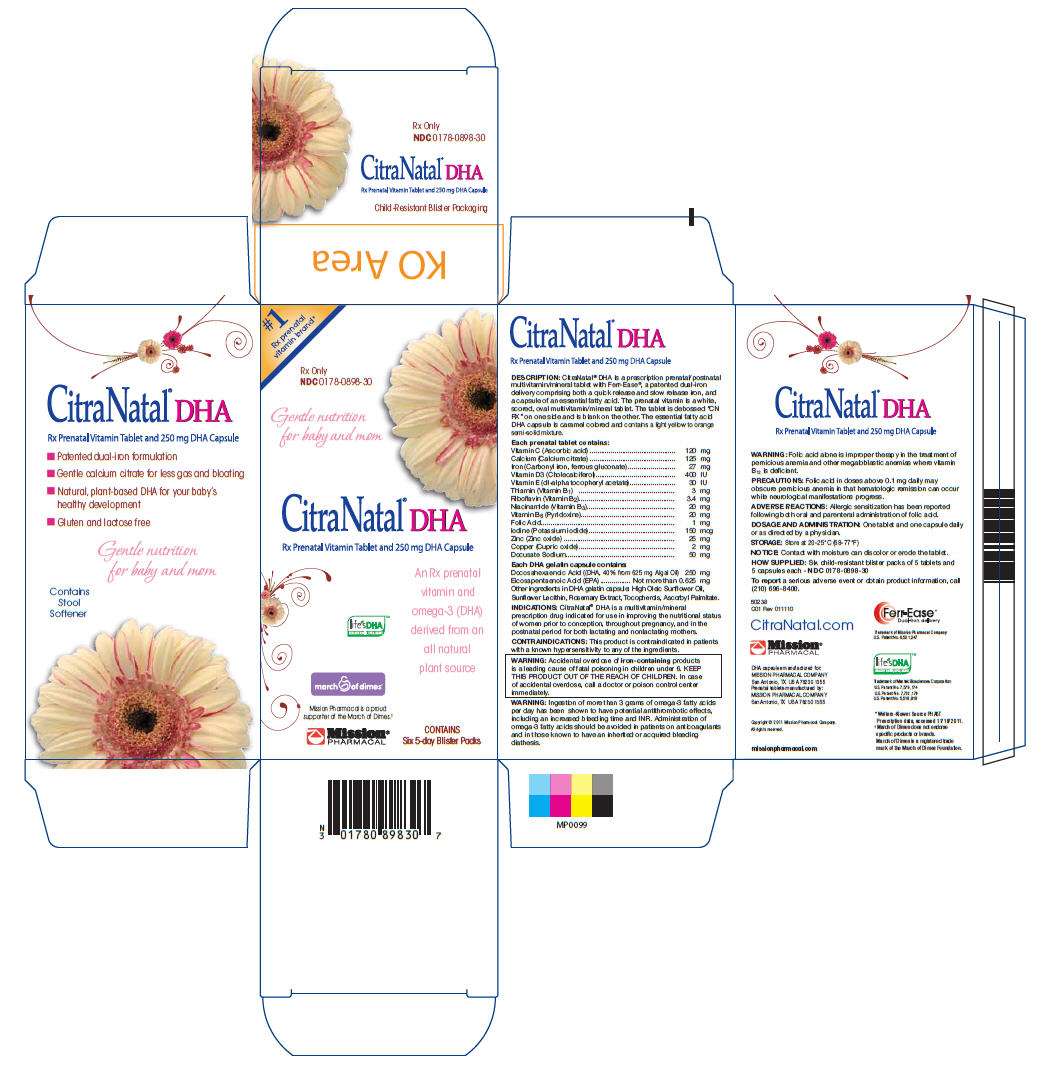
CitraNatal DHA
Mission Pharmacal Company
Mission Pharmacal Company
CitraNatal DHA
FULL PRESCRIBING INFORMATION: CONTENTS*
- CITRANATAL DHA DESCRIPTION
- INDICATIONS
- WARNING
- PRECAUTIONS
- CITRANATAL DHA ADVERSE REACTIONS
- CITRANATAL DHA DOSAGE AND ADMINISTRATION
- STORAGE
- HOW SUPPLIED
FULL PRESCRIBING INFORMATION
Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. KEEP THIS PRODUCT OUT OF THE REACH OF CHILDREN. In case of accidental overdose, call a doctor or poison control center immediately.
