EMBEDA
FULL PRESCRIBING INFORMATION
EMBEDA Capsules contain pellets of morphine sulfate and naltrexone hydrochloride at a ratio of 100:4. Morphine sulfate is an agonist and naltrexone hydrochloride is an antagonist at the mu-opioid receptor.
The chemical name of morphine sulfate is 7,8-didehydro-4,5 α-epoxy-17-methyl-morphinan-3,6 α-diol sulfate (2:1) (salt) pentahydrate. The empirical formula is (C17H19NO3)2●H2SO4●5H2O and its molecular weight is 758.85.
Morphine sulfate is an odorless, white, crystalline powder with a bitter taste. It has a solubility of 1 in 21 parts of water and 1 in 1000 parts of alcohol, but is practically insoluble in chloroform or ether. The octanol: water partition coefficient of morphine is 1.42 at physiologic pH and the pKb is 7.9 for the tertiary nitrogen (mostly ionized at pH 7.4). Its structural formula is:
 STRUCTURE IMAGE 1
STRUCTURE IMAGE 1
The chemical name of naltrexone hydrochloride is (5α)-17-(Cyclopropylmethyl)-4,5-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride. The empirical formula is C20H23NO4•HCl and its molecular weight is 377.46.
Naltrexone hydrochloride is a white to slightly off-white powder that is soluble in water. Its structural formula is
 STRUCTURE IMAGE 2
STRUCTURE IMAGE 2
Each capsule contains the following inactive ingredients common to all strengths: talc, ammonio methacrylate copolymer, sugar spheres, ethylcellulose, sodium chloride, polyethylene glycol, hydroxypropyl cellulose, dibutyl sebacate, methacrylic acid copolymer, diethyl phthalate, magnesium stearate, sodium lauryl sulfate, and ascorbic acid. The capsule shells contain gelatin, titanium dioxide, and grey ink, D and C yellow #10 (EMBEDA 20 mg/0.8 mg), FD and C red #3, FD and C blue #1 (EMBEDA 30 mg/1.2 mg), D and C red #28, FD and C red #40, FD and C blue #1 (EMBEDA 50 mg/2 mg), D and C red #28, FD and C red #40, FD and C blue #1 (EMBEDA 60 mg/2.4 mg), FD and C blue #1, FD and C red #40, FD and C yellow #6 (EMBEDA 80 mg/3.2 mg), D and C yellow #10, FD and C blue #1 (EMBEDA 100 mg/4 mg).
EMBEDA contains no gluten.
Morphine sulfate, a pure opioid agonist, is relatively selective for the mu receptor, although it can interact with other opioid receptors at higher doses. In addition to analgesia, the widely diverse effects of morphine sulfate include analgesia, dysphoria, euphoria, somnolence, respiratory depression, diminished gastrointestinal motility, altered circulatory dynamics, histamine release, physical dependence, and alterations of the endocrine and autonomic nervous systems.
Morphine produces both its therapeutic and its adverse effects by interaction with one or more classes of specific opioid receptors located throughout the body. Morphine acts as a pure agonist, binding with and activating opioid receptors at sites in the peri-aqueductal and peri-ventricular grey matter, the ventro-medial medulla and the spinal cord to produce analgesia.
Effects on the Central Nervous System
The principal actions of therapeutic value of morphine are analgesia and sedation (i.e., sleepiness and anxiolysis). Specific CNS opiate receptors and endogenous compounds with morphine-like activity have been identified throughout the brain and spinal cord and are likely to play a role in the expression of analgesic effects. In addition, when morphine binds to mu-opioid receptors, it results in positive subjective effects, such as drug liking, euphoria, and high. Morphine produces respiratory depression by direct action on brainstem respiratory centers. The mechanism of respiratory depression involves a reduction in the responsiveness of the brainstem respiratory centers to increases in carbon dioxide tension, and to electrical stimulation. Morphine depresses the cough reflex by direct effect on the cough center in the medulla. Antitussive effects may occur with doses lower than those usually required for analgesia. Morphine causes miosis, even in total darkness, and little tolerance develops to this effect. Pinpoint pupils are a sign of opioid overdose but are not pathognomonic (e.g., pontine lesions of hemorrhagic or ischemic origins may produce similar findings). Marked mydriasis rather than miosis may be seen with worsening hypoxia in the setting of EMBEDA overdose [see OVERDOSAGE (10)].
Effects on the Gastrointestinal Tract and Other Smooth Muscle
Gastric, biliary, and pancreatic secretions are decreased by morphine. Morphine causes a reduction in motility associated with an increase in tone in the antrum of the stomach and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. Propulsive peristaltic waves in the colon are decreased, while tone is increased to the point of spasm. The end result is constipation. Morphine can cause a marked increase in biliary tract pressure as a result of spasm of the sphincter of Oddi.
Effects on the Cardiovascular System
Morphine produces peripheral vasodilation which may result in orthostatic hypotension or syncope. Release of histamine may be induced by morphine and can contribute to opioid-induced hypotension. Manifestations of histamine release and/or peripheral vasodilation may include pruritus, flushing, red eyes and sweating.
Mechanism of Action of Naltrexone
Naltrexone is a pure, centrally acting mu-opioid antagonist that reverses the subjective and analgesic effects of mu-opioid receptor agonists by competitively binding at mu-opioid receptors.
Plasma Level-Analgesia Relationships
In any particular patient, both analgesic effects and plasma morphine concentrations are related to the morphine dose.
While plasma morphine-efficacy relationships can be demonstrated in non-tolerant individuals, they are influenced by a wide variety of factors and are not generally useful as a guide to the clinical use of morphine. The effective dose in opioid-tolerant patients may be 10-50 times as great (or greater) than the appropriate dose for opioid-naïve individuals. Dosages of morphine should be chosen and must be titrated on the basis of clinical evaluation of the patient and the balance between therapeutic and adverse effects.
For any fixed dose and dosing interval, EMBEDA will have, at steady-state, a lower Cmax and a higher Cmin than conventional immediate-release morphine.
The pharmacodynamic effect of naltrexone in the setting of crushed EMBEDA was examined in two clinical trials. In a randomized double-blind, triple-dummy, four-way cross-over study, 32 non-dependent recreational opioid users received 120 mg of EMBEDA whole and crushed, 120 mg of immediate-release morphine sulfate and placebo. Overall, 87.5% of subjects had some degree of reduced drug liking after receiving crushed EMBEDA, while 12.5% had no reduction in drug liking. There was considerable individual variability in the degree of reduction in drug liking, ranging between 10 and 50%. Similarly, 69% of subjects showed some degree of a decrease in euphoria with crushed EMBEDA compared to IR morphine and 31% of subjects did not report a reduction in euphoria. There was similar individual variability in the degree of reduction in euphoria.
A randomized double-blind, placebo-controlled, three-way cross-over trial in 28 non-dependent recreational opioid-users was performed using 30 mg of IV morphine alone and 30 mg of IV morphine in combination with 1.2 mg of IV naltrexone to simulate parenteral use of crushed EMBEDA. The combination of morphine with naltrexone resulted in 71% of subjects reporting a reduction in euphoria compared to morphine alone. Note that the intravenous injection of crushed EMBEDA may result in serious injury and death due to a morphine overdose or an embolic event. Intravenous injection of crushed EMBEDA may preciptitate a severe withdrawal syndrome in opioid-dependent patients.
