Digiclean
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active Ingredient
- Purpose
- Digiclean Uses
- Warnings
- Directions
- Digiclean Other information
- Inactive ingredients
- Principal Display Panel and Representative Label
FULL PRESCRIBING INFORMATION
Active Ingredient
Triclosan, 0.3%
Purpose
Antiseptic handwash
Digiclean Uses
- for handwashing to decrease bacteria on the skin
Warnings
-
For external use only
Do not use
- In eyes
When using this product
- if in eyes, rinse promptly and thoroughly with water
- discontinue use if irritation and redness develop
Stop use and ask a doctor if
- skin irritation and redness persist for more than 72 hours
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.
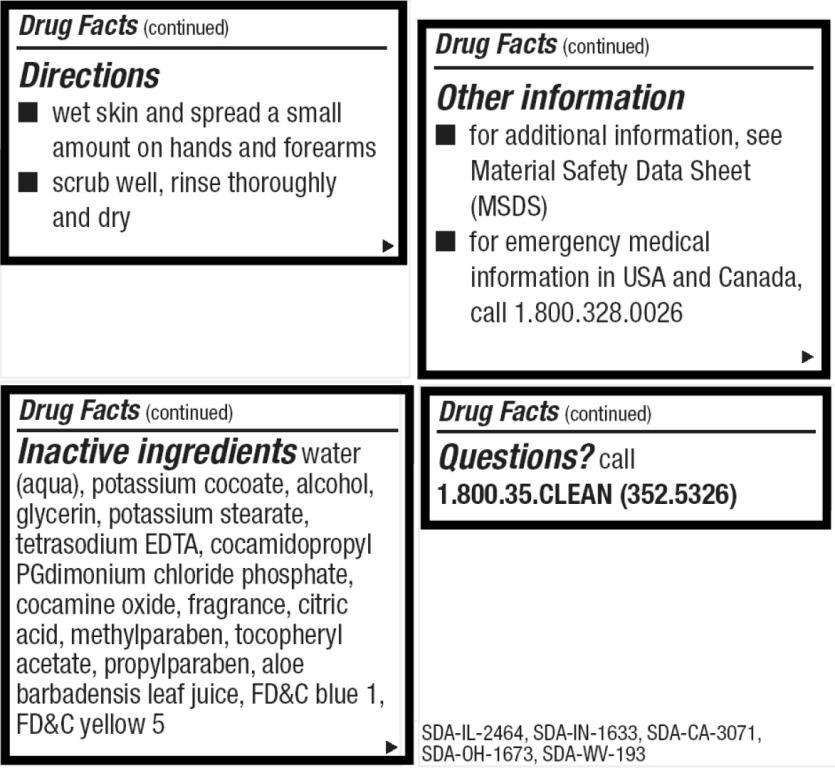
Directions
- wet skin and spread a small amount on hands and forearms
- scrub well, rinse throughly and dry
Digiclean Other information
- for additional information, see Material Safety Data Sheet (MSDS)
- for emergency medical information in USA and Canada, call 1.800.328.0026
Inactive ingredients
water, potassium cocoate, alcohol, glycerin, potassium stearate, tetrasodium EDTA, cocamidopropyl PG-dimonium chloride phosphate, cocamine oxide, fragrance, citric acid, methylparaben, tocopheryl acetate, propylparaben, aloe barbadensis leaf juice, FDC blue 1, FDC yellow 5
Questions?
call 1.800.35.CLEAN (352.5326)
Principal Display Panel and Representative Label
DIGITS
NDC 47593-390-41
DigiClean
Healthcare Personnel Handwash Foam
22638
Net Contents: 750 mL/25 fl oz
ECOLAB
Ecolab, 370 Wabasha Street N, St Paul MN 55102-1390 USA
copyright 2010 Ecolab USA Inc, All rights reserved
Made in United States, 707008/5403/0710

DigicleanTriclosan SOLUTION
| |||||||||||||||||||||||||||||||||||||||||||||||||