EPIVIR
These highlights do not include all the information needed to use EPIVIR safely and effectively. See full prescribing information for EPIVIR.EPIVIR (lamivudine) Tablets and Oral SolutionInitial U.S. Approval: 1995
FULL PRESCRIBING INFORMATION: CONTENTS*
- RECENT MAJOR CHANGES
- Boxed Warning section
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
- DOSAGE FORMS & STRENGTHS
- EPIVIR CONTRAINDICATIONS
- WARNINGS AND PRECAUTIONS
- EPIVIR ADVERSE REACTIONS
- DRUG INTERACTIONS
- USE IN SPECIFIC POPULATIONS
- OVERDOSAGE
- EPIVIR DESCRIPTION
- CLINICAL PHARMACOLOGY
- NONCLINICAL TOXICOLOGY
- CLINICAL STUDIES
- HOW SUPPLIED
- INFORMATION FOR PATIENTS
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
Boxed Warning section
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogues alone or in combination, including lamivudine and other antiretrovirals. Suspend treatment if clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity occur [see Warnings and Precautions (5.1)].
Severe acute exacerbations of hepatitis B have been reported in patients who are co-infected with hepatitis B virus (HBV) and human immunodeficiency virus (HIV-1) and have discontinued EPIVIR. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue EPIVIR and are co-infected with HIV-1 and HBV. If appropriate, initiation of anti-hepatitis B therapy may be warranted [see Warnings and Precautions (5.2)].
EPIVIR Tablets and Oral Solution (used to treat HIV-1 infection) contain a higher dose of the active ingredient (lamivudine) than EPIVIR-HBV® Tablets and Oral Solution (used to treat chronic HBV infection). Patients with HIV-1 infection should receive only dosage forms appropriate for treatment of HIV-1 [see Warnings and Precautions (5.2)] .
INDICATIONS & USAGE
EPIVIR is a nucleoside analogue indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) infection. Limitation of use: The dosage of this product is for HIV-1 and not for HBV.
DOSAGE & ADMINISTRATION
- Adults and adolescents >16 years of age: 300 mg daily, administered as either 150 mg twice daily or 300 mg once daily. (2.1)
- Pediatric patients 3 months up to 16 years of age: Dosage should be based on body weight. (2.2)
- Patients With Renal Impairment: Doses of EPIVIR must be adjusted in accordance with renal function. (2.3)
The recommended oral dose of EPIVIR in HIV-1-infected adults and adolescents >16 years of age is 300 mg daily, administered as either 150 mg twice daily or 300 mg once daily, in combination with other antiretroviral agents. If lamivudine is administered to a patient infected with HIV-1 and HBV, the dosage indicated for HIV-1 therapy should be used as part of an appropriate combination regimen [see Warnings and Precautions (5.2)].
The recommended oral dose of EPIVIR Oral Solution in HIV-1-infected pediatric patients 3 months to 16 years of age is 4 mg/kg twice daily (up to a maximum of 150 mg twice a day), administered in combination with other antiretroviral agents.
EPIVIR is also available as a scored tablet for HIV-1-infected pediatric patients who weigh ≥14 kg and for whom a solid dosage form is appropriate. Before prescribing EPIVIR Tablets, children should be assessed for the ability to swallow tablets. If a child is unable to reliably swallow EPIVIR Tablets, the oral solution formulation should be prescribed. The recommended oral dosage of EPIVIR Tablets for HIV-1-infected pediatric patients is presented in Table 1.
|
Weight (kg) |
Dosage Regimen Using Scored 150-mg Tablet |
Total Daily Dose |
|
| AM Dose | PM Dose | ||
| 14 to 21 | ½ tablet (75 mg) | ½ tablet (75 mg) | 150 mg |
| >21 to <30 | ½ tablet (75 mg) | 1 tablet (150 mg) | 225 mg |
| ≥30 | 1 tablet (150 mg) | 1 tablet (150 mg) | 300 mg |
Dosing of EPIVIR is adjusted in accordance with renal function. Dosage adjustments are listed in Table 2 [see Clinical Pharmacology (12.3)].
| Creatinine Clearance (mL/min) | Recommended Dosage of EPIVIR |
| ≥50 | 150 mg twice daily or 300 mg once daily |
| 30-49 | 150 mg once daily |
| 15-29 | 150 mg first dose, then 100 mg once daily |
| 5-14 | 150 mg first dose, then 50 mg once daily |
| <5 | 50 mg first dose, then 25 mg once daily |
No additional dosing of EPIVIR is required after routine (4-hour) hemodialysis or peritoneal dialysis.
Although there are insufficient data to recommend a specific dose adjustment of EPIVIR in pediatric patients with renal impairment, a reduction in the dose and/or an increase in the dosing interval should be considered.
DOSAGE FORMS & STRENGTHS
-
EPIVIR Scored Tablets
150 mg, are white, diamond-shaped, scored, film-coated tablets debossed with “GX CJ7” on both sides.
-
EPIVIR Tablets
300 mg, are gray, modified diamond-shaped, film-coated tablets engraved with “GX EJ7” on one side and plain on the reverse side.
