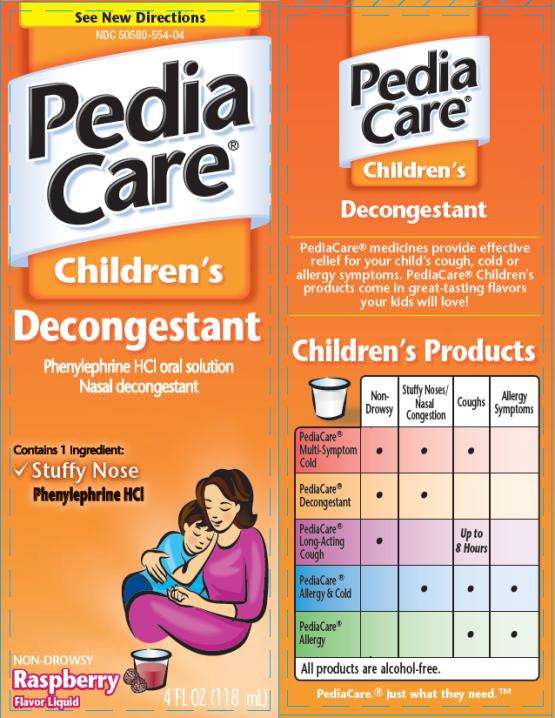
diaCare Childrens Decongestant
PediaCare Childrens Decongestant
FULL PRESCRIBING INFORMATION
PediaCare Children’s Decongestant
Drug Facts
Phenylephrine HCl 2.5 mg
Nasal decongestant
*5 mL = one teaspoon
temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
Do not use
- in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your child’s prescription drug contains an MAOI, ask a doctor or pharmacist before giving this product.
if your child has
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- a sodium-restricted diet
do not exceed recommended dose
- nervousness, dizziness, or sleeplessness occur
- symptoms do not improve within 7 days or occur with a fever
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
- find right dose on chart below
- use only enclosed dosing cup designed for use with this product. Do not use any other dosing device.
- if needed, repeat dose every 4 hours
- do not give more than 6 times in 24 hours
| Age | Does (tsp) |
| Under 4 years | Do not use |
| 4 to 5 years | 1 teaspoonful (5 mL) |
| 6 to 11 years | 2 teaspoonfuls (10 mL) |
Attention: use only enclosed dosing cup specifically designed for use with this product. Do not use any other dosing device.
- each teaspoon contains: sodium 14 mg
- store between 20-25°C (68-77°F). Protect from light. Store in outer carton until contents are used.
-
do not use if bottle wrap, or foil inner seal imprinted
“Safety Seal®” is broken or missing
- see bottom panel for lot number and expiration date
carboxymethylcellulose sodium, citric acid, edetate disodium, FD&C red #40, flavors, glycerin, sodium benzoate, sodium citrate, sorbitol, sucralose, water
call 1-888-474-3099
See New Directions
PediaCare
Children’s
Decongestant
Phenylephrine
HCI oral solution
4 FL OZ (118
mL
)
diaCare Childrens DecongestantPhenylephrine HCl LIQUID
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||