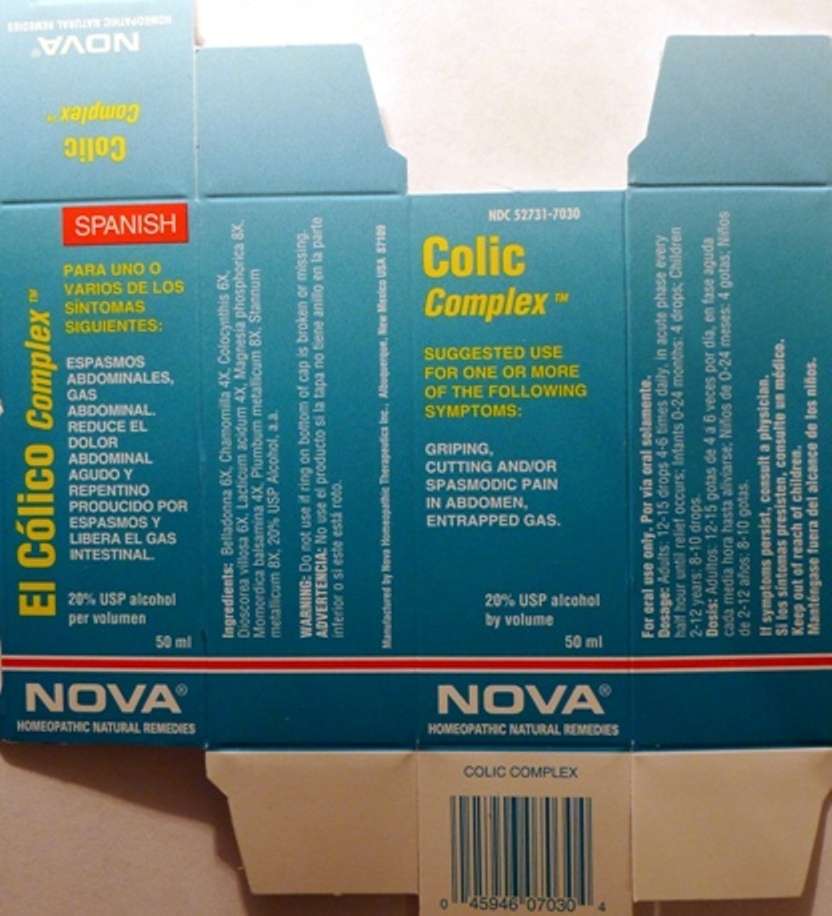
Colic Complex
Nova Homeopathic Therapeutics, Inc.
Nova Homeopathic Therapeutics, Inc.
Colic Complex
FULL PRESCRIBING INFORMATION: CONTENTS*
FULL PRESCRIBING INFORMATION
Purpose:
Suggested use for one or more of the following symptoms:
Abdominal spasms, entrapped gas.
Usage and Dosage:
For oral use only.
Adults:
In Acute Phase:
12-15 drops, every half hour until relief occurs
When Relief Occurs:
12-15 drops, 4-6 times daily
Children 2-12 years:
8-10 drops, 4-6 times daily
Infants 0-24 months:
4 drops, 4-6 times daily
Warnings:
Keep out of reach of children.
If symptoms persist, consult a physician.
Do not use if ring on bottom of cap is broken or missing.
Active Ingredients:
Belladonna 6X, Chamomilla 4X, Colocynthis 6X, Dioscorea villosa 6X, Lacticum acidum 4X, Magnesia phosphorica 8X, Momordica balsamina 4X, Plumbum metallicum 8X, Stannum metallicum 8X
Inactive Ingredients:
Alcohol, 20% USP
Colic Complex Product

Colic Complex Bottle Label

Colic Complex Box
Colic ComplexBelladonna, Chamomilla, Colocynthis, Dioscorea villosa, Lacticum acidum, Magnesia phosphorica, Momordica balsamina, Plumbum metallicum, Stannum metallicum LIQUID
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||