The clinical significance of the degree of reduction in drug liking and euphoria reported in these studies has not yet been established. There is no evidence that the naltrexone in EMBEDA reduces the abuse liability of EMBEDA.
Absorption
EMBEDA Capsules contain extended-release pellets of morphine sulfate that release morphine slowly compared to an oral morphine solution. Following the administration of oral morphine solution, approximately 50% of the morphine absorbed reaches the systemic circulation within 30 minutes. However, following the administration of an equal amount of EMBEDA to healthy volunteers, this occurs, on average, after 8 hours. As with most forms of oral morphine, because of pre-systemic elimination, only about 20 to 40% of the administered dose reaches the systemic circulation.
EMBEDA is bioequivalent to a similar morphine sulfate extended release capsules product with regard to rate and extent of plasma morphine absorption. The median time to peak plasma morphine levels (Tmax) was shorter for EMBEDA (7.5 hrs) compared to KADIAN® (10 hrs). Dose-related increase in steady-state pre-dose plasma concentrations of morphine were noted following multiple dose administration of EMBEDA in patients.
Food effect: While concurrent administration of high fat food decreases the rate and extent of morphine absorption from EMBEDA, the total bioavailability is not affected. Co-administration of a high-fat meal with EMBEDA did not compromise sequestration of naltrexone.
When taken as directed, the sequestered naltrexone in EMBEDA is not consistently absorbed into systemic circulation following single dose administration. In some subjects, a limited number (~2%) of blood samples had low and highly variable plasma naltrexone levels (median = 7.74 pg/mL, range 4.05-132 pg/mL) following single dose administration of 60 – 120 mg EMBEDA compared to oral naltrexone solution. In patients titrated up to 60 – 80 mg BID EMBEDA, naltrexone levels (4-25.5 pg/mL) were detected in 13 out of 67 patients at steady-state. In a long-term safety study where an average dose of EMBEDA was up to 860 mg administered twice a day for 12 months, 11.0% of blood samples at pre-dose timepoints at steady-state had detectable plasma naltrexone concentrations ranging from 4.03 to 145 pg/mL.
Compared to 2.4 mg naltrexone oral solution, which produced mean (SD) naltrexone plasma levels of 689 (+ 429 pg/mL) and mean (SD) 6β-naltrexol plasma levels of 3920 (+ 1350 pg/mL), administration of intact 60 mg EMBEDA produced no naltrexone plasma levels and mean (SD) 6β-naltrexol plasma levels of 16.7 (+ 13.5 pg/mL). Trough levels of plasma naltrexone and 6-β-naltrexol did not accumulate upon repeated administration of EMBEDA.
Tampering with the EMBEDA formulation by crushing or chewing the pellets, results in the rapid release and absorption of both morphine and naltrexone comparable to an oral solution. This has not been shown to reduce the abuse liability of EMBEDA.
Distribution
Once absorbed, morphine is distributed to skeletal muscle, kidneys, liver, intestinal tract, lungs, spleen, and brain. The volume of distribution of morphine is approximately 3 to 4 L/kg. Morphine is 30 to 35% reversibly bound to plasma proteins. Although the primary site of action of morphine is in the CNS, only small quantities pass the blood brain barrier. Morphine also crosses the placental membranes [see USE IN SPECIFIC POPULATIONS (8.1)] and has been found in breast milk [see USE IN SPECIFIC POPULATIONS (8.3) ].
Metabolism
Major pathways of morphine metabolism include glucuronidation and sulfation in the liver to produce including morphine-3-glucuronide, M3G (about 50%) and morphine-6-glucuronide, M6G (about 5 to 15%) or morphine-3-etheral sulfate. A small fraction (less than 5%) of morphine is demethylated. M3G has no significant contribution to the analgesic activity. Although M6G does not readily cross the blood-brain barrier, it has been shown to have opioid agonist and analgesic activity in humans.
Naltrexone is extensively metabolized into 6-β-naltrexol.
Excretion
Approximately 10% of morphine dose is excreted unchanged in the urine. Elimination of morphine is primarily via hepatic metabolism to glucuronide metabolites M3G and M6G (55 to 65%) which are then renally excreted. A small amount of the glucuronide metabolites is excreted in the bile and there is some minor enterohepatic cycling.
The mean adult plasma clearance is about 20-30 mL/minute/kg. The effective half-life of morphine after IV administration is reported to be approximately 2.0 hours. The terminal elimination half-life of morphine following single dose EMBEDA administration is approximately 29 hours.
Special Populations
Geriatric: Pharmacokinetics of EMBEDA have not been investigated in elderly patients (greater than 65 years) although such patients were included in clinical studies. However, in a long-term open label safety study, the pre-dose plasma morphine concentrations after dose normalization were similar for subjects less than 65 years and those greater than or equal to 65 years of age [see USE IN SPECIFIC POPULATIONS (8.5)].
Pediatric: Pharmacokinetics of EMBEDA were not evaluated in pediatric population.
Gender: No meaningful differences were noted between male and female patients in the analysis of pharmacokinetic data of morphine from clinical studies.
Race: Pharmacokinetic differences due to race may exist. Additionally, Chinese subjects given intravenous morphine in one study had a higher clearance when compared to Caucasian subjects (1852 + 116 mL/min versus 1495 + 80 mL/min).
Hepatic Failure: The pharmacokinetics of morphine was found to be significantly altered in individuals with alcoholic cirrhosis. The clearance was found to decrease with a corresponding increase in half-life. The morphine-3-glucuronide (M3G) and morphine-6-glucuronide (M6G) to morphine plasma AUC ratios also decreased in these patients indicating a decrease in metabolic activity.
Renal Insufficiency: The pharmacokinetics of morphine is altered in renal failure patients. AUC is increased and clearance is decreased. The metabolites, M3G and M6G, accumulate several fold in renal failure patients compared with healthy subjects.
Drug Interaction/Alcohol Interaction: As such additive pharmacodynamic effects may be expected when EMBEDA is used in conjunction with alcohol, other opioids, or illicit drugs that cause central nervous system depression. Additionally, a pharmacokinetic drug interaction is noted with concomitant administration of 40% alcohol and EMBEDA, where an average 2-fold (range 1.4- to 5-fold increase) higher Cmax of morphine was noted compared to EMBEDA consumed with water
Uses
EMBEDA is an extended-release oral formulation of morphine sulfate and naltrexone hydrochloride indicated for the management of moderate to severe pain when a continuous, around-the-clock opioid analgesic is needed for an extended period of time.
EMBEDA is NOT intended for use as a prn analgesic.
EMBEDA is not indicated for acute/postoperative pain or if the pain is mild or not expected to persist for an extended period of time. EMBEDA is only indicated for postoperative use if the patient is already receiving chronic opioid therapy prior to surgery or if the postoperative pain is expected to be moderate to severe and persist for an extended period of time. Physicians should individualize treatment, moving from parenteral to oral analgesics as appropriate
EMBEDA is contraindicated in patients with a known hypersensitivity to morphine, morphine salts, naltrexone, or in any situation where opioids are contraindicated.
