Enalapril Maleate
FULL PRESCRIBING INFORMATION: CONTENTS*
- PREGNANCY
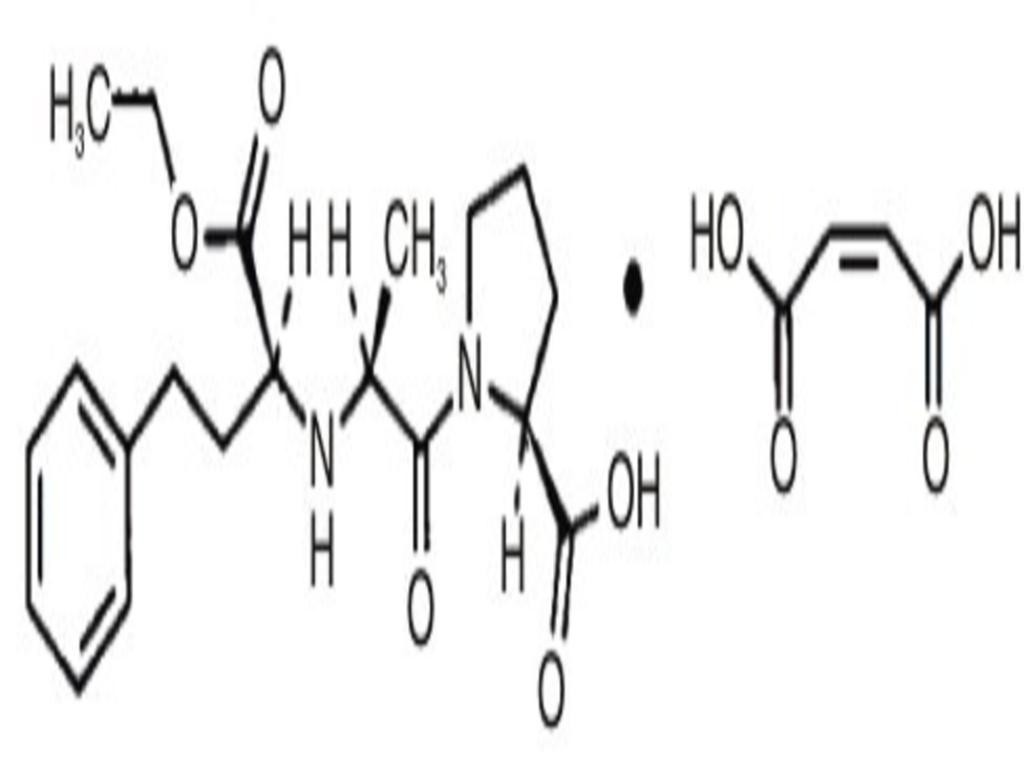
- ENALAPRIL MALEATE DESCRIPTION
- CLINICAL PHARMACOLOGY
- INDICATIONS & USAGE
- ENALAPRIL MALEATE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- ENALAPRIL MALEATE ADVERSE REACTIONS
- SPL UNCLASSIFIED
- DOSAGE & ADMINISTRATION
- OVERDOSAGE
- HOW SUPPLIED
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
PREGNANCY
ENALAPRIL MALEATE DESCRIPTION
CLINICAL PHARMACOLOGY
Mechanism of ActionPharmacokinetics and Metabolism
Pharmacodynamics and Clinical Effects
SURVIVAL (%)
INDICATIONS & USAGE
HypertensionHeart Failure
Asymptomatic Left Ventricular Dysfunction
ENALAPRIL MALEATE CONTRAINDICATIONS
WARNINGS
Anaphylactoid and Possibly Related ReactionsHypotension
Neutropenia/Agranulocytosis
Hepatic Failure
Fetal/Neonatal Morbidity and Mortality
PRECAUTIONS
GeneralInformation for Patients
Drug Interactions
Carcinogenesis, Mutagenesis, Impairment of Fertility
Pregnancy
Nursing Mothers
Pediatric Use
ENALAPRIL MALEATE ADVERSE REACTIONS
SPL UNCLASSIFIED
HEART FAILURE
Clinical Laboratory Test Findings
DOSAGE & ADMINISTRATION
HypertensionOVERDOSAGE
HOW SUPPLIED
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION

Enalapril MaleateEnalapril Maleate TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!