-
EPIVIR Oral Solution
A clear, colorless to pale yellow, strawberry-banana flavored liquid, containing 10 mg of lamivudine per 1 mL.
EPIVIR CONTRAINDICATIONS
EPIVIR Tablets and Oral Solution are contraindicated in patients with previously demonstrated clinically significant hypersensitivity (e.g., anaphylaxis) to any of the components of the products.
WARNINGS AND PRECAUTIONS
- Lactic acidosis and severe hepatomegaly with steatosis: Reported with the use of nucleoside analogues. Suspend treatment if clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatoxicity occur. (5.1)
- Severe acute exacerbations of hepatitis: Reported in patients who are co-infected with hepatitis B virus and HIV-1 and discontinued EPIVIR. Monitor hepatic function closely in these patients and, if appropriate, initiate anti-hepatitis B treatment. (5.2)
- Patients with HIV-1 infection should receive only dosage forms of EPIVIR appropriate for treatment of HIV-1. (5.2)
- Co-infected HIV-1/HBV Patients: Emergence of lamivudine-resistant HBV variants associated with lamivudine-containing antiretroviral regimens has been reported. (5.2)
- Emtricitabine should not be administered concomitantly with lamivudine-containing products. (5.3)
- Hepatic decompensation (some fatal) has occurred in HIV-1/HCV co-infected patients receiving interferon and ribavirin-based regimens. Monitor for treatment-associated toxicities. Discontinue EPIVIR as medically appropriate and consider dose reduction or discontinuation of interferon alfa, ribavirin, or both. (5.4)
- Pancreatitis: Use with caution in pediatric patients with a history of pancreatitis or other significant risk factors for pancreatitis. Discontinue treatment as clinically appropriate. (5.5)
- Immune reconstitution syndrome (5.6) and redistribution/accumulation of body fat (5.7) have been reported in patients treated with combination antiretroviral therapy.
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogues alone or in combination, including lamivudine and other antiretrovirals. A majority of these cases have been in women. Obesity and prolonged nucleoside exposure may be risk factors. Particular caution should be exercised when administering EPIVIR to any patient with known risk factors for liver disease; however, cases also have been reported in patients with no known risk factors. Treatment with EPIVIR should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
Posttreatment Exacerbations of Hepatitis: In clinical trials in non-HIV-1-infected patients treated with lamivudine for chronic hepatitis B, clinical and laboratory evidence of exacerbations of hepatitis have occurred after discontinuation of lamivudine. These exacerbations have been detected primarily by serum ALT elevations in addition to re-emergence of HBV DNA. Although most events appear to have been self-limited, fatalities have been reported in some cases. Similar events have been reported from postmarketing experience after changes from lamivudine-containing HIV-1 treatment regimens to non-lamivudine-containing regimens in patients infected with both HIV-1 and HBV. The causal relationship to discontinuation of lamivudine treatment is unknown. Patients should be closely monitored with both clinical and laboratory follow-up for at least several months after stopping treatment. There is insufficient evidence to determine whether re-initiation of lamivudine alters the course of posttreatment exacerbations of hepatitis.
Important Differences Among Lamivudine-Containing Products: EPIVIR Tablets and Oral Solution contain a higher dose of the same active ingredient (lamivudine) than EPIVIR-HBV Tablets and EPIVIR-HBV Oral Solution. EPIVIR-HBV was developed for patients with chronic hepatitis B. The formulation and dosage of lamivudine in EPIVIR-HBV are not appropriate for patients co-infected with HIV-1 and HBV. Safety and efficacy of lamivudine have not been established for treatment of chronic hepatitis B in patients co-infected with HIV-1 and HBV. If treatment with EPIVIR-HBV is prescribed for chronic hepatitis B for a patient with unrecognized or untreated HIV-1 infection, rapid emergence of HIV-1 resistance is likely to result because of the subtherapeutic dose and the inappropriateness of monotherapy HIV-1 treatment. If a decision is made to administer lamivudine to patients co-infected with HIV-1 and HBV, EPIVIR Tablets, EPIVIR Oral Solution, COMBIVIR® (lamivudine/zidovudine) Tablets, EPZICOM® (abacavir sulfate and lamivudine) Tablets, or TRIZIVIR® (abacavir sulfate, lamivudine, and zidovudine) Tablets should be used as part of an appropriate combination regimen.
Emergence of Lamivudine-Resistant HBV: In non–HIV-1-infected patients treated with lamivudine for chronic hepatitis B, emergence of lamivudine-resistant HBV has been detected and has been associated with diminished treatment response (see full prescribing information for EPIVIR-HBV for additional information). Emergence of hepatitis B virus variants associated with resistance to lamivudine has also been reported in HIV-1-infected patients who have received lamivudine-containing antiretroviral regimens in the presence of concurrent infection with hepatitis B virus.
EPIVIR should not be administered concomitantly with other lamivudine-containing products including EPIVIR-HBV Tablets, EPIVIR Oral Solution, COMBIVIR (lamivudine/zidovudine) Tablets, EPZICOM (abacavir sulfate and lamivudine) Tablets, or TRIZIVIR (abacavir sulfate, lamivudine, and zidovudine) or emtricitabine-containing products, including ATRIPLA® (efavirenz, emtricitabine, and tenofovir), EMTRIVA® (emtricitabine), or TRUVADA® (emtricitabine and tenofovir).
