Dermazene
Dermazene Cream (hydrocortisone 1% – iodoquinol 1%)
FULL PRESCRIBING INFORMATION: CONTENTS*
- DERMAZENE DESCRIPTION
- CLINICAL PHARMACOLOGY
- DERMAZENE CONTRAINDICATIONS
- WARNINGS AND PRECAUTIONS
- DERMAZENE ADVERSE REACTIONS
- DERMAZENE DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- PRINCIPAL DISPLAY PANEL - 28.4 gram Box
FULL PRESCRIBING INFORMATION
Rx only
DERMAZENE DESCRIPTION
Each gram of Dermazene® Cream 1% contains 10 mg of hydrocortisone and 10 mg of iodoquinol in a cream base of purified water, propylene glycol, cerasynt SE, amerchol L101, isopropyl palmitate, cetyl alcohol, arlacel 60, myrj 52, tween 60, sorbic acid, methyl paraben and propyl paraben.
Chemically, hydrocortisone is [Pregn-4-ene-3,20-dione, 11, 17, 21-trihydroxy-(11β)-] with the molecular formula (C21H30O5) and is represented by the following structural formula:
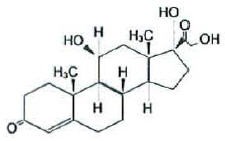
and iodoquinol, 5,7-diiodo-8-quinolinol (C9H5I2NO) is represented by the following structure:

Hydrocortisone is an anti-inflammatory and antipruritic agent, while iodoquinol is an antifungal and antibacterial agent.
CLINICAL PHARMACOLOGY
Hydrocortisone has anti-inflammatory, antipruritic and vasoconstrictor properties. The mechanism of anti-inflammatory activity is unclear. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man.
Iodoquinol has both antifungal and antibacterial properties.
Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings.
Hydrocortisone can be absorbed from normal intact skin. Inflammation and/or other inflammatory disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids.
Once absorbed through the skin, hydrocortisone is metabolized in the liver and most body tissues to hydrogenated and degraded forms such as tetrahydrocortisone and tetrahydrocortisol. These are excreted in the urine, mainly conjugated as glucuronides, together with a very small proportion of unchanged hydrocortisone.
There are no data available regarding the percutaneous absorption of Iodoquinol; however, following oral administration, 3-5% of the dose was recovered in the urine as a glucuronide.
Uses
| INDICATIONS AND USAGE |
| Based on a review of a related drug by the National Research Council and subsequent FDA classification for that drug, the indications are as follows: "Possibly" Effective: Contact or atopic dermatitis; impetiginized eczema; nummular eczema; infantile eczema; endogenous chronic infectious dermatitis; stasis dermatitis; pyoderma; nuchal eczema and chronic eczematoid otitis externa; acne urticata; localized or disseminated neurodermatitis; lichen simplex chronicus; anogenital pruritus (vulvae, scroti, ani); folliculitis; bacterial declatoses; mycotic dermatoses such as tinea (capitis, cruris, corporis, pedis); moniliasis, intertrigo. Final classification of the less-than-effective indications requires further investigation. |
DERMAZENE CONTRAINDICATIONS
Dermazene Cream is contraindicated in those patients with a history of hypersensitivity to hydrocortisone, iodoquinol or any other components of the preparation.
WARNINGS AND PRECAUTIONS
For external use only. Keep away from eyes. If irritation develops, the use of Dermazene Cream should be discontinued and appropriate therapy instituted. Staining of the skin and fabrics may occur. If extensive areas are treated or if the occlusive technique is used, the possibility exists of increased systemic absorption of the corticosteroid, and suitable precautions should be taken. Children may absorb proportionally larger amounts of the corticosteroids and thus be more susceptible to systemic toxicity. Parents of pediatric patients should be advised not to use tightfitting diapers or plastic pants on a child being treated in the diaper area, as these garments may constitute occlusive dressings. Iodoquinol may be absorbed through the skin and interfere with thyroid function tests. If such tests are contemplated, wait at least one month after discontinuance of therapy to perform these tests. The ferric chloride test for phenylketonuria (PKU) can yield a false positive result if iodoquinol is present in the diaper or urine.
Prolonged use may result in overgrowth of non-susceptible organisms requiring appropriate therapy. Keep out of reach of children.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Long term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of hydrocortisone or Iodoquinol.
In vitro studies to determine mutagenicity with hydrocortisone have revealed negative results. Mutagenicity studies have not been conducted with iodoquinol.
Pregnancy Category C
Animal reproductive studies have not been conducted with Dermazene Cream. It is not known whether Dermazene Cream can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. Dermazene Cream should be given to a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Dermazene Cream is administered to a nursing woman.
Pediatric Use
Safety and effectiveness in children under the age of 12 have not been established.
DERMAZENE ADVERSE REACTIONS
The following local adverse reactions are reported infrequently with topical corticosteroids. These reactions are listed in an approximate decreasing order of occurrence:
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
DERMAZENE DOSAGE AND ADMINISTRATION
Apply to affected area 3 to 4 times daily in accordance with physician's directions.
HOW SUPPLIED
| SIZE | NDC NUMBER |
|---|---|
| 1 OZ tube | 58980-811-10 |
Distributed by:
STRATUS
PHARMACEUTICALS INC
Manufactured by: Sonar Products Inc., Carlstadt, NJ 07072 for
Stratus Pharmaceuticals Inc., 14377 Southwest 142nd Street,
Miami, Florida 33186-6727
Customer Service: 1-800-442-7882 / fax 305-254-6875
©1999 Stratus Pharmaceuticals Inc
JG-DCI2007-06 (12/99)
PRINCIPAL DISPLAY PANEL - 28.4 gram Box
NDC 58980-811-10
NET WT 1.0 OZ (28.4 grams)
DERMAZENE™ CREAM
(hydrocortisone 1% - iodoquinol 1%)
Rx only
Distributed by STRATUS PHARMACEUTICALS INC. 14377 Southwest 142nd Street, Miami, Florida 33186-6727
DERMAZENE
1 OZ TUBE

Dermazenehydrocortisone and Iodoquinol CREAM
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||