EMBEDA is contraindicated in patients with significant respiratory depression in unmonitored settings or the absence of resuscitative equipment.
EMBEDA is contraindicated in patients with acute or severe bronchial asthma or hypercapnia in unmonitored settings or the absence of resuscitative equipment [see WARNINGS AND PRECAUTIONS (5.1) ].
EMBEDA is contraindicated in any patient who has or is suspected of having paralytic ileus.
EMBEDA is to be swallowed whole or the contents of the capsules sprinkled on apple sauce. The pellets in the capsules are not to be crushed, dissolved, or chewed. The resulting morphine dose may be fatal, particularly in opioid-naïve individuals. In opioid-tolerant individuals, the absorption of naltrexone may increase the risk of precipitating withdrawal.
EMBEDA 100 mg/4 mg is for use in opioid-tolerant patients only. Ingestion of these capsules or of the pellets within the capsules may cause fatal respiratory depression when administered to patients not already tolerant to high doses of opioids.
EMBEDA contains morphine, an opioid agonist, and is a Schedule II controlled substance. Opioid agonists have the potential for being abused and are sought by drug abusers and people with addiction disorders and are subject to criminal diversion.
Morphine can be abused in a manner similar to other opioid agonists, legal or illicit. This should be considered when prescribing or dispensing EMBEDA in situations where the physician or pharmacist is concerned about an increased risk of misuse, abuse, or diversion.
Abuse of EMBEDA by crushing, chewing, snorting, or injecting the dissolved product will result in the uncontrolled delivery of the opioid and pose a significant risk to the abuser that could result in overdose and death [see DRUG ABUSE AND DEPENDENCE (9)].
Concerns about abuse and addiction should not prevent the proper management of pain. Healthcare professionals should contact their State Professional Licensing Board or State Controlled Substances Authority for information on how to prevent and detect abuse of this product.
EMBEDA may be expected to have additive effects when used in conjunction with alcohol, other opioids, or illicit drugs that cause central nervous system depression because respiratory depression, hypotension, and profound sedation or coma may result.
Patients should not consume alcoholic beverages, prescription or non-prescription medications containing alcohol while on EMBEDA therapy. The co-ingestion of alcohol with EMBEDA can result in an increase of morphine plasma levels and potentially fatal overdose of morphine [see CLINICAL PHARMACOLOGY (12.3)].
Respiratory depression is the chief hazard of all morphine preparations such as EMBEDA. Respiratory depression occurs more frequently and is more dangerous in elderly and debilitated patients, and those suffering from conditions accompanied by hypoxia, hypercapnia, or upper airway obstruction (when even moderate therapeutic doses may significantly decrease pulmonary ventilation).
EMBEDA should be used with extreme caution in patients with chronic obstructive pulmonary disease or cor pulmonale, and in patients having a substantially decreased respiratory reserve (e.g., severe kyphoscoliosis), hypoxia, hypercapnia, or pre-existing respiratory depression. In such patients, even usual therapeutic doses of morphine may increase airway resistance and decrease respiratory drive to the point of apnea. In these patients, alternative non-opioid analgesics should be considered, and opioids should be employed only under careful medical supervision at the lowest effective dose.
The respiratory depressant effects of morphine with carbon dioxide retention and secondary elevation of cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions, or a pre-existing increase in intracranial pressure. EMBEDA can produce effects on pupillary response and consciousness, which may obscure neurologic signs of further increases in pressure in patients with head injuries. EMBEDA should only be administered under such circumstances when considered essential and then with extreme care.
EMBEDA may cause severe hypotension. There is an added risk to individuals whose ability to maintain blood pressure has already been compromised by a reduced blood volume or a concurrent administration of drugs such as phenothiazines or general anesthetics [see DRUG INTERACTIONS (7.1)]. EMBEDA may produce orthostatic hypotension and syncope in ambulatory patients.
EMBEDA should be administered with caution to patients in circulatory shock, as vasodilation produced by the drug may further reduce cardiac output and blood pressure.
EMBEDA should be used with caution and in reduced dosage in patients who are concurrently receiving other central nervous system depressants including sedatives or hypnotics, general anesthetics, phenothiazines, other tranquilizers, and alcohol because respiratory depression, hypotension, and profound sedation or coma may result [see DRUG INTERACTIONS (7.1) ].
EMBEDA should not be given to patients with gastrointestinal obstruction, particularly paralytic ileus, as there is a risk of the product remaining in the stomach for an extended period and the subsequent release of a bolus of morphine when normal gut motility is restored. As with other solid morphine formulations diarrhea may reduce morphine absorption.
The administration of morphine may obscure the diagnosis or clinical course in patients with acute abdominal condition.
Patients taking EMBEDA who are scheduled for cordotomy or other interruption of pain transmission pathways should have EMBEDA ceased 24 hours prior to the procedure and the pain controlled by parenteral short-acting opioids. In addition, the post-procedure titration of analgesics for such patients should be individualized to avoid either oversedation or withdrawal syndromes.
EMBEDA may cause spasm of the sphincter of Oddi and should be used with caution in patients with biliary tract disease, including acute pancreatitis. Opioids may cause increases in the serum amylase level.
Tolerance is the need for increasing doses of opioids to maintain a defined effect such as analgesia (in the absence of disease progression or other external factors). Physical dependence is manifested by withdrawal symptoms after abrupt discontinuation of a drug or upon administration of an antagonist. Physical dependence and tolerance are common during chronic opioid therapy.
The opioid abstinence or withdrawal syndrome is characterized by some or all of the following: restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other symptoms also may develop, including: irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate.
EMBEDA should not be abruptly discontinued [see DOSAGE AND ADMINISTRATION (2.5)].
EMBEDA should be administered with caution, and in reduced dosages in elderly or debilitated patients; patients with severe renal or hepatic insufficiency; patients with Addison's disease; myxedema; hypothyroidism; prostatic hypertrophy or urethral stricture.
Caution should also be exercised in the administration of EMBEDA to patients with CNS depression, toxic psychosis, acute alcoholism, and delirium tremens.
All opioids may aggravate convulsions in patients with convulsive disorders, and all opioids may induce or aggravate seizures in some clinical settings.
EMBEDA may impair the mental and/or physical abilities needed to perform potentially hazardous activities such as driving a car or operating machinery. Patients must be cautioned accordingly. Patients should also be warned about the potential combined effects of EMBEDA with other CNS depressants, including other opioids, phenothiazines, sedative/hypnotics, and alcohol [see DRUG INTERACTIONS (7.1)].
Although extremely rare, cases of anaphylaxis have been reported with the use of a similar extended release morphine formulation.
Agonist/antagonist analgesics (i.e., pentazocine, nalbuphine, butorphanol) should be administered with caution to a patient who has received or is receiving a course of therapy with EMBEDA. In this situation, mixed agonist/antagonist analgesics may reduce the analgesic effect of EMBEDA and/or may precipitate withdrawal symptoms in these patients.
Consuming EMBEDA that have been tampered by crushing, chewing, or dissolving the extended-release formulation can release sufficient naltrexone to precipitate withdrawal in opioid-dependent individuals. Symptoms of withdrawal usually appear within five minutes of ingestion of naltrexone and can last for up to 48 hours. Mental status changes can include confusion, somnolence, and visual hallucinations. Significant fluid losses from vomiting and diarrhea can require intravenous fluid administration. Patients should be closely monitored and therapy with non-opioid medications tailored to meet individual requirements.