In vitro studies have shown ribavirin can reduce the phosphorylation of pyrimidine nucleoside analogues such as lamivudine. Although no evidence of a pharmacokinetic or pharmacodynamic interaction (e.g., loss of HIV-1/HCV virologic suppression) was seen when ribavirin was coadministered with lamivudine in HIV-1/HCV co-infected patients [see Clinical Pharmacology (12.3)], hepatic decompensation (some fatal) has occurred in HIV-1/HCV co-infected patients receiving combination antiretroviral therapy for HIV-1 and interferon alfa with or without ribavirin. Patients receiving interferon alfa with or without ribavirin and EPIVIR should be closely monitored for treatment-associated toxicities, especially hepatic decompensation. Discontinuation of EPIVIR should be considered as medically appropriate. Dose reduction or discontinuation of interferon alfa, ribavirin, or both should also be considered if worsening clinical toxicities are observed, including hepatic decompensation (e.g., Child-Pugh >6). See the complete prescribing information for interferon and ribavirin.
In pediatric patients with a history of prior antiretroviral nucleoside exposure, a history of pancreatitis, or other significant risk factors for the development of pancreatitis, EPIVIR should be used with caution. Treatment with EPIVIR should be stopped immediately if clinical signs, symptoms, or laboratory abnormalities suggestive of pancreatitis occur [see Adverse Reactions (6.1)].
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including EPIVIR. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Redistribution/accumulation of body fat including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and “cushingoid appearance” have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
EPIVIR ADVERSE REACTIONS
- The most common reported adverse reactions (incidence ≥15%) in adults were headache, nausea, malaise and fatigue, nasal signs and symptoms, diarrhea, and cough. (6.1)
- The most common reported adverse reactions (incidence ≥15%) in pediatric patients were fever and cough. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact GlaxoSmithKline at 1-888-825-5249 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Lactic acidosis and severe hepatomegaly with steatosis [see Boxed Warning, Warnings and Precautions (5.1)].
- Severe acute exacerbations of hepatitis B [see Boxed Warning, Warnings and Precautions (5.2)].
- Hepatic decompensation in patients co-infected with HIV-1 and Hepatitis C [see Warnings and Precautions (5.4)].
- Pancreatitis [see Warnings and Precautions (5.5)].
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adults - Clinical Trials in HIV-1: The safety profile of EPIVIR in adults is primarily based on 3,568 HIV-1-infected patients in 7 clinical trials.
The most common adverse reactions are headache, nausea, malaise, fatigue, nasal signs and symptoms, diarrhea and cough.
Selected clinical adverse reactions in ≥5% of patients during therapy with EPIVIR 150 mg twice daily plus RETROVIR® 200 mg 3 times daily for up to 24 weeks are listed in Table 3.
| Adverse Reaction |
EPIVIR 150 mg Twice Daily plus RETROVIR (n = 251) |
RETROVIRa (n = 230) |
| Body as a Whole | ||
| Headache |
35% |
27% |
| Malaise & fatigue |
27% |
23% |
| Fever or chills |
10% |
12% |
| Digestive | ||
| Nausea |
33% |
29% |
| Diarrhea |
18% |
22% |
| Nausea & vomiting |
13% |
12% |
| Anorexia and/or decreased appetite |
10% |
7% |
| Abdominal pain |
9% |
11% |
| Abdominal cramps |
6% |
3% |
| Dyspepsia |
5% |
5% |
| Nervous System | ||
| Neuropathy |
12% |
10% |
| Insomnia & other sleep disorders |
11% |
7% |
| Dizziness |
10% |
4% |
| Depressive disorders |
9% |
4% |
| Respiratory | ||
| Nasal signs & symptoms |
20% |
11% |
| Cough |
18% |
13% |
| Skin | ||
| Skin rashes |
9% |
6% |
| Musculoskeletal | ||
| Musculoskeletal pain |
12% |
10% |
| Myalgia |
8% |
6% |
| Arthralgia |
5% |
5% |
| a Either zidovudine monotherapy or zidovudine in combination with zalcitabine. |
Pancreatitis: Pancreatitis was observed in 9 out of 2,613 adult patients (0.3%) who received EPIVIR in controlled clinical trials EPV20001, NUCA3001, NUCB3001, NUCA3002, NUCB3002, and NUCB3007 [see Warnings and Precautions (5.5)].
EPIVIR 300 mg Once Daily: The types and frequencies of clinical adverse reactions reported in patients receiving EPIVIR 300 mg once daily or EPIVIR 150 mg twice daily (in 3-drug combination regimens in EPV20001 and EPV40001) for 48 weeks were similar.
