DIOR SNOW PROTECTION UV
NIPPON SHIKIZAI INC.
CHRISTIAN DIOR INC.
DIORSNOW WHITE REVEAL UV SHIELD
FULL PRESCRIBING INFORMATION: CONTENTS*
- WARNINGS
- ACTIVE INGREDIENTS
- OTHER INGREDIENTS
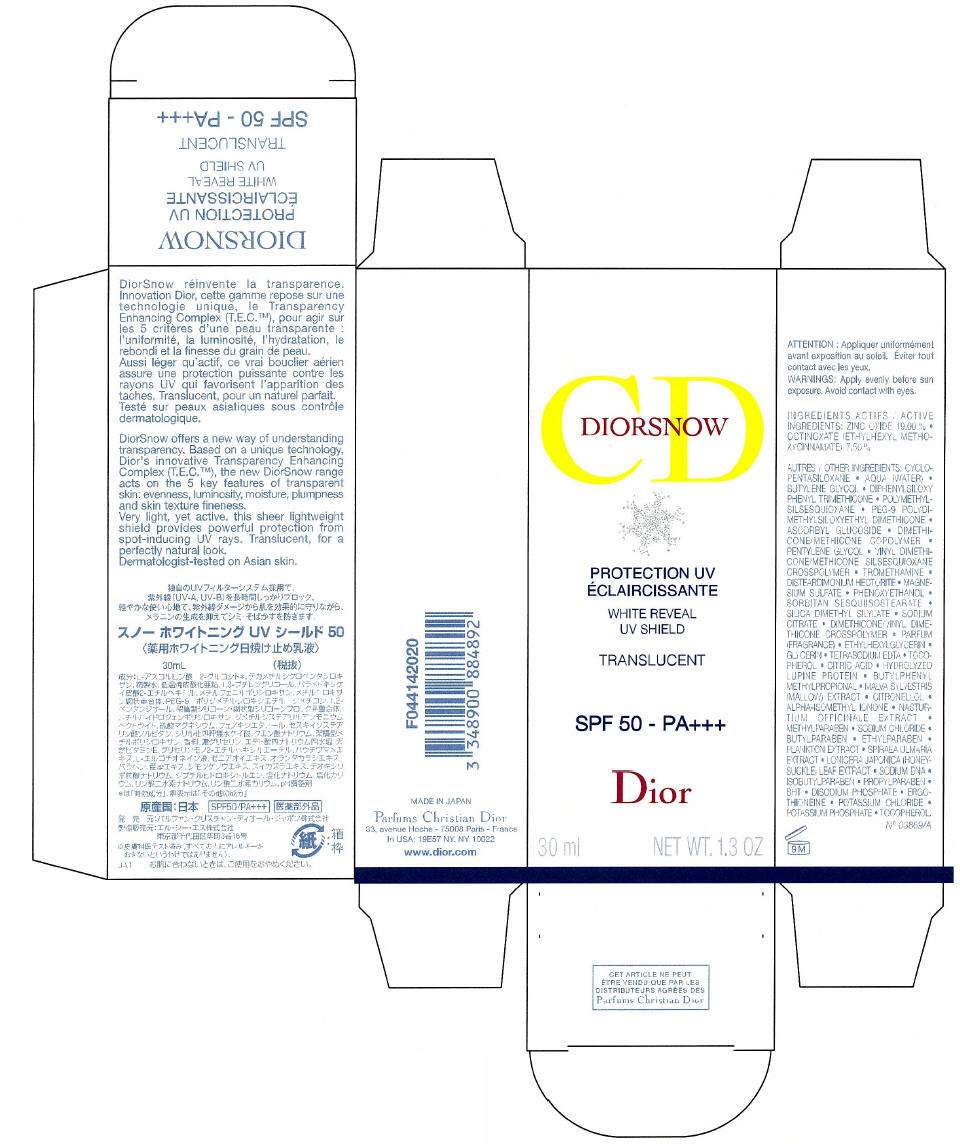
- PRINCIPAL DISPLAY PANEL - 30 ml Bottle Box (Translucent)
- PRINCIPAL DISPLAY PANEL - 30 ml Bottle Box (Pearly White)
- PRINCIPAL DISPLAY PANEL - 30 ml Bottle Box (Beige)
- PRINCIPAL DISPLAY PANEL - 30 ml Bottle Box (Mauve)
FULL PRESCRIBING INFORMATION
WARNINGS
Apply evenly before sun exposure. Avoid contact with eyes.
ACTIVE INGREDIENTS
ZINC OXIDE 19.00 %, OCTINOXATE (ETHYLHEXYL METHOXYCINNAMATE) 7.50 %
OTHER INGREDIENTS
CYCLOPENTASILOXANE, AQUA (WATER), BUTYLENE GLYCOL, DIPHENYLSILOXY PHENYL TRIMETHICONE, POLYMETHYLSILSESQUIOXANE, PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE, ASCORBYL GLUCOSIDE, DIMETHICONE/METHICONE COPOLYMER, PENTYLENE GLYCOL, VINYL DIMETHICONE/METHICONE SILSESQUIOXANE CROSSPOLYMER, TROMETHAMINE, DISTEARDIMONIUM HECTORITE, MAGNESIUM SULFATE, PHENOXYETHANOL, SORBITAN SESQUIISOSTEARATE, SILICA DIMETHYL SILYLATE, SODIUM CITRATE, DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER, PARFUM (FRAGRANCE), ETHYLHEXYLGLYCERIN, GLYCERIN, TETRASODIUM EDTA, TOCOPHEROL, CITRIC ACID, HYDROLYZED LUPINE PROTEIN, BUTYLPHENYL METHYLPROPIONAL, MALVA SYLVESTRIS (MALLOW) EXTRACT, CITRONELLOL, ALPHA-ISOMETHYL