Naltrexone does not interfere with thin-layer, gas-liquid, and high pressure liquid chromatographic methods which may be used for the separation and detection of morphine, methadone, or quinine in the urine. Naltrexone may or may not interfere with enzymatic methods for the detection of opioids depending on the specificity of the test. Please consult the test manufacturer for specific details.
Serious adverse reactions that may be associated with EMBEDA therapy in clinical use include: respiratory depression, respiratory arrest, apnea, circulatory depression, cardiac arrest, hypotension, and/or shock [see OVERDOSAGE (10), WARNINGS AND PRECAUTIONS (5)].
The common adverse events seen on initiation of therapy with EMBEDA are dose dependent, and their frequency depends on the clinical setting, the patient's level of opioid tolerance, and host factors specific to the individual. They should be expected and managed as a part of opioid analgesia. The most frequent of these include drowsiness, dizziness, constipation, and nausea.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
There were 1251 subjects exposed to at least one dose of EMBEDA in the clinical program. During late phase clinical development, 618 subjects received EMBEDA in two randomized, controlled, double-blind studies in subjects with osteoarthritis of the hip or knee. An additional 465 subjects received EMBEDA in an open-label, year-long safety study of subjects with chronic, non-cancer pain, 208 subjects for at least six months and 124 for 12 months. The remaining 168 subjects were exposed to a single dose of EMBEDA in early PK/PD studies.
Short-Term (12-Week) Randomized Study
Adverse reactions observed in at least 2% of subjects treated with EMBEDA
This study utilized an enriched enrollment with a randomized withdrawal design in which subjects were titrated to effect on open-label EMBEDA for up to 45 days. Once their pain was controlled, subjects were randomized to either active treatment with EMBEDA or were tapered off EMBEDA using a double-dummy design and placed on placebo. The Maintenance Period was 12 weeks. The most common adverse reactions leading to study discontinuation were nausea, constipation, vomiting, fatigue, dizziness, pruritus, and somnolence. Adverse reactions, defined as treatment-related adverse events assessed by the investigators, reported by greater than or equal to 2.0% of subjects in either the titration or maintenance phase of the 12-week study are presented in Table 1.
Long-Term Open-Label Safety Study
In the long-term open-label safety study, 465 patients with chronic non-malignant pain were enrolled and 124 patients were treated for up to 1 year. The distributions of adverse events were similar to that of the randomized, controlled studies, and were consistent with the most common opioid related adverse events. Adverse reactions, defined as treatment-related adverse events assessed by the investigators, reported by greater than or equal to 2.0% of subjects are presented in Table 2.
Adverse Reactions Observed in the Phase 2/3 Studies
Most common (greater than 10%): constipation, nausea, somnolence
Common (greater than or equal to 1% to less than 10%): vomiting, headache, dizziness, pruritus, dry mouth, diarrhea, fatigue, insomnia, hyperhidrosis, anxiety, chills, abdominal pain, lethargy, edema peripheral, dyspepsia, anorexia, muscle spasms, depression, flatulence, restlessness, decreased appetite, irritability, stomach discomfort, tremor, arthralgia, hot flush, sedation
Adverse Reactions Observed in the Phase 2/3 Studies
Most common (greater than or equal to 10%):
Gastrointestinal disorders: constipation, nausea;
Nervous system disorders: somnolence.
Common (greater than or equal to 1% to less than 10%):
Gastrointestinal disorders: abdominal pain, diarrhea, dry mouth, dyspepsia, flatulence, stomach discomfort, vomiting;
General disorders and administration site conditions: chills, edema peripheral, fatigue, irritability;
Metabolism and nutrition disorders: anorexia, decreased appetite;
Musculoskeletal and connective tissue disorders: arthralgia, muscle spasms;
Nervous system disorders: dizziness, headache, lethargy, sedation, tremor;
Psychiatric disorders: anxiety, depression, insomnia, restlessness;
Skin and subcutaneous tissue disorders: hyperhidrosis, pruritus;
Vascular disorders: hot flush.
Less Common (less than 1%):
Eye disorders: vision blurred, orthostatic hypotension;
Gastrointestinal disorders: abdominal distension, pancreatitis, abdominal discomfort, fecaloma, abdominal pain lower, abdominal tenderness;
General disorders and administration site conditions: malaise, asthenia, feeling jittery, drug withdrawal syndrome;
Hepatobiliary disorders: cholecystitis;
Investigations: alanine aminotransferase increased, aspartate aminotransferase increased;
Musculoskeletal and connective tissue disorders: myalgia, muscular weakness;
Nervous system disorders: depressed level of consciousness, mental impairment, memory impairment, disturbance in attention, stupor, paraesthesia, coordination abnormal;
Psychiatric disorders: disorientation, thinking abnormal, mental status changes, confusional state, euphoric mood, hallucination, abnormal dreams, mood swings, nervousness;
Renal and urinary disorders: urinary retention, dysuria;
Reproductive system and breast disorders: erectile dysfunction;
Respiratory, thoracic and mediastinal disorders: dyspnea, rhinorrhoea;
Skin and subcutaneous tissue disorders: rash, piloerection, cold sweat, night sweats;
Vascular disorders: hypotension, flushing.
Acute overdosage with morphine is manifested by respiratory depression, somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, and, sometimes, pulmonary edema, bradycardia, hypotension, and death. Marked mydriasis rather than miosis may be seen due to severe hypoxia in overdose situations.
Primary attention should be given to the re-establishment of a patent and protected airway and institution of assisted or controlled ventilation if needed. Other supportive measures (including oxygen, vasopressors) should be employed in the management of circulatory shock and pulmonary edema accompanying overdose as indicated. Cardiac arrest or arrhythmias will require advanced life support techniques.
The pure opioid antagonists, naloxone or nalmefene, are specific antidotes to respiratory depression which results from opioid overdose. Since the duration of reversal would be expected to be less than the duration of action of morphine in EMBEDA, the patient must be carefully monitored until spontaneous respiration is reliably re-established. EMBEDA will continue to release and add to the morphine load for up to 24 hours after administration and the management of an overdose should be monitored accordingly. If the response to opioid antagonists is suboptimal or not sustained, additional antagonist should be given as directed by the manufacturer of the product.
Opioid antagonists should not be administered in the absence of clinically significant respiratory or circulatory depression secondary to morphine overdose. Such agents should be administered cautiously to persons who are known, or suspected to be physically dependent on EMBEDA. In such cases, an abrupt or complete reversal of opioid effects may precipitate an acute withdrawal syndrome.
The sequestered naltrexone in EMBEDA has no role in the treatment of opioid overdose.