Selected laboratory abnormalities observed during therapy are summarized in Table 4.
|
Test (Threshold Level) |
24-Week Surrogate Endpoint Studiesa |
Clinical Endpoint Studya |
||
|
EPIVIR plus RETROVIR |
RETROVIRb |
EPIVIR plus Current Therapy |
Placebo plus Current Therapyc |
|
|
Absolute neutrophil count (<750/mm3) |
7.2% |
5.4% |
15% |
13% |
|
Hemoglobin (<8.0 g/dL) |
2.9% |
1.8% |
2.2% |
3.4% |
|
Platelets (<50,000/mm3) |
0.4% |
1.3% |
2.8% |
3.8% |
|
ALT (>5.0 x ULN) |
3.7% |
3.6% |
3.8% |
1.9% |
|
AST (>5.0 x ULN) |
1.7% |
1.8% |
4.0% |
2.1% |
|
Bilirubin (>2.5 x ULN) |
0.8% |
0.4% |
ND |
ND |
|
Amylase (>2.0 x ULN) |
4.2% |
1.5% |
2.2% |
1.1% |
| a The median duration on study was 12 months. | ||||
| b Either zidovudine monotherapy or zidovudine in combination with zalcitabine. | ||||
| c Current therapy was either zidovudine, zidovudine plus didanosine, or zidovudine plus zalcitabine. | ||||
| ULN = Upper limit of normal. | ||||
| ND = Not done. |
The frequencies of selected laboratory abnormalities reported in patients receiving EPIVIR 300 mg once daily or EPIVIR 150 mg twice daily (in 3-drug combination regimens in EPV20001 and EPV40001) were similar.
Pediatric Patients – Clinical Trials in HIV-1: EPIVIR Oral Solution has been studied in 638 pediatric patients 3 months to 18 years of age in 3 clinical trials.
Selected clinical adverse reactions and physical findings with a ≥5% frequency during therapy with EPIVIR 4 mg/kg twice daily plus RETROVIR 160 mg/m2 3 times daily in therapy-naive (≤56 days of antiretroviral therapy) pediatric patients are listed in Table 5.
|
Adverse Reaction |
EPIVIR plus RETROVIR (n = 236) |
Didanosine (n = 235) |
|
Body as a Whole |
||
|
Fever |
25% |
32% |
|
Digestive |
||
|
Hepatomegaly |
11% |
11% |
|
Nausea & vomiting |
8% |
7% |
|
Diarrhea |
8% |
6% |
|
Stomatitis |
6% |
12% |
|
Splenomegaly |
5% |
8% |
|
Respiratory |
||
|
Cough |
15% |
18% |
|
Abnormal breath sounds/wheezing |
7% |
9% |
|
Ear, Nose, and Throat |
||
|
Signs or symptoms of earsa |
7% |
6% |
|
Nasal discharge or congestion |
8% |
11% |
|
Other |
||
|
Skin rashes |
12% |
14% |
|
Lymphadenopathy |
9% |
11% |
| a Includes pain, discharge, erythema, or swelling of an ear. |
Pancreatitis: Pancreatitis, which has been fatal in some cases, has been observed in antiretroviral nucleoside-experienced pediatric patients receiving EPIVIR alone or in combination with other antiretroviral agents. In an open-label dose-escalation study (NUCA2002), 14 patients (14%) developed pancreatitis while receiving monotherapy with EPIVIR. Three of these patients died of complications of pancreatitis. In a second open-label study (NUCA2005), 12 patients (18%) developed pancreatitis. In Study ACTG300, pancreatitis was not observed in 236 patients randomized to EPIVIR plus RETROVIR. Pancreatitis was observed in 1 patient in this study who received open-label EPIVIR in combination with RETROVIR and ritonavir following discontinuation of didanosine monotherapy [see Warnings and Precautions (5.5)].
Paresthesias and Peripheral Neuropathies: Paresthesias and peripheral neuropathies were reported in 15 patients (15%) in Study NUCA2002, 6 patients (9%) in Study NUCA2005, and 2 patients (<1%) in Study ACTG300.
Selected laboratory abnormalities experienced by therapy-naive (≤56 days of antiretroviral therapy) pediatric patients are listed in Table 6.
|
Test (Threshold Level) |
EPIVIR plus RETROVIR |
Didanosine |
|
Absolute neutrophil count (<400/mm3) |
8% |
3% |
|
Hemoglobin (<7.0 g/dL) |
4% |
2% |
|
Platelets (<50,000/mm3) |
1% |
3% |
|
ALT (>10 x ULN) |
1% |
3% |
|
AST (>10 x ULN) |
2% |
4% |
|
Lipase (>2.5 x ULN) |
3% |
3% |
|
Total Amylase (>2.5 x ULN) |
3% |
3% |
| ULN = Upper limit of normal. |
Neonates - Clinical Trials in HIV-1: Limited short-term safety information is available from 2 small, uncontrolled studies in South Africa in neonates receiving lamivudine with or without zidovudine for the first week of life following maternal treatment starting at Week 38 or 36 of gestation [see Clinical Pharmacology (12.3)]. Selected adverse reactions reported in these neonates included increased liver function tests, anemia, diarrhea, electrolyte disturbances, hypoglycemia, jaundice and hepatomegaly, rash, respiratory infections, and sepsis; 3 neonates died (1 from gastroenteritis with acidosis and convulsions, 1 from traumatic injury, and 1 from unknown causes). Two other nonfatal gastroenteritis or diarrhea cases were reported, including 1 with convulsions; 1 infant had transient renal insufficiency associated with dehydration. The absence of control groups limits assessments of causality, but it should be assumed that perinatally exposed infants may be at risk for adverse reactions comparable to those reported in pediatric and adult HIV-1-infected patients treated with lamivudine-containing combination regimens. Long-term effects of in utero and infant lamivudine exposure are not known.
