most strains of Plasmodium falciparum (but not the gametocytes of P. falciparum).
The mechanism of plasmodicidal action of chloroquine is not completely certain. While the drug can inhibit certain enzymes, its effect is believed to result, at least in part, from its interaction with DNA.
Chloroquine is rapidly and almost completely absorbed from the gastrointestinal tract, and only a small proportion of the administered dose is found in the stools. Approximately 55% of the drug in the plasma is bound to nondiffusible plasma constituents. Excretion of chloroquine is quite slow, but is increased by acidification of the urine. Chloroquine is deposited in the tissues in considerable amounts. In animals, from 200 to 700 times the plasma concentration may be found in the liver, spleen, kidney, and lung; leukocytes also concentrate the drug. The brain and spinal cord, in contrast, contain only 10 to 30 times the amount present in plasma.
Chloroquine undergoes appreciable degradation in the body. The main metabolite is desethylchloroquine, which accounts for one fourth of the total material appearing in the the urine; bisdesethylchloroquine, a carboxylic acid derivative, and other metabolic products as yet uncharacterized are found in small amounts. Slightly more than half of the urinary drug products can be accounted for as unchanged chloroquine.
Microbiology
Chloroquine phosphate has been found to be highly active again the erythrocytic forms of Plasmodium vivax and malariae and most strains of Plasmodium falciparum (but not the gametocytes of P. falciparum). The precise mechanism of action of the drug is not known.
In vitro studies with trophozoites of Entamoeba histolytica have demonstrated that chloroquine phosphate also possesses amebicidal activity comparable to that of emetine.
CHLOROQUINE PHOSPHATE INDICATIONS AND USAGE
Chloroquine phosphate tablets are indicated for the suppressive treatment and for acute attacks of malaria due to P. vivax, P. malariae, P. ovale, and susceptible strains of P. falciparum. The drug is also indicated for the treatment of extraintestinal amebiasis.
Chloroquine phosphate does not prevent relapses in patients with vivax or malariae malaria because it is not effective against exo-erythrocytic forms of the parasite, nor will it prevent vivax or malariae infection when administered as a prophylactic. It is highly effective as a suppressive agent in patients with vivax or malariae malaria, in terminating acute attacks, and significantly lengthening the interval between treatment and relapse. In patients with falciparum malaria it abolishes the acute attack and effects complete cure of the infection, unless due to a resistant strain of P. falciparum.
CHLOROQUINE PHOSPHATE CONTRAINDICATIONS
Use of this drug is contraindicated in the presence of retinal or visual field changes either attributable to 4-aminoquinoline compounds or to any other etiology, and in patients with known hypersensitivity to 4-aminoquinoline compounds. However, in the treatment of acute attacks of malaria caused by susceptible strains of plasmodia, the physician may elect to use this drug after carefully weighing the possible benefits and risks to the patient.
WARNINGS
In recent years it has been found that certain strains of P. falciparum have become resistant to 4-aminoquinoline compounds (including chloroquine and hydroxychloroquine) as shown by the fact that normally adequate doses have failed to prevent or cure clinical malaria or parasitemia. Treatment with quinine or other specific forms of therapy is therefore advised for patients infected with a resistant strain of parasites.
Irreversible retinal damage has been observed in some patients who had received long-term or high-dosage 4-aminoquinoline therapy. Retinopathy has been reported to be dose related.
When prolonged therapy with any antimalarial compound is contemplated, initial (base line) and periodic ophthalmologic examinations (including visual acuity, expert slit-lamp, funduscopic, and visual field tests) should be performed.
If there is any indication (past or present) or abnormality in the visual acuity, visual field, or retinal macular areas (such as pigmentary changes, loss of foveal reflex), or any visual symptoms (such as light flashes and streaks) which are not fully explainable by difficulties of accommodation or corneal opacities, the drug should be discontinued immediately and the patient closely observed for possible progression. Retinal changes (and visual disturbances) may progress even after cessation of therapy.
All patients on long-term therapy with this preparation should be questioned and examined periodically, including testing knee and ankle reflexes, to detect any evidence of muscular weakness. If weakness occurs, discontinue the drug.
A number of fatalities have been reported following the accidental ingestion of chloroquine, sometimes in relatively small doses (0.75 g or 1 g chloroquine phosphate in one 3-year-old child). Patients should be strongly warned to keep this drug out of the reach of children because they are especially sensitive to the 4-aminoquinoline compounds.
Use of chloroquine phosphate in patients with psoriasis may precipitate a severe attack of psoriasis. When used in patients with porphyria the condition may be exacerbated. The drug should not be used in these conditions unless in the judgment of the physician the benefit to the patient outweighs the possible hazard.
PRECAUTIONS
General
If any severe blood disorder appears which is not attributable to the disease under treatment, discontinuance of the drug should be considered.
Since this drug is known to concentrate in the liver, it should be used with caution in patients with hepatic disease or alcoholism or in conjunction with known hepatotoxic drugs.
The drug should be administered with caution to patients having G-6-PD (glucose-6 phosphate dehydrogenase) deficiency.
Laboratory Tests
Complete blood cell counts should be made periodically if patients are given prolonged therapy.
Nursing Mothers
Because of the potential for serious adverse reactions in nursing infants from chloroquine, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
See WARNINGS and DOSAGE AND ADMINISTRATION.
CHLOROQUINE PHOSPHATE ADVERSE REACTIONS
Ocular reactions: Irreversible retinal damage in patients receiving long-term or high-dose 4-aminoquinoline therapy; visual disturbances (blurring of vision and difficulty of focusing or accommodation); nyctalopia; scotomatous vision with field defects of paracentral, pericentral ring types, and typically temporal scotomas, e.g., difficulty in reading with words tending to disappear, seeing half an object, misty vision, and fog before the eyes.