CITRANATAL DHA DESCRIPTION
CitraNatal® DHA is a prescription prenatal/postnatal multivitamin/mineral tablet with Ferr-Ease®, a patented dual-iron delivery comprising both a quick release and slow release iron, and a capsule of an essential fatty acid. The prenatal vitamin is a white, scored, oval multivitamin/mineral tablet. The tablet is debossed “CN RX” on one side and is blank on the other. The essential fatty acid DHA capsule is caramel colored and contains a light yellow to orange semi-solid mixture.
| Vitamin C (Ascorbic acid) | 120 mg |
| Calcium (Calcium citrate) | 125 mg |
| Iron (Carbonyl iron, ferrous gluconate) | 27 mg |
| Vitamin D3 (Cholecalciferol) | 400 IU |
| Vitamin E (dl-alpha tocopheryl acetate) | 30 IU |
| Thiamin (Vitamin B1) | 3 mg |
| Riboflavin (Vitamin B2) | 3.4 mg |
| Niacinamide (Vitamin B3) | 20 mg |
| Vitamin B6 (Pyridoxine) | 20 mg |
| Folic Acid | 1 mg |
| Iodine (Potassium iodide) | 150 mcg |
| Zinc (Zinc oxide) | 25 mg |
| Copper (Cupric oxide) | 2 mg |
| Docusate Sodium | 50 mg |
Each DHA gelatin capsule contains:
Docosahexaenoic Acid ((DHA, 40% from 625 mg Algal Oil) 250 mg Eicosapentaenoic Acid (EPA)................. Not more than 0.625 mg Other ingredients in DHA gelatin capsule: High Oleic Sunflower Oil, Sunflower Lecithin, Rosemary Extract, Tocopherols, Ascorbyl Palmitate.
Active ingredient
Purpose
INDICATIONS
[Insert Clinical Pharmacology General text here]
CONTRAINDICATIONS
[Insert Mechanism of Action text here]
WARNING
Ingestion of more than 3 grams of omega-3 fatty acids per day has been shown to have potential antithrombotic effects, including an increased bleeding time and INR. Administration of omega-3 fatty acids should be avoided in patients on anticoagulants and in those known to have an inherited or acquired bleeding diathesis.
WARNING: Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B12 is deficient.
PRECAUTIONS
Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations progress.
CITRANATAL DHA ADVERSE REACTIONS
Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
CITRANATAL DHA DOSAGE AND ADMINISTRATION
One tablet and one capsule daily or as directed by a physician.
STORAGE
Store at 20-25°C (68-77°F)
NOTICE: Contact with moisture can discolor or erode the tablet.
HOW SUPPLIED
Six child-resistant blister packs of 5 tablets and 5 capsules each - NDC 0178-0898-30
To report a serious adverse event or obtain product information, call (210) 696-8400.
500238
C01 Rev 011110
DHA capsules manufactured for:
MISSION PHARMACAL COMPANY
San Antonio, TX USA 78230 1355
Prenatal tablets manufactured by:
MISSION PHARMACAL COMPANY
San Antonio, TX USA 78230 1355
CitraNatal® DHA Carton
NDC: 0178-0898-30
CitraNatal DHAVitamin C, Calcium, Iron, Vitamin D3, Vitamin E, Thiamin, Riboflavin, Niacinamide, Vitamin B6, Folic Acid, Iodine, Zinc, Copper, Docusate Sodium TABLET
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||