In addition to adverse reactions reported from clinical trials, the following adverse reactions have been reported during postmarketing use of EPIVIR. Because these reactions are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to lamivudine.
Body as a Whole: Redistribution/accumulation of body fat [see Warnings and Precautions (5.7)].
Endocrine and Metabolic: Hyperglycemia.
General: Weakness.
Hemic and Lymphatic: Anemia (including pure red cell aplasia and severe anemias progressing on therapy).
Hepatic and Pancreatic: Lactic acidosis and hepatic steatosis, posttreatment exacerbation of hepatitis B [see Boxed Warning, Warnings and Precautions (5.1, 5.2)].
Hypersensitivity: Anaphylaxis, urticaria.
Musculoskeletal: Muscle weakness, CPK elevation, rhabdomyolysis.
Skin: Alopecia, pruritus.
DRUG INTERACTIONS
Lamivudine is predominantly eliminated in the urine by active organic cationic secretion. The possibility of interactions with other drugs administered concurrently should be considered, particularly when their main route of elimination is active renal secretion via the organic cationic transport system (e.g., trimethoprim). No data are available regarding interactions with other drugs that have renal clearance mechanisms similar to that of lamivudine.
Although no evidence of a pharmacokinetic or pharmacodynamic interaction (e.g., loss of HIV-1/HCV virologic suppression) was seen when ribavirin was coadministered with lamivudine in HIV-1/HCV co-infected patients, hepatic decompensation (some fatal) has occurred in HIV-1/HCV co-infected patients receiving combination antiretroviral therapy for HIV-1 and interferon alfa with or without ribavirin [see Warnings and Precautions (5.4), Clinical Pharmacology (12.3)].
Lamivudine and zalcitabine may inhibit the intracellular phosphorylation of one another. Therefore, use of lamivudine in combination with zalcitabine is not recommended.
No change in dose of either drug is recommended. There is no information regarding the effect on lamivudine pharmacokinetics of higher doses of TMP/SMX such as those used to treat PCP.
A drug interaction study showed no clinically significant interaction between EPIVIR and zidovudine.
USE IN SPECIFIC POPULATIONS
- Pregnancy: Physicians are encouraged to register patients in the Antiretroviral Pregnancy Registry by calling 1-800-258-4263. (8.1)
Revised: April 2009
EPV:2PI
Pregnancy Category C. There are no adequate and well-controlled studies of EPIVIR in pregnant women. Animal reproduction studies in rats and rabbits revealed no evidence of teratogenicity. Increased early embryolethality occurred in rabbits at exposure levels similar to those in humans. EPIVIR should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Lamivudine pharmacokinetics were studied in pregnant women during 2 clinical studies conducted in South Africa. The study assessed pharmacokinetics in: 16 women at 36 weeks gestation using 150 mg lamivudine twice daily with zidovudine, 10 women at 38 weeks gestation using 150 mg lamivudine twice daily with zidovudine, and 10 women at 38 weeks gestation using lamivudine 300 mg twice daily without other antiretrovirals. These studies were not designed or powered to provide efficacy information. Lamivudine pharmacokinetics in pregnant women were similar to those seen in non-pregnant adults and in postpartum women. Lamivudine concentrations were generally similar in maternal, neonatal, and umbilical cord serum samples. In a subset of subjects, lamivudine amniotic fluid specimens were collected following natural rupture of membranes. Amniotic fluid concentrations of lamivudine were typically 2 times greater than maternal serum levels and ranged from 1.2 to 2.5 mcg/mL (150 mg twice daily) and 2.1 to 5.2 mcg/mL (300 mg twice daily). It is not known whether risks of adverse events associated with lamivudine are altered in pregnant women compared with other HIV-1-infected patients.
Animal reproduction studies performed at oral doses up to 130 and 60 times the adult dose in rats and rabbits, respectively, revealed no evidence of teratogenicity due to lamivudine. Increased early embryolethality occurred in rabbits at exposure levels similar to those in humans. However, there was no indication of this effect in rats at exposure levels up to 35 times those in humans. Based on animal studies, lamivudine crosses the placenta and is transferred to the fetus [see Nonclinical Toxicology (13.2)].
Antiretroviral Pregnancy Registry: To monitor maternal-fetal outcomes of pregnant women exposed to lamivudine, a Pregnancy Registry has been established. Physicians are encouraged to register patients by calling 1-800-258-4263.
The Centers for Disease Control and Prevention recommend that HIV-1-infected mothers in the United States not breastfeed their infants to avoid risking postnatal transmission of HIV-1 infection. Because of the potential for serious adverse reactions in nursing infants and HIV-1 transmission, mothers should be instructed not to breastfeed if they are receiving lamivudine.
Lamivudine is excreted into human milk. Samples of breast milk obtained from 20 mothers receiving lamivudine monotherapy (300 mg twice daily) or combination therapy (150 mg lamivudine twice daily and 300 mg zidovudine twice daily) had measurable concentrations of lamivudine.