In an individual physically dependent on opioids, administration of an opioid receptor antagonist may precipitate an acute withdrawal. The severity of the withdrawal produced will depend on the degree of physical dependence and the dose of the antagonist administered. Use of an opioid antagonist should be reserved for cases where such treatment is clearly needed. If it is necessary to treat serious respiratory depression in the physically dependent patient, administration of the antagonist should be begun with care and by titration with smaller than usual doses of the antagonist
Selection of patients for treatment with morphine sulfate should be governed by the same principles that apply to the use of similar opioid analgesics. Physicians should individualize treatment in every case, using non-opioid analgesics, opioids on an as needed basis and/or combination products, and chronic opioid therapy in a progressive plan of pain management such as outlined by the World Health Organization and Federation of State Medical Boards Model Guidelines.
Care should be taken to use low initial doses of EMBEDA in patients who are not already opioid-tolerant, especially those who are receiving concurrent treatment with muscle relaxants, sedatives, or other CNS active medications.
The 100 mg/4 mg capsules are for use only in opioid-tolerant patients.
EMBEDA is to be swallowed whole or the contents of the capsules sprinkled on apple sauce and taken by mouth. The pellets in the capsules are not to be crushed, dissolved, or chewed before swallowing.
It is critical to adjust the dosing regimen for each patient individually, taking into account the patient's prior analgesic treatment experience. In the selection of the initial dose of EMBEDA, attention should be given to:
1) the total daily dose, potency, and kind of opioid the patient has been
taking previously;
2) the reliability of the relative potency estimate used
to calculate the equivalent dose of morphine needed (Note: potency estimates may
vary with the route of administration);
3) the patient's degree of opioid
experience and opioid tolerance;
4) the general condition and medical status
of the patient;
5) concurrent medication;
6) the type and severity of the
patient's pain.
The following dosing recommendations can be considered approaches to what is actually a series of clinical decisions over time in the management of the pain of an individual patient:
Use of EMBEDA as the First Opioid Analgesic
The lowest dose of EMBEDA should be used as the initial opioid analgesic in patients with chronic pain. Patients may subsequently be titrated to a once or twice a day dosage which adequately manages their pain.
Conversion from Other Oral Morphine Formulations to EMBEDA
Patients on other oral morphine formulations may be converted to EMBEDA by administering one-half of the patient's total daily oral morphine dose as EMBEDA every 12 hours (twice-a-day) or by administering the total daily oral morphine dose as EMBEDA every 24 hours (once-a-day). EMBEDA should not be given more frequently than every 12 hours.
Conversion from Oral Opioids, Parenteral Morphine, or Other Parenteral Opioids to EMBEDA
EMBEDA can be administered to patients previously receiving treatment with parenteral morphine or other opioids. While there are useful tables of oral and parenteral equivalents in cancer analgesia, there is substantial inter-patient variation in the relative potency of different opioid drugs and formulations. For these reasons, it is better to underestimate the patient's 24-hour oral morphine requirement and provide rescue medication than to overestimate and manage an adverse event. The following general points should be considered:
- Parenteral to Oral Morphine Ratio: It may take anywhere from 2-6 mg of oral morphine to provide analgesia equivalent to 1 mg of parenteral morphine. A dose of oral morphine three times the daily parenteral morphine requirement may be sufficient in chronic use settings.
- Other Oral or Parenteral Opioids to Oral Morphine Sulfate: There is lack of systematic evidence bearing on these types of analgesic substitutions. Therefore, specific recommendations are not possible. Physicians are advised to refer to published relative potency data, keeping in mind that such ratios are only approximate. In general, it is safest to give half of the estimated daily morphine demand as the initial dose and to manage inadequate analgesia by supplementation with immediate-release morphine.
The first dose of EMBEDA may be taken with the last dose of any immediate-release (short-acting) opioid medication due to the extended-release characteristics of EMBEDA.
Patients may develop some degree of tolerance, requiring dosage adjustment until they have achieved their individual balance between effective analgesia and opioid side effects such as confusion, sedation, and constipation.
- EMBEDA should be titrated no more frequently than every-other-day to allow patients to stabilize before escalating the dose.
- If breakthrough pain occurs, the dose may be supplemented with a small dose (less than 20% of the total daily dose) of a short-acting analgesic.
- Patients who exhibit signs of excessive opioid side effects such as sedation should have their dose reduced.
- Patients who experience inadequate analgesia on once-daily dosing should be switched to twice-a-day.
- EMBEDA should not be dosed more frequently than every 12 hours.
During periods of changing analgesic requirements including initial titration, frequent communication is recommended between physician, other members of the healthcare team, the patient, and the caregiver/family.
Patients who have difficulty swallowing whole capsules or tablets may benefit from an alternative method of administration. EMBEDA pellets may be sprinkled over apple sauce. Other foods have not been tested and should not be substituted for apple sauce.
- Sprinkle the pellets onto a small amount of apple sauce and use immediately.
- The patient must be cautioned not to chew the pellets [see WARNINGS AND PRECAUTIONS (5)].
- Rinse mouth to ensure all pellets have been swallowed.
- Patients should consume entire portion and should not divide apple sauce into separate doses.
Do not administer EMBEDA pellets through a nasogastric or gastric tubes.
Continual re-evaluation of the patient receiving morphine sulfate is important, with special attention to the maintenance of pain control and the relative incidence of side effects associated with therapy. If the level of pain increases, effort should be made to identify the source of increased pain, while adjusting the dose as described above to decrease the level of pain. During chronic therapy, especially for non-cancer-related pain (or pain associated with other terminal illnesses), the continued need for the use of opioid analgesics should be re-assessed as appropriate.
In general, EMBEDA should not be abruptly discontinued. However, EMBEDA, like other opioids, can be safely discontinued without the development of withdrawal symptoms by slowly tapering the daily dose.
HOW SUPPLIED/STORAGE AND HANDLING
|
|
EMBEDA 20 mg/0.8 mg | EMBEDA 30 mg/1.2 mg | EMBEDA 50 mg/2 mg | EMBEDA 60 mg/2.4 mg | EMBEDA 80 mg/3.2 mg | EMBEDA 100 mg/4 mg |
| Morphine sulfate | 20 mg | 30 mg | 50 mg | 60 mg | 80 mg | 100 mg |
| Sequestered naltrexone hydrochloride |
0.8 mg |
1.2 mg | 2 mg | 2.4 mg | 3.2 mg | 4 mg |
| Capsule Description | size 4, two-toned, yellow opaque hard gelatin capsule. The darker-toned cap has “EMBEDA” printed in grey ink and a single grey band around ¾ of the circumference. The lighter-toned body has “20” reverse-printed in a grey circle. | size 3, two-toned, blue violet opaque hard gelatin capsule. The darker-toned cap has “EMBEDA” printed in grey ink and a single grey band around ¾ of the circumference. The lighter-toned body has “30” reverse-printed in a grey circle. | size 1, two-toned, blue opaque hard gelatin capsule. The darker-toned cap has “EMBEDA” printed in grey ink and a single grey band around ¾ of the circumference. The lighter-toned body has “50” reverse-printed in a grey circle. | size 0, two-toned, pink opaque hard gelatin capsule. The darker-toned cap has “EMBEDA” printed in grey ink and a single grey band around ¾ of the circumference. The lighter-toned body has “60” reverse-printed in a grey circle. | size 0, two-toned, light peach opaque elongated hard gelatin capsule. The darker-toned cap has “EMBEDA” printed in grey ink and a single grey band around ¾ of the circumference. The lighter-toned body has “80” reverse-printed in a grey circle. | size 00, two-toned, green opaque hard gelatin capsule. The darker-toned cap has “EMBEDA” printed in grey ink and a single grey band around ¾ of the circumference. The lighter-toned body has “100” reverse-printed in a grey circle. |
| Bottle Size | 100 | 100 | 100 | 100 | 100 | 100 |
| NDC # | 60793-430-01 | 60793-431-01 | 60793-433-01 | 60793-434-01 | 60793-435-01 | 60793-437-01 |
Store at 25°C (77°F); excursions permitted between 15° and 30°C (59° and 86°F).