IONONE, NASTURTIUM OFFICINALE EXTRACT, METHYLPARABEN, SODIUM CHLORIDE, BUTYLPARABEN, ETHYLPARABEN, PLANKTON EXTRACT, SPIRAEA ULMARIA EXTRACT, LONICERA JAPONICA (HONEYSUCKLE) LEAF EXTRACT, SODIUM DNA, ISOBUTYLPARABEN, PROPYLPARABEN, BHT, DISODIUM PHOSPHATE, ERGOTHIONEINE, POTASSIUM CHLORIDE, POTASSIUM PHOSPHATE, TOCOPHEROL.
N° 03869/A
PRINCIPAL DISPLAY PANEL - 30 ml Bottle Box (Translucent)
CD
DIROSNOW
PROTECTION UV
WHITE REVEAL
UV SHIELD
TRANSLUCENT
SPF 50 - PA+++
Dior
30 ml
NET WT. 1.3 OZ
PRINCIPAL DISPLAY PANEL - 30 ml Bottle Box (Pearly White)
CD
DIROSNOW
PROTECTION UV
WHITE REVEAL
UV SHIELD
PEARLY WHITE
SPF 50 - PA+++
Dior
30 ml
NET WT. 1.3 OZ

PRINCIPAL DISPLAY PANEL - 30 ml Bottle Box (Beige)
CD
DIROSNOW
BASE UV
WHITE REVEAL
MAKEUP UV BASE
BEIGE
SPF 35 - PA+++
Dior
30 ml
1 FL OZ

PRINCIPAL DISPLAY PANEL - 30 ml Bottle Box (Mauve)
CD
DIROSNOW
BASE UV
WHITE REVEAL
MAKEUP UV BASE
MAUVE
SPF 35 - PA+++
Dior
30 ml
1 FL OZ

DIOR SNOW PROTECTION UVOCTINOXATE and ZINC OXIDE CREAM
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DIOR SNOW PROTECTION UVOCTINOXATE and ZINC OXIDE CREAM
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DIOR SNOW PROTECTION UVOCTINOXATE and ZINC OXIDE CREAM
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DIOR SNOW PROTECTION UVOCTINOXATE and ZINC OXIDE CREAM
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||