The safety and effectiveness of twice-daily EPIVIR in combination with other antiretroviral agents have been established in pediatric patients 3 months and older [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.2)].
Clinical studies of EPIVIR did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. In particular, because lamivudine is substantially excreted by the kidney and elderly patients are more likely to have decreased renal function, renal function should be monitored and dosage adjustments should be made accordingly [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)].
Reduction of the dosage of EPIVIR is recommended for patients with impaired renal function [see Dosage and Administration (2.3), Clinical Pharmacology (12.3)].
OVERDOSAGE
There is no known antidote for EPIVIR. One case of an adult ingesting 6 g of EPIVIR was reported; there were no clinical signs or symptoms noted and hematologic tests remained normal. Two cases of pediatric overdose were reported in Study ACTG300. One case involved a single dose of 7 mg/kg of EPIVIR; the second case involved use of 5 mg/kg of EPIVIR twice daily for 30 days. There were no clinical signs or symptoms noted in either case. Because a negligible amount of lamivudine was removed via (4-hour) hemodialysis, continuous ambulatory peritoneal dialysis, and automated peritoneal dialysis, it is not known if continuous hemodialysis would provide clinical benefit in a lamivudine overdose event. If overdose occurs, the patient should be monitored, and standard supportive treatment applied as required.
EPIVIR DESCRIPTION
EPIVIR (also known as 3TC) is a brand name for lamivudine, a synthetic nucleoside analogue with activity against HIV-1 and HBV. The chemical name of lamivudine is (2R,cis)-4-amino-1-(2-hydroxymethyl-1,3-oxathiolan-5-yl)-(1H)-pyrimidin-2-one. Lamivudine is the (-)enantiomer of a dideoxy analogue of cytidine. Lamivudine has also been referred to as (-)2′,3′-dideoxy, 3′-thiacytidine. It has a molecular formula of C8H11N3O3S and a molecular weight of 229.3. It has the following structural formula:

Lamivudine is a white to off-white crystalline solid with a solubility of approximately 70 mg/mL in water at 20°C.
EPIVIR Tablets are for oral administration. Each scored 150-mg film-coated tablet contains 150 mg of lamivudine and the inactive ingredients hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium starch glycolate, and titanium dioxide.
Each 300-mg film-coated tablet contains 300 mg of lamivudine and the inactive ingredients black iron oxide, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium starch glycolate, and titanium dioxide.
EPIVIR Oral Solution is for oral administration. One milliliter (1 mL) of EPIVIR Oral Solution contains 10 mg of lamivudine (10 mg/mL) in an aqueous solution and the inactive ingredients artificial strawberry and banana flavors, citric acid (anhydrous), methylparaben, propylene glycol, propylparaben, sodium citrate (dihydrate), and sucrose (200 mg).
CLINICAL PHARMACOLOGY
Lamivudine is an antiviral agent [see Clinical Pharmacology (12.4)].
Pharmacokinetics in Adults: The pharmacokinetic properties of lamivudine have been studied in asymptomatic, HIV-1-infected adult patients after administration of single intravenous (IV) doses ranging from 0.25 to 8 mg/kg, as well as single and multiple (twice-daily regimen) oral doses ranging from 0.25 to 10 mg/kg.
The pharmacokinetic properties of lamivudine have also been studied as single and multiple oral doses ranging from 5 mg to 600 mg/day administered to HBV-infected patients.
The steady-state pharmacokinetic properties of the EPIVIR 300-mg tablet once daily for 7 days compared with the EPIVIR 150-mg tablet twice daily for 7 days were assessed in a crossover study in 60 healthy volunteers. EPIVIR 300 mg once daily resulted in lamivudine exposures that were similar to EPIVIR 150 mg twice daily with respect to plasma AUC24,ss; however, Cmax,ss was 66% higher and the trough value was 53% lower compared with the 150-mg twice-daily regimen. Intracellular lamivudine triphosphate exposures in peripheral blood mononuclear cells were also similar with respect to AUC24,ss and Cmax24,ss; however, trough values were lower compared with the 150-mg twice-daily regimen. Inter-subject variability was greater for intracellular lamivudine triphosphate concentrations versus lamivudine plasma trough concentrations. The clinical significance of observed differences for both plasma lamivudine concentrations and intracellular lamivudine triphosphate concentrations is not known.
Absorption and Bioavailability: Lamivudine was rapidly absorbed after oral administration in HIV-1-infected patients. Absolute bioavailability in 12 adult patients was 86% ± 16% (mean ± SD) for the 150-mg tablet and 87% ± 13% for the oral solution. After oral administration of 2 mg/kg twice a day to 9 adults with HIV-1, the peak serum lamivudine concentration (Cmax) was 1.5 ± 0.5 mcg/mL (mean ± SD). The area under the plasma concentration versus time curve (AUC) and Cmax increased in proportion to oral dose over the range from 0.25 to 10 mg/kg.
The accumulation ratio of lamivudine in HIV-1-positive asymptomatic adults with normal renal function was 1.50 following 15 days of oral administration of 2 mg/kg twice daily.
