Epinephrine
General Injectables and Vaccines, Inc.
FULL PRESCRIBING INFORMATION: CONTENTS*
- EPINEPHRINE DESCRIPTION
- CLINICAL PHARMACOLOGY
- EPINEPHRINE INDICATIONS AND USAGE
- EPINEPHRINE CONTRAINDICATIONS
- WARNINGS SECTION
- PRECAUTIONS
- EPINEPHRINE ADVERSE REACTIONS
- EPINEPHRINE DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- SAMPLE OUTER PACKAGE LABEL
FULL PRESCRIBING INFORMATION
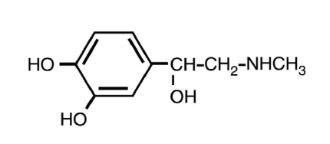
EPINEPHRINE DESCRIPTION
CLINICAL PHARMACOLOGY
Epinephrine is a sympathomimetic drug. It activates an adrenergic receptive mechanism on effector cells and imitates all actions of the sympathetic
nervous system except those on the arteries of the face and sweat glands. Epinephrine acts on both alpha and beta receptors and is the most potent alpha receptor activator.
INDICATIONS AND USAGE
In general, the most common uses of epinephrine are to relieve respiratory distress due to bronchospasm, to provide rapid relief of hypersensitivity reactions to drugs and other allergens, and to prolong the action of infiltration anesthetics. Its cardiac effects
may be of use in restoring cardiac rhythm in cardiac arrest due to various causes, but it is not used in cardiac failure or in hemorrhagic, traumatic, or cardiogenic shock.
Epinephrine is used as a hemostatic agent. It is also used in treating mucosal congestion of hay fever, rhinitis, and acute sinusitis; to relieve bronchial asthmatic paroxysms; in syncope due to complete heart block or carotid sinus hypersensitivity; for symptomatic relief of serum sickness,
urticaria, angioneurotic edema; for resuscitation in cardiac arrest following anesthetic accidents; in simple (open angle) glaucoma; for relaxation of uterine musculature and to inhibit uterine contractions. Epinephrine injection can be utilized to prolong the action of intraspinal and local
anesthetics (see CONTRAINDICATIONS).
EPINEPHRINE CONTRAINDICATIONS
Epinephrine is contraindicated in narrow-angle (congestive) glaucoma, shock, during general anesthesia with halogenated hydrocarbons or cyclopropane and in individuals with organic brain damage. Epinephrine is also contraindicated with local anesthesia
of certain areas, e.g., fingers, toes, because of the danger of vasoconstriction producing sloughing of tissue; in labor because it may delay the second stage; in cardiac dilatation and coronary insufficiency.
WARNINGS SECTION
Administer with caution to elderly people; to those with cardiovascular disease, hypertension, diabetes, or hyperthyroidism; in psychoneurotic individuals; and in pregnancy. Patients with long-standing bronchial asthma and emphysema who have developed degenerative heart disease should be administered the drug with extreme caution.
Overdosage or inadvertent intravenous injection of epinephrine may cause cerebrovascular hemorrhage resulting from the sharp rise in blood pressure.
Fatalities may also result from pulmonary edema because of the peripheral constriction and cardiac stimulation produced. Rapidly acting vasodilators such as nitrites, or alpha-blocking agents may counteract the marked pressor effects of epinephrine.
epinephrine is the preferred treatment of serious allergic or other emergency situations even though this product contains sodium metabisulfite, a sulfite that may in other products cause allergic-type reactions including anaphylactic symptoms or life-threatening or less severe asthmatic episodes in certain susceptible persons. The alternative to using epinephrine in a life-threatening situation may not be satisfactory. The presence of a sulfite in this product should not deter administration of the drug for treatment of serious allergic or other emergency situations.
PRECAUTIONS
Epinephrine Injection should be protected from exposure to light. Do not remove vial from carton until ready to use. The solution should not be used if its color is pinkish or darker than slightly yellow or if it contains a precipitate.
Epinephrine is readily destroyed by alkalies and oxidizing agents. In the latter category are oxygen, chlorine, bromine, iodine, permanganates, chromates, nitrites, and salts of easily reducible metals, especially iron.
EPINEPHRINE ADVERSE REACTIONS
Transient and minor side effects of anxiety, headache, fear, and palpitations often occur with therapeutic doses, especially in hyperthyroid individuals. Repeated local injections can result in necrosis at sites of injection from vascular constriction. “Epinephrine-fastness”can occur with prolonged use.
EPINEPHRINE DOSAGE AND ADMINISTRATION
Subcutaneously or intramuscularly — 0.2 to 1 mL. Start with a small dose and increase if required.
Note: The subcutaneous is the preferred route of administration. If given intramuscularly,
injection into the buttocks should be avoided.
For bronchial asthma and certain allergic manifestations, e.g., angioedema, urticaria, serum sickness, anaphylactic shock, use epinephrine subcutaneously. For bronchial asthma in pediatric patients, administer 0.01 mg/kg or 0.3 mg/m2 to a maximum of 0.5 mg subcutaneously, repeated
every four hours if required.
For cardiac resuscitation — A dose of 0.5 mL (0.5 mg) diluted to 10 mL with sodium chloride injection can be administered intravenously or intracardially to restore myocardial contractility. External cardiac massage should follow intracardial administration to permit the drug to enter coronary circulation. The drug should be used secondarily to unsuccessful attempts with physical or electromechanical methods.
Store at 20°-25°C (68°-77°F). excursions permitted to 15°-30°C (59° - 86°F) (See USP Controlled Room Temperature).
Protect from light
Parenteral drug products should be inspected visually for particulate matter and discoloration,
whenever solution and container permit.
HOW SUPPLIED
Epinephrine Injection, USP 1:1000 (1 mg/ml)
NDC 0517-1130-05 30 mL Multiple Dose Vial Boxes of 5
IN1130
Rev. 11/05
MG# 10069(1)
SAMPLE OUTER PACKAGE LABEL

EpinephrineEpinephrine INJECTION, SOLUTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||