Diltiazem Hydrochloride
FULL PRESCRIBING INFORMATION: CONTENTS*
- DILTIAZEM HYDROCHLORIDE DESCRIPTION
- CLINICAL PHARMACOLOGY
- INDICATIONS & USAGE
- DILTIAZEM HYDROCHLORIDE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- DRUG INTERACTIONS
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- PREGNANCY
- NURSING MOTHERS
- PEDIATRIC USE
- GERIATRIC USE
- DILTIAZEM HYDROCHLORIDE ADVERSE REACTIONS
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- STORAGE AND HANDLING
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
DILTIAZEM HYDROCHLORIDE DESCRIPTION
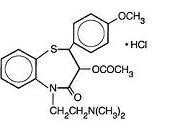
CLINICAL PHARMACOLOGY
Mechanisms of Action
Angina Due to Coronary Artery Spasm:Diltiazem has been shown to be a potent dilator of coronary arteries both epicardial and subendocardial. Spontaneous and ergonovine-induced coronary artery spasm are inhibited by diltiazem.
Exertional Angina:Diltiazem has been shown to produce increases in exercise tolerance, probably due to its ability to reduce myocardial oxygen demand. This is accomplished via reductions in heart rate and systemic blood pressure at submaximal and maximal exercise workloads.
Hemodynamic and Electrophysiologic Effects
Pharmacokinetics and Metabolism
INDICATIONS & USAGE
DILTIAZEM HYDROCHLORIDE CONTRAINDICATIONS
WARNINGS
1.Cardiac Conduction:Diltiazem prolongs AV node refractory periods without significantly prolonging sinus node recovery time, except in patients with sick sinus syndrome. This effect may rarely result in abnormally slow heart rates (particularly in patients with sick sinus syndrome) or second- or third-degree AV block (six of 1243 patients for 0.48%). Concomitant use of diltiazem with beta-blockers or digitalis may result in additive effects on cardiac conduction. A patient with Prinzmetal's angina developed periods of asystole (2 to 5 seconds) after a single dose of 60 mg of diltiazem (seeADVERSE REACTIONS).2.Congestive Heart Failure:Although diltiazem has a negative inotropic effect in isolated animal tissue preparations, hemodynamic studies in humans with normal ventricular function have not shown a reduction in cardiac index nor consistent negative effects on contractility (dp/dt). Experience with the use of diltiazem alone or in combination with beta-blockers in patients with impaired ventricular function is very limited. Caution should be exercised when using the drug in such patients.
3.Hypotension:Decreases in blood pressure associated with diltiazem therapy may occasionally result in symptomatic hypotension.
4.Acute Hepatic Injury:In rare instances, significant elevations in enzymes such as alkaline phosphatase, LDH, SGOT, SGPT, and other phenomena consistent with acute hepatic injury have been noted. These reactions have been reversible upon discontinuation of drug therapy. The relationship to diltiazem is uncertain in most cases, but probable in some (seePRECAUTIONS).
PRECAUTIONS
GeneralDilitiazem is extensively metabolized by the liver and excreted by the kidneys and in bile. As with any drug given over prolonged periods, laboratory parameters of renal and hepatic function should be monitored at regular intervals. The drug should be used with caution in patients with impaired renal or hepatic function. In subacute and chronic dog and rat studies designed to produce toxicity, high doses of dilitiazem were associated with histological changes in the liver, which were reversible when the drug was discontinued. In dogs, doses of 20 mg/kg were also associated with hepatic changes; however, these changes were reversible with continued dosing. Dermatological events (see ADVERSE REACTIONS) may be transient and may disappear despite continued use of diltiazem. However, skin eruptions progressing to erythema multiforme and/or exfoliative dermatitis have also been infrequently reported. Should a dermatologic reaction persist, the drug should be discontinued.
DRUG INTERACTIONS
WARNINGS).WARNINGS).
WARNINGS).
WARNINGS).
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
PREGNANCY
NURSING MOTHERS
PEDIATRIC USE
GERIATRIC USE
DILTIAZEM HYDROCHLORIDE ADVERSE REACTIONS
Cardiovascular:Angina, arrhythmia, AV block (first-degree), AV block (second- or third-degreeseeWARNINGS, Cardiac Conduction), bradycardia, bundle branch block, congestive heart failure, ECG abnormality, flushing, hypotension, palpitations, syncope, tachycardia, ventricular extrasystoles.
Nervous System:Abnormal dreams, amnesia, depression, gait abnormality, hallucinations, insomnia, nervousness, paresthesia, personality change, somnolence, tremor.
Gastrointestinal:Anorexia, constipation, diarrhea, dysgeusia, dyspepsia, mild elevations of alkaline phosphatase, SGOT, SGPT, and LDH (seeWARNINGS, Acute Hepatic Injury), thirst, vomiting, weight increase.
Dermatological:Petechiae, photosensitivity, pruritus, urticaria.
Other:Amblyopia, CPK elevation, dry mouth, dyspnea, epistaxis, eye irritation, hyperglycemia, hyperuricemia, impotence, muscle cramps, nasal congestion, nocturia, osteoarticular pain, polyuria, sexual difficulties, tinnitus.
OVERDOSAGE
Bradycardia:Administer atropine (0.6 to 1 mg). If there is no response to vagal blockade, administer isoproterenol cautiously.
High-Degree AV Block:Treat as for bradycardia above. Fixed high-degree AV block should be treated with cardiac pacing.
Cardiac Failure:Administer inotropic agents (isoproterenol, dopamine, or dobutamine) and diuretics.
Hypotension:Vasopressors (e.g., dopamine or norepinephrine).
DOSAGE & ADMINISTRATION
Exertional Angina Pectoris Due to Atherosclerotic Coronary Artery Disease or Angina Pectoris at Rest Due to Coronary Artery SpasmConcomitant Use With Other Cardiovascular Agents
Sublingual NTGmay be taken as required to abort acute anginal attacks during diltiazem therapy.
Prophylactic Nitrate Therapy:Diltiazem may be safely coadministered with short- and long-acting nitrates, but there have been no controlled studies to evaluate the antianginal effectiveness of this combination.
Beta-blockers.(SeeWARNINGSandPRECAUTIONS.)
HOW SUPPLIED
STORAGE AND HANDLING
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION


Diltiazem HydrochlorideDILTIAZEM HYDROCHLORIDE TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!