Effects of Food on Oral Absorption: An investigational 25-mg dosage form of lamivudine was administered orally to 12 asymptomatic, HIV-1-infected patients on 2 occasions, once in the fasted state and once with food (1,099 kcal; 75 grams fat, 34 grams protein, 72 grams carbohydrate). Absorption of lamivudine was slower in the fed state (Tmax: 3.2 ± 1.3 hours) compared with the fasted state (Tmax: 0.9 ± 0.3 hours); Cmax in the fed state was 40% ± 23% (mean ± SD) lower than in the fasted state. There was no significant difference in systemic exposure (AUC∞) in the fed and fasted states; therefore, EPIVIR Tablets and Oral Solution may be administered with or without food.
Distribution: The apparent volume of distribution after IV administration of lamivudine to 20 patients was 1.3 ± 0.4 L/kg, suggesting that lamivudine distributes into extravascular spaces. Volume of distribution was independent of dose and did not correlate with body weight.
Binding of lamivudine to human plasma proteins is low (<36%). In vitro studies showed that over the concentration range of 0.1 to 100 mcg/mL, the amount of lamivudine associated with erythrocytes ranged from 53% to 57% and was independent of concentration.
Metabolism: Metabolism of lamivudine is a minor route of elimination. In man, the only known metabolite of lamivudine is the trans-sulfoxide metabolite. Within 12 hours after a single oral dose of lamivudine in 6 HIV-1-infected adults, 5.2% ± 1.4% (mean ± SD) of the dose was excreted as the trans-sulfoxide metabolite in the urine. Serum concentrations of this metabolite have not been determined.
Elimination: The majority of lamivudine is eliminated unchanged in urine by active organic cationic secretion. In 9 healthy subjects given a single 300-mg oral dose of lamivudine, renal clearance was 199.7 ± 56.9 mL/min (mean ± SD). In 20 HIV-1-infected patients given a single IV dose, renal clearance was 280.4 ± 75.2 mL/min (mean ± SD), representing 71% ± 16% (mean ± SD) of total clearance of lamivudine.
In most single-dose studies in HIV-1-infected patients, HBV-infected patients, or healthy subjects with serum sampling for 24 hours after dosing, the observed mean elimination half-life (t½) ranged from 5 to 7 hours. In HIV-1-infected patients, total clearance was 398.5 ± 69.1 mL/min (mean ± SD). Oral clearance and elimination half-life were independent of dose and body weight over an oral dosing range of 0.25 to 10 mg/kg.
Special Populations: Renal Impairment: The pharmacokinetic properties of lamivudine have been determined in a small group of HIV-1-infected adults with impaired renal function (Table 7).
|
Parameter |
Creatinine Clearance Criterion (Number of Subjects) |
||
|
>60 mL/min (n = 6) |
10-30 mL/min (n = 4) |
<10 mL/min (n = 6) |
|
|
Creatinine clearance (mL/min) |
111 ± 14 |
28 ± 8 |
6 ± 2 |
|
Cmax (mcg/mL) |
2.6 ± 0.5 |
3.6 ± 0.8 |
5.8 ± 1.2 |
|
AUC∞ (mcg•hr/mL) |
11.0 ± 1.7 |
48.0 ± 19 |
157 ± 74 |
|
Cl/F (mL/min) |
464 ± 76 |
114 ± 34 |
36 ± 11 |
Exposure (AUC∞), Cmax, and half-life increased with diminishing renal function (as expressed by creatinine clearance). Apparent total oral clearance (Cl/F) of lamivudine decreased as creatinine clearance decreased. Tmax was not significantly affected by renal function. Based on these observations, it is recommended that the dosage of lamivudine be modified in patients with renal impairment [see Dosage and Administration (2.3)].
Based on a study in otherwise healthy subjects with impaired renal function, hemodialysis increased lamivudine clearance from a mean of 64 to 88 mL/min; however, the length of time of hemodialysis (4 hours) was insufficient to significantly alter mean lamivudine exposure after a single-dose administration. Continuous ambulatory peritoneal dialysis and automated peritoneal dialysis have negligible effects on lamivudine clearance. Therefore, it is recommended, following correction of dose for creatinine clearance, that no additional dose modification be made after routine hemodialysis or peritoneal dialysis.
It is not known whether lamivudine can be removed by continuous (24-hour) hemodialysis.
The effects of renal impairment on lamivudine pharmacokinetics in pediatric patients are not known.
Hepatic Impairment: The pharmacokinetic properties of lamivudine have been determined in adults with impaired hepatic function. Pharmacokinetic parameters were not altered by diminishing hepatic function; therefore, no dose adjustment for lamivudine is required for patients with impaired hepatic function. Safety and efficacy of lamivudine have not been established in the presence of decompensated liver disease.
Pediatric Patients: In Study NUCA2002, pharmacokinetic properties of lamivudine were assessed in a subset of 57 HIV-1-infected pediatric patients (age range: 4.8 months to 16 years, weight range: 5 to 66 kg) after oral and IV administration of 1, 2, 4, 8, 12, and 20 mg/kg/day. In the 9 infants and children (range: 5 months to 12 years of age) receiving oral solution 4 mg/kg twice daily (the usual recommended pediatric dose), absolute bioavailability was 66% ± 26% (mean ± SD), which was less than the 86% ± 16% (mean ± SD) observed in adults. The mechanism for the diminished absolute bioavailability of lamivudine in infants and children is unknown.