Dispense in a sealed, tamper-evident, childproof, light-resistant container.
Patients receiving EMBEDA should be given the following instructions by the physician:
- Patients should be advised that EMBEDA contains morphine and naltrexone and should be taken only as directed.
- The dose of morphine sulfate should not be adjusted without consulting with a physician or other healthcare professional. EMBEDA should be swallowed whole (not crushed, dissolved, or chewed) due to a risk of fatal morphine overdose or naltrexone precipitated withdrawal symptoms. Alternately, EMBEDA Capsules may be opened and the entire contents sprinkled on a small amount of apple sauce immediately prior to ingestion [see DOSAGE AND ADMINISTRATION (2.3)].
- Patients should not consume alcoholic beverages while on EMBEDA therapy. Additionally, patients must not use prescription or non-prescription medications containing alcohol while on EMBEDA therapy. The co-ingestion of alcohol with EMBEDA may result in an increase of plasma levels and potentially fatal overdose of morphine.
- Patients should be advised of the most common adverse reactions that may occur while taking EMBEDA: constipation, nausea, somnolence, vomiting, dizziness, pruritus, and headache.
- Patients should be advised that EMBEDA may cause drowsiness, dizziness, or lightheadedness and may impair mental and/or physical ability required for the performance of potentially hazardous tasks (e.g., driving, operating machinery). Patients started on EMBEDA or patients whose dose has been adjusted should refrain from any potentially dangerous activity until it is established that they are not adversely affected.
- Patients should not combine EMBEDA with central nervous system depressants (sleep aids, tranquilizers) except by the orders of the prescribing physician because dangerous additive effects may occur resulting in serious injury or death.
- Patients should be advised that EMBEDA is a potential drug of abuse. They should protect it from theft.
- Special care must be taken to avoid accidental ingestion or use by individuals (including children) other than the patient for whom it was originally prescribed, as such unsupervised use may have severe, even fatal, consequences [see WARNINGS AND PRECAUTIONS (5)].
- Patients should be advised that EMBEDA 100 mg/4 mg is for use only in opioid-tolerant patients.
- Women of childbearing potential who become or are planning to become pregnant should consult a physician prior to initiating or continuing therapy with EMBEDA.
- Safe use in pregnancy has not been established. Prolonged use of opioid analgesics during pregnancy may cause fetal neonatal physical dependence, and neonatal withdrawal may occur.
- As with other opioids, patients taking EMBEDA should be advised of the potential for severe constipation; appropriate laxatives and/or stool softeners as well as other appropriate treatments should be initiated from the onset of opioid therapy.
- Patients should be advised to seek medical attention immediately if signs of a serious allergic reaction occur, such as swelling of the face, throat, or tongue, trouble breathing, feeling dizzy or faint, pounding heart beat, chest pain or feeling of doom [see WARNINGS AND PRECAUTIONS (5.13) ].
Patients should be advised to report episodes of breakthrough pain and adverse experiences occurring during therapy. Individualization of dosage is essential to make optimal use of this medication.
Patients should be advised that EMBEDA may impair mental and/or physical ability required for the performance of potentially hazardous tasks (e.g., driving, operating machinery). Patients started on EMBEDA or whose dose has been changed should refrain from dangerous activity until it is established that they are not adversely affected [see WARNINGS AND PRECAUTIONS (5.12)].
Patients should be advised that EMBEDA should not be taken with alcohol, prescription or non-prescription medications containing alcohol, or other CNS depressants (sleeping medication, tranquilizers) except by the orders of the prescribing healthcare provider because dangerous additive effects may occur resulting in serious injury or death [see WARNINGS AND PRECAUTIONS (5.2), (5.6)].
Women of childbearing potential who become or are planning to become pregnant, should consult their prescribing healthcare provider prior to initiating or continuing therapy with EMBEDA [see USE IN SPECIFIC POPULATIONS (8.1)].
Patients should be advised that if they have been receiving treatment with EMBEDA for more than a few weeks and cessation of therapy is indicated, it may be appropriate to taper the EMBEDA dose, rather than abruptly discontinue it, due to the risk of precipitating withdrawal symptoms. Their prescribing healthcare provider should provide a dose schedule to accomplish a gradual discontinuation of the medication.
Patients should be advised that EMBEDA is a potential drug of abuse. They should protect it from theft, and it should never be given to anyone other than the individual for whom it was prescribed [see WARNINGS AND PRECAUTIONS (5.1)].
Patients should be advised that severe constipation could occur as a result of taking EMBEDA and appropriate laxatives, stool softeners and other appropriate treatments should be initiated from the beginning of opioid therapy.
Patients should be instructed to keep EMBEDA in a secure place out of the reach of children. When EMBEDA is no longer needed, the unused capsules should be destroyed by flushing down the toilet.
[See separate leaflet.]
Manufactured for: King Pharmaceuticals, Inc., 501 Fifth Street, Bristol, TN 37620
(Telephone: 1-800-776-3637)
by: Actavis Elizabeth LLC, 200 Elmora Avenue, Elizabeth, NJ 07207 USA
EMBEDA is a trademark of Alpharma Pharmaceuticals LLC, a wholly owned subsidiary of King Pharmaceuticals, Inc.
KADIAN is a registered trademark of Actavis Elizabeth LLC
To report SUSPECTED ADVERSE REACTIONS, contact King Pharmaceuticals, Inc. at 1-800-546-4905 or DSP@Kingpharm.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
U.S. Patent Numbers:
5,202,128
5,378,474
5,330,766
40-9096
Revised – June 2009
v.10
EMBEDA™ capsules contain morphine, an opioid agonist and a Schedule II controlled substance with an abuse liability similar to other opioid agonists. EMBEDA can be abused in a manner similar to other opioid agonists, legal or illicit. This should be considered when prescribing or dispensing EMBEDA in situations where the physician or pharmacist is concerned about an increased risk of misuse, abuse, or diversion.
EMBEDA contains pellets of an extended-release oral formulation of morphine sulfate, an opioid receptor agonist, surrounding an inner core of naltrexone hydrochloride, an opioid receptor antagonist indicated for the management of moderate to severe pain when a continuous, around-the-clock opioid analgesic is needed for an extended period of time.
EMBEDA is NOT intended for use as a prn analgesic.
EMBEDA 100 mg/4 mg IS FOR USE IN OPIOID-TOLERANT PATIENTS ONLY. Ingestion of these capsules or the pellets within the capsules may cause fatal respiratory depression when administered to patients not already tolerant to high doses of opioids.