Neuromuscular reactions: Convulsive seizures.
Auditory reactions: Nerve type deafness; tinnitus, reduced hearing in patients with preexisting auditory damage.
Gastrointestinal reactions: Anorexia, nausea, vomiting, diarrhea, abdominal cramps.
Dermatologic reactions: Pleomorphic skin eruptions, skin and mucosal pigmentary changes; lichen planus-like eruptions, pruritus, and hair loss.
CNS reactions: Mild and transient headache; psychic stimulation.
Cardiovascular reactions: Rarely, hypotension, electrocardiographic change.
OVERDOSAGE
Symptoms
Chloroquine is very rapidly and completely absorbed after ingestion. Toxic doses of chloroquine can be fatal. As little as 1 g may be fatal in children. Toxic symptoms can occur within minutes. These consist of headache, drowsiness, visual disturbances, nausea and vomiting, cardiovascular collapse, and convulsions following by sudden and early respiratory and cardiac arrest. The electrocardiogram may reveal atrial standstill, nodal rhythm, prolonged intraventricular conduction time, and progressive bradycardia leading to ventricular fibrillation and/or arrest.
Treatment
Treatment is symptomatic and must be prompt with immediate evacuation of the stomach by emesis (at home, before transportation to the hospital) or gastric lavage until the stomach is completely emptied. If finely powdered, activated charcoal is introduced by stomach tube, after lavage, and within 30 minutes after ingestion of the antimalarial, it may inhibit further intestinal absorption of the drug. To be effective, the dose of activated charcoal should be at least five times the estimated dose of chloroquine ingested.
Convulsions, if present, should be controlled before attempting gastric lavage. If due to cerebral stimulation, cautious administration of an ultra short-acting barbituate may be tried, but if due to anoxia, it should be corrected by oxygen administration and artificial respiration. In shock with hypotension, a potent vasopressor should be administered. Because of the importance of supporting respiration, tracheal intubation or tracheostomy, followed by gastric lavage, may also be necessary. Peritoneal dialysis and exchange transfusions have also been suggested to reduce the level of the drug in the blood.
A patient who survives the acute phase and is asymptomatic should be closely observed for at least six hours. Fluids may be forced, and sufficient ammonium chloride (8 g daily in divided doses for adults) may be administered for a few days to acidify the urine to help promote urinary excretion in cases of both overdosage or sensitivity.
CHLOROQUINE PHOSPHATE DOSAGE AND ADMINISTRATION
The dosage of chloroquine phosphate is often expressed in terms of equivalent chloroquine base. Each 250 mg tablet of chloroquine phosphate contains the equivalent of 150 mg chloroquine base. In infants and children the dosage is preferably calculated on the body weight.
Malaria
Suppression-Adult Dose: 500 mg (= 300 mg base) on exactly the same day of each week.
Pediatric Dose: The weekly suppressive dosage is 5 mg calculated as base, per kg of body weight, but should not exceed the adult dose regardless of weight.
If circumstances permit, suppressive therapy should begin two weeks prior to exposure. However, failing this in adults, an initial double (loading) dose of 1 g (= 600 mg base), or in children 10 mg base/kg may be taken in two divided doses, six hours apart. The suppressive therapy should be continued for eight weeks after leaving the endemic area.
For treatment of acute attack, adults
An initial dose of 1 g (= 600 mg base) followed by an additional 500 mg (= 300 mg base) after six to eight hours and a single dose of 500 mg (= 300 mg base) on each of two consecutive days. This represents a total dose of 2.5 g chloroquine phosphate or 1.5 g base in three days.
The dosage for adults may also be calculated on the basis of body weight; this method is preferred for infants and children. A total dose representing 25 mg of base per kg of body weight is administered in three days, as follows:
First dose: 10 mg base per kg (but not exceeding a single dose of 600 mg base).
Second dose: 5 mg base per kg (but not exceeding a single dose of 300 mg base) 6 hours after first dose.
Third dose: 5 mg base per kg 18 hours after second dose.
Fourth dose: 5 mg base per kg 24 hours after third dose.
For radical cure of vivax and malariae malaria concomitant therapy with an 8-aminoquinoline compound is necessary.
Extraintestinal Amebiasis
Adults, 1 g (600 mg base) daily for two days, followed by 500 mg (300 mg base) daily for at least two to three weeks. Treatment is usually combined with an effective intestinal amebicide.
HOW SUPPLIED
Chloroquine Phosphate Tablets USP 250 mg: Each round, single-scored, white to off-white compressed tablet contains 250 mg of chloroquine phosphate, equivalent to 150 mg of chloroquine base. Each tablet is debossed with 0115 2790
Bottles of 50 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .NDC 0115-2790-06
Bottles of 500 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .NDC 0115-2790-02
Store at controlled room temperature, 15°C-30°C (59°-86°F) in a tight, light-resistant container, as defined in the USP.
Mfg. by: IMPAX Laboratories, Inc.
Hayward, CA 94544 USA
Dist. by: Global Pharmaceuticals
Division of IMPAX Laboratories, Inc.
Philadelphia, PA 19124 USA
Rev. 06/03
099-05
PRINCIPAL DISPLAY PANEL - 250 mg Tablet Bottle Label
GLOBAL®
NDC 0115-2790-06
Chloroquine
Phosphate
Tablets, USP
250 mg
Rx only
50 TABLETS

Chloroquine PhosphateCHLOROQUINE PHOSPHATE TABLET
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||