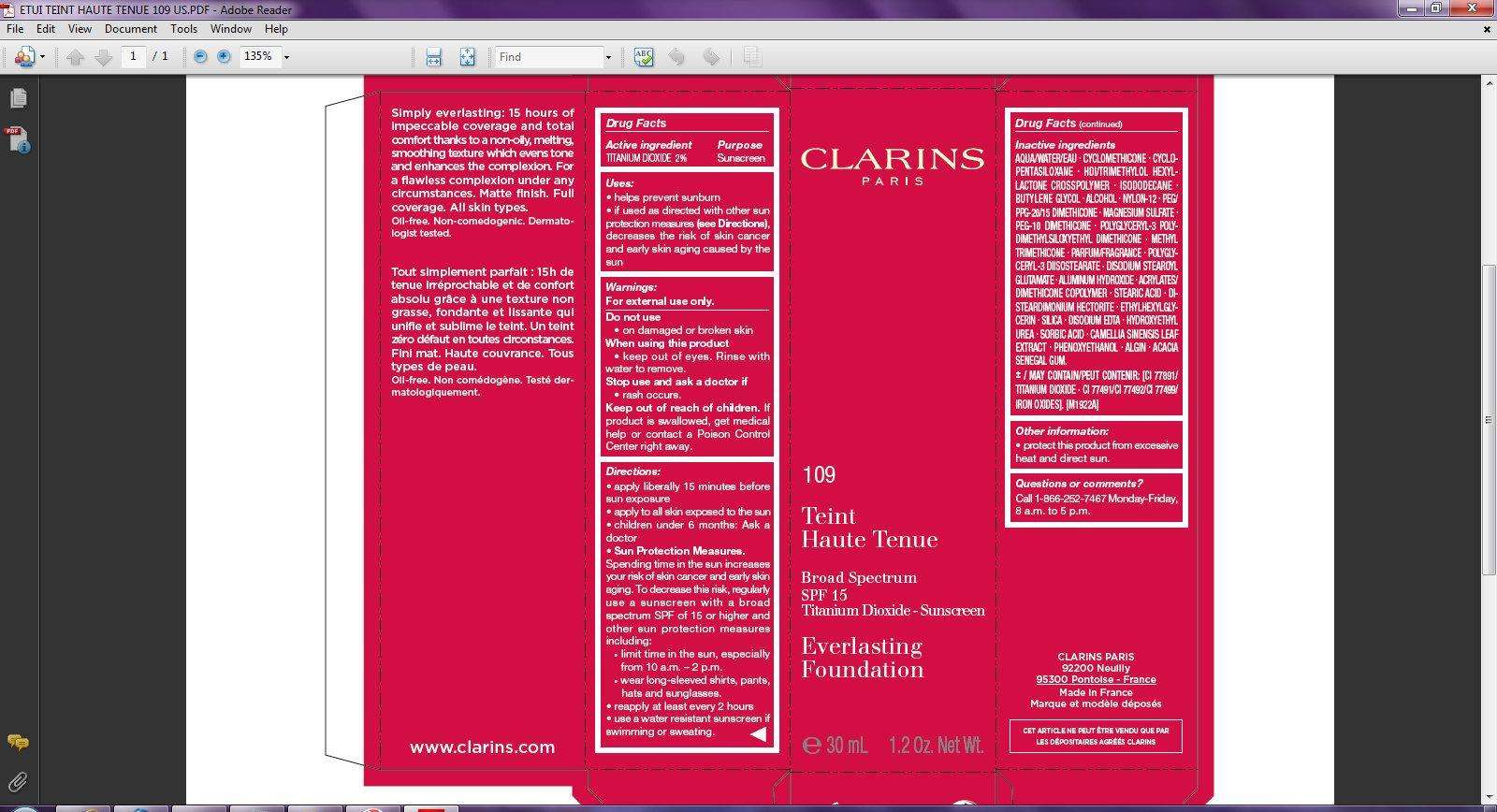
CLARINS Broad Spectrum SPF 15 Everlasting Foundation Tint 109
CLARINS Broad Spectrum SPF 15 Everlasting Foundation Tint 109
FULL PRESCRIBING INFORMATION: CONTENTS*
- Drug Facts
- Active ingredient
- Purpose
- CLARINS Broad Spectrum SPF 15 Everlasting Foundation Tint 109 Uses:
- Warnings:
- Directions:
- Inactive ingredients
- CLARINS Broad Spectrum SPF 15 Everlasting Foundation Tint 109 Other information:
- Questions or comments?
- CLARINS
- Product Labels
FULL PRESCRIBING INFORMATION
Drug Facts
Active ingredient
TITANIUM DIOXIDE 2%
Purpose
Sunscreen
Uses:
• helps prevent sunburn
• if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
Warnings:
For external use only.
Do not use
• on damaged or broken skin
When using this product
• keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if
• rash occurs.
Keep out of reach of children.
If product is swallowed, get medical help or contact a Poison Control Center right away.
Directions:
• apply liberally 15 minutes before sun exposure
• apply to all skin exposed to the sun
• children under 6 months: Ask a doctor
• Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m. – 2 p.m.
• wear long-sleeved shirts, pants, hats and sunglasses.
• reapply at least every 2 hours
• use a water resistant sunscreen if swimming or sweating.
Inactive ingredients
AQUA/WATER/EAU - CYCLOMETHICONE - CYCLOPENTASILOXANE - HDI/TRIMETHYLOL HEXYLLACTONE CROSSPOLYMER - SODODECANE - BUTYLENE GLYCOL - ALCOHOL - NYLON-12 - PEG/PPG-20/15 DIMETHICONE - MAGNESIUM SULFATE - PEG-10 DIMETHICONE - POLYGLYCERYL-3 POLYDIMETHYLSILOXYETHYL DIMETHICONE - METHYL TRIMETHICONE - PARFUM/FRAGRANCE - POLYGLYCERYL- 3 DIISOSTEARATE - DISODIUM STEAROYL GLUTAMATE - ALUMINUM HYDROXIDE - ACRYLATES/DIMETHICONE COPOLYMER - STEARIC ACID - DIDISTEARDIMONIUM HECTORITE - ETHYLHEXYLGLYCERIN - SILICA - DISODIUM EDTA - HYDROXYETHYL UREA - SORBIC ACID - CAMELLIA SINENSIS LEAF EXTRACT - PHENOXYETHANOL - ALGIN - ACACIA SENEGAL GUM.
± / MAY CONTAIN/PEUT CONTENIR: [CI 77891/TITANIUM DIOXIDE . CI 77491/CI 77492/CI 77499/IRON OXIDES]. [M1922A]
Other information:
• protect this product from excessive heat and direct sun.
Questions or comments?
Call 1-866-252-7467 Monday-Friday, 8 a.m. to 5 p.m.
CLARINS
Simply everlasting: 15 hours of impeccable coverage and total comfort thanks to a non-oily, melting, smoothing texture which evens tone and enhances the complexion. For a flawless complexion under any circumstances. Matte finish. Full coverage. All skin types. Oil-free. Non-comedogenic. Dermatologist tested. www.clarins.com CLARINS PARIS 92200 Neuilly 95300 Pontoise - France Made in France
CLARINS PARIS 109 Teint Haute Tenue Broad Spectrum SPF 15 Titanium Dioxide - Sunscreen Everlasting Foundation e 30 mL 1.2 Oz. Net Wt.
Product Labels

CLARINS Broad Spectrum SPF 15 Everlasting Foundation Tint 109TITANIUM DIOXIDE LIQUID
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||