Patients should not consume alcoholic beverages while on EMBEDA therapy. Additionally, patients must not use prescription or non-prescription medications containing alcohol while on EMBEDA therapy. The co-ingestion of alcohol with EMBEDA may result in an increase of plasma levels and potentially fatal overdose of morphine. EMBEDA is to be swallowed whole or the contents of the capsules sprinkled on apple sauce. The pellets in the capsules are not to be crushed, dissolved, or chewed due to the risk of rapid release and absorption of a potentially fatal dose of morphine.
Crushing, chewing, or dissolving EMBEDA will also result in the release of naltrexone which may precipitate withdrawal in opioid-tolerant individuals.
[See separate leaflet.]
Manufactured for: King Pharmaceuticals, Inc., 501 Fifth Street, Bristol, TN 37620
(Telephone: 1-800-776-3637)
by: Actavis Elizabeth LLC, 200 Elmora Avenue, Elizabeth, NJ 07207 USA
EMBEDA is a trademark of Alpharma Pharmaceuticals LLC, a wholly owned subsidiary of King Pharmaceuticals, Inc.
KADIAN is a registered trademark of Actavis Elizabeth LLC
To report SUSPECTED ADVERSE REACTIONS, contact King Pharmaceuticals, Inc. at 1-800-546-4905 or DSP@Kingpharm.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
U.S. Patent Numbers:
5,202,128
5,378,474
5,330,766
40-9096
Revised – June 2009
EMBEDA™ (im-bed-a)
(morphine sulfate and
naltrexone hydrochloride)
Extended Release Capsules CII
IMPORTANT: Keep EMBEDA in a safe place away from children. Accidental use by a child is a medical emergency and can result in death. If a child accidentally takes EMBEDA, get emergency help right away.
Read the Medication Guide that comes with EMBEDA before you start taking it and each time you get a new prescription. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. Share this important information with members of your household.
What Is the Most Important Information I Should Know About EMBEDA?
-
Do not crush, dissolve, or chew EMBEDA capsules or the
capsule contents before swallowing. If EMBEDA is taken in this way, both the
morphine and naltrexone in EMBEDA will be released too fast.
This is dangerous. It may cause you to have trouble breathing, and
lead to death.
- If your body is not used to taking opioids and your body absorbs too much morphine, you could overdose and die.
- If you have been taking opioids (narcotics) for a period of time, and your body absorbs the naltrexone in EMBEDA, this could cause you to have uncomfortable withdrawal symptoms.
- Take EMBEDA exactly as prescribed by your healthcare provider.
- EMBEDA is not for use to treat pain that you only have once in a while (“as needed”).
- If you cannot swallow capsules, tell your healthcare provider. There may be another way to take EMBEDA that may be right for you. See “How should I take EMBEDA?”
- Do not take the highest dose of EMBEDA (morphine sulfate 100 mg and naltrexone hydrochloride 4 mg) unless you are “opioid tolerant.” Opioid tolerant means that you regularly use another opioid medicine for your constant (around the clock) pain and your body is used to it.
- Do not drink alcohol, or use prescription or non-prescription medicines that contain alcohol while you are being treated with EMBEDA. Alcohol can cause very high levels of morphine in your blood and you can die due to an overdose of morphine.
- Prevent theft, misuse or abuse. Keep EMBEDA in a safe place to protect it from being stolen. EMBEDA can be a target for people who misuse or abuse prescription medicines or street drugs.
- Never give EMBEDA to anyone else, even if they have the same symptoms you have. It may harm them or even cause death.
See the section “What are the possible side effects of EMBEDA?” for more information about side effects.
What is EMBEDA?
- EMBEDA is a prescription medicine that contains morphine sulfate, an opioid receptor agonist (narcotic pain medicine) and naltrexone hydrochloride, an opioid receptor antagonist. Naltrexone hydrochloride is in the middle of each pellet and has a special coating to protect it from being released. If you crush or chew EMBEDA, the naltrexone will be released all at one time. See “What is the most important information I should know about EMBEDA?
- EMBEDA is a federally controlled substance (CII) because it is a strong opioid pain medicine that can be abused by people who abuse prescription medicines or street drugs.
- EMBEDA is used to manage moderate to severe pain that continues around-the-clock and is expected to last for a long period of time.
- It is not known if EMBEDA is safe and works in children under the age of 18.
Who Should Not Take EMBEDA?
Do not take EMBEDA if you:
- are having an asthma attack or have severe asthma, trouble breathing, or lung problems.
- have a bowel blockage called paralytic ileus.
- are allergic to morphine, morphine salts, naltrexone, or any of the ingredients in EMBEDA. See the end of this Medication Guide for a complete list of ingredients in EMBEDA.
What Should I Tell My Healthcare Provider Before Starting EMBEDA?
-
EMBEDA may not be right for you. Tell your healthcare
provider about all of your medical conditions, especially if you:
- have trouble breathing or lung problems
- have a head injury or brain problem
- have liver or kidney problems
- have adrenal gland problems, such as Addison’s disease
- have convulsions or seizures
- have thyroid problems
- have problems urinating or prostate problems
- have constipation or other bowel problems
- have problems with your pancreas or gallbladder
- have severe scoliosis
- have a drinking problem or alcoholism
- have severe mental problems or hallucinations (see or hear things that are not really there)
- have or have had drug abuse or drug addiction problems
- are planning to have surgery (cordotomy) or another procedure that will interrupt the pain signals to your body.
- are pregnant or plan to become pregnant. EMBEDA may harm your unborn baby.
- are breastfeeding. EMBEDA may pass through your milk and may harm your baby. You should not breastfeed while taking EMBEDA.
- Tell your healthcare provider about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal supplements. Some medicines may cause serious problems when taken with EMBEDA. Sometimes, the doses of certain medicines and EMBEDA may need to be changed if used together.
- Be especially careful about taking other medicines that make you sleepy such
as:
- other pain medicines
- anti-depressant medicines
- sleeping pills
- anti-anxiety medicines
- muscle relaxants
- antihistamines
- anti-nausea medicines
- tranquilizers
Also tell your healthcare provider if you take:
- cimetidine (Tagamet)
- a water pill (diuretic)
- an anticholinergic medicine
- Do not take EMBEDA if you already take a monoamine oxidase inhibitor medicine (MAOI) or within 14 days after you stop taking an MAOI medicine.
- Do not take any new medicine while using EMBEDA until you have talked to your healthcare provider or pharmacist. They will tell you if it is safe to take other medicines with EMBEDA.
Ask your healthcare provider if you are not sure if your medicine is one listed above.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How Should I Take EMBEDA?
- Take EMBEDA exactly as prescribed by your healthcare provider. Do not change your dose unless your healthcare provider tells you to.
- You can take EMBEDA with or without food.
- Swallow EMBEDA capsule whole. Do not crush, dissolve, or chew EMBEDA or the pellets in the capsules before swallowing. See “What is the most important information I should know about EMBEDA?”
- If you cannot swallow capsules, tell your healthcare provider. There may be another way to take EMBEDA that may be right for you. If your doctor tells you that you can take EMBEDA using this other way, follow these steps:
EMBEDA can be opened and the pellets inside the capsule can be sprinkled over apple sauce, as follows:
- Open the EMBEDA capsule and sprinkle the pellets over approximately one
tablespoon of apple sauce (Figure 1).
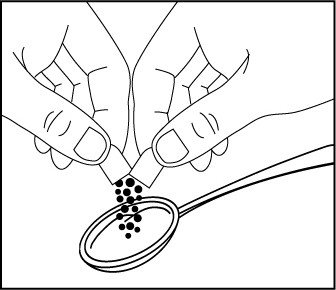 Figure 1 Swallow all of the apple sauce and pellets right away. Do not save any of
the apple sauce and pellets for another dose (Figure
2).
Figure 1 Swallow all of the apple sauce and pellets right away. Do not save any of
the apple sauce and pellets for another dose (Figure
2).
 Figure 2 Rinse your mouth to make sure you have swallowed all of the pellets. Do not
chew the pellets (Figure 3).
Figure 2 Rinse your mouth to make sure you have swallowed all of the pellets. Do not
chew the pellets (Figure 3).
 Figure 3 Flush the empty capsule down the toilet right away (Figure 4).
Figure 3 Flush the empty capsule down the toilet right away (Figure 4).
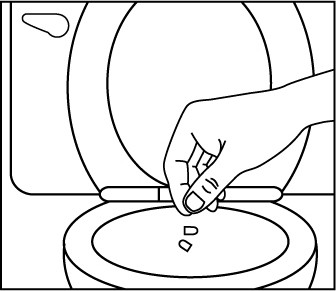 Figure 4 You should not receive EMBEDA through a nasogastric tube or gastric tube
(stomach tube).
Figure 4 You should not receive EMBEDA through a nasogastric tube or gastric tube
(stomach tube).
- If you miss a dose, take it as soon as possible. If it is almost time for your next dose, skip the missed dose. Just take the next dose at your regular time. Do not take 2 doses at the same time unless your healthcare provider tells you to. If you are not sure about your dosing, call your healthcare provider.
- If you take too much EMBEDA or overdose, call 911 or poison control center right away.
- Call your healthcare provider if the dose of EMBEDA that you are taking does not relieve your pain.
What Should I Avoid While Taking EMBEDA?
- Do not drive, operate heavy machinery, or do other dangerous activities, especially when you start taking EMBEDA and when your dose is changed, until you know how you react to this medicine. EMBEDA can make you sleepy, and also cause you to feel dizzy and lightheaded. Ask your healthcare provider to tell you when it is okay to do these activities.
What are the Possible Side Effects of EMBEDA?
EMBEDA can cause serious side effects, including:
- See “What is the most important information I should know about EMBEDA?”
-
EMBEDA can cause serious breathing problems that can become
life-threatening, especially if used the wrong way.
Call your healthcare provider or get medical help right away if:
- your breathing slows down
- you have shallow breathing (little chest movement with breathing)
- you feel faint, dizzy, confused, or
- have any other unusual symptoms
These can be symptoms that you have taken too much EMBEDA (overdose) or the dose is too high for you. These symptoms may lead to serious problems or death if not treated right away.
- EMBEDA can cause your blood pressure to drop. This can make you feel dizzy and faint if you get up too fast from sitting or lying down. Low blood pressure is also more likely to happen if you take other medicines that can also lower your blood pressure. Severe low blood pressure can happen if you lose blood or take certain other medicines.
- EMBEDA can cause physical dependence. Do not stop taking EMBEDA or any other opioid without talking to your healthcare provider. You could become sick with uncomfortable withdrawal symptoms because your body has become used to these medicines. Physical dependence is not the same as drug addiction.
- There is a chance of abuse or addiction with EMBEDA. The chance is higher if you are or have been addicted to or abused other medicines, street drugs, or alcohol, or if you have a history of mental problems.
-
Serious allergic reactions. Rarely, severe allergic
reactions happen in people who take a long-acting morphine medicine that is like
EMBEDA. Get medical help right away if you have any of these symptoms of a
severe allergic reaction:
- feel dizzy or faint
- trouble breathing
- pounding heart beat
- chest pain
- swelling of the face, throat, or tongue
- feeling of doom
The most common side effects of EMBEDA are
- constipation
- nausea
- sleepiness
- vomiting
- dizziness
- itching
- headache
These side effects may decrease with continued use. Talk to your healthcare provider if you continue to have these side effects. These are not all the possible side effects of EMBEDA. For a complete list, ask your healthcare provider or pharmacist.
Constipation (not often enough or hard bowel movements) is a common side effect of pain medicines (opioids) including EMBEDA and is unlikely to go away without treatment. Talk to your healthcare provider about dietary changes, and the use of laxatives (medicines to treat constipation) and stool softeners to prevent or treat constipation while taking EMBEDA.
Talk to your healthcare provider if you have any side effects that bother you or do not go away.
These are not all the possible side effects of EMBEDA. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
- Selling or giving away this medicine is against the law
How should I store EMBEDA?
- See “What is the most important information I should know about EMBEDA?"
- Keep EMBEDA out of the reach of children.
- Keep EMBEDA in the container it comes in.
- Keep EMBEDA at room temperature between 59° to 86°F (15° to 30°C).
- After you stop taking EMBEDA, flush the unused capsules down the toilet.
General Information about EMBEDA
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use EMBEDA for conditions for which it was not prescribed. Do not give EMBEDA to other people, even if they have the same symptoms you have. It may harm them and even cause death. Sharing EMBEDA is against the law.
This medication guide summarizes the most important information about EMBEDA. If you would like more information, talk with your healthcare provider. Also, you can ask your pharmacist or healthcare provider for information about EMBEDA that is written for healthcare professionals. For more information call 1-800-776-3637 or go to www.kingpharm.com.
What are the ingredients in EMBEDA?
Active Ingredients: pellets of morphine sulfate and naltrexone hydrochloride
Inactive Ingredients common to all strengths: talc, ammonio methacrylate copolymer, sugar spheres, ethylcellulose, sodium chloride, polyethylene glycol, hydroxypropyl cellulose, dibutyl sebacate, methacrylic acid copolymer, diethyl phthalate, magnesium stearate, sodium lauryl sulfate, and ascorbic acid. The capsule shells contain gelatin, titanium dioxide, and grey ink, D and C yellow #10 (EMBEDA 20 mg/0.8 mg), FD and C red #3, FD and C blue #1 (EMBEDA 30 mg/1.2 mg), D and C red #28, FD and C red #40, FD and C blue #1 (EMBEDA 50 mg/2 mg), D and C red #28, FD and C red #40, FD and C blue #1 (EMBEDA 60 mg/2.4 mg), FD and C blue #1, FD and C red #40, FD and C yellow #6 (EMBEDA 80 mg/3.2 mg), D and C yellow #10, FD and C blue #1 (EMBEDA 100 mg/4 mg).
Manufactured for: King Pharmaceuticals, Inc., 501 Fifth Street, Bristol, TN 37620
(Telephone: 1-800-776-3637)
by: Actavis Elizabeth LLC, 200 Elmora Avenue, Elizabeth, NJ 07207 USA
EMBEDA is a trademark of Alpharma Pharmaceuticals LLC, a wholly owned subsidiary of King Pharmaceuticals, Inc.
41-1126
Revision: June 2009
This Medication Guide has been approved by the U.S. Food and Drug Administration
v.10

EMBEDAMORPHINE SUFATE CAPSULE, EXTENDED RELEASE
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||