Systemic clearance decreased with increasing age in pediatric patients, as shown in Figure 1.
Figure 1. Systemic Clearance (L/hr•kg) of Lamivudine in Relation to Age

After oral administration of lamivudine 4 mg/kg twice daily to 11 pediatric patients ranging from 4 months to 14 years of age, Cmax was 1.1 ± 0.6 mcg/mL and half-life was 2.0 ± 0.6 hours. (In adults with similar blood sampling, the half-life was 3.7 ± 1 hours.) Total exposure to lamivudine, as reflected by mean AUC values, was comparable between pediatric patients receiving an 8-mg/kg/day dose and adults receiving a 4-mg/kg/day dose.
Distribution of lamivudine into cerebrospinal fluid (CSF) was assessed in 38 pediatric patients after multiple oral dosing with lamivudine. CSF samples were collected between 2 and 4 hours postdose. At the dose of 8 mg/kg/day, CSF lamivudine concentrations in 8 patients ranged from 5.6% to 30.9% (mean ± SD of 14.2% ± 7.9%) of the concentration in a simultaneous serum sample, with CSF lamivudine concentrations ranging from 0.04 to 0.3 mcg/mL.
Limited, uncontrolled pharmacokinetic and safety data are available from administration of lamivudine (and zidovudine) to 36 infants up to 1 week of age in 2 studies in South Africa. In these studies, lamivudine clearance was substantially reduced in 1-week-old neonates relative to pediatric patients (>3 months of age) studied previously. There is insufficient information to establish the time course of changes in clearance between the immediate neonatal period and the age-ranges >3 months old [see Adverse Reactions (6.1)].
Geriatric Patients: The pharmacokinetics of lamivudine after administration of EPIVIR to patients over 65 years of age have not been studied [see Use in Specific Populations (8.5)].
Gender: There are no significant gender differences in lamivudine pharmacokinetics.
Race: There are no significant racial differences in lamivudine pharmacokinetics.
Drug Interactions: Interferon Alfa: There was no significant pharmacokinetic interaction between lamivudine and interferon alfa in a study of 19 healthy male subjects [see Warnings and Precautions (5.4)].
Ribavirin: In vitro data indicate ribavirin reduces phosphorylation of lamivudine, stavudine, and zidovudine. However, no pharmacokinetic (e.g., plasma concentrations or intracellular triphosphorylated active metabolite concentrations) or pharmacodynamic (e.g., loss of HIV-1/HCV virologic suppression) interaction was observed when ribavirin and lamivudine (n = 18), stavudine (n = 10), or zidovudine (n = 6) were coadministered as part of a multi-drug regimen to HIV-1/HCV co-infected patients [see Warnings and Precautions (5.4)].
Trimethoprim/Sulfamethoxazole: Lamivudine and TMP/SMX were coadministered to 14 HIV-1-positive patients in a single-center, open-label, randomized, crossover study. Each patient received treatment with a single 300-mg dose of lamivudine and TMP 160 mg/SMX 800 mg once a day for 5 days with concomitant administration of lamivudine 300 mg with the fifth dose in a crossover design. Coadministration of TMP/SMX with lamivudine resulted in an increase of 43% ± 23% (mean ± SD) in lamivudine AUC∞, a decrease of 29% ± 13% in lamivudine oral clearance, and a decrease of 30% ± 36% in lamivudine renal clearance. The pharmacokinetic properties of TMP and SMX were not altered by coadministration with lamivudine [see Drug Interactions (7.3)].
Zidovudine: No clinically significant alterations in lamivudine or zidovudine pharmacokinetics were observed in 12 asymptomatic HIV-1-infected adult patients given a single dose of zidovudine (200 mg) in combination with multiple doses of lamivudine (300 mg q 12 hr) [see Drug Interactions (7.4)].
Mechanism of Action: Intracellularly, lamivudine is phosphorylated to its active 5′-triphosphate metabolite, lamivudine triphosphate (3TC-TP). The principal mode of action of 3TC-TP is the inhibition of HIV-1 reverse transcriptase (RT) via DNA chain termination after incorporation of the nucleotide analogue into viral DNA. 3TC-TP is a weak inhibitor of mammalian DNA polymerases α, β, and γ.
Antiviral Activity: The antiviral activity of lamivudine against HIV-1 was assessed in a number of cell lines (including monocytes and fresh human peripheral blood lymphocytes) using standard susceptibility assays. EC50 values (50% effective concentrations) were in the range of 0.003 to 15 µM (1 μM = 0.23 mcg/mL). HIV-1 from therapy-naive subjects with no amino acid substitutions associated with resistance gave median EC50 values of 0.429 µM (range: 0.200 to 2.007 µM) from Virco (n = 92 baseline samples from COLA40263) and 2.35 µM (1.37 to 3.68 µM) from Monogram Biosciences (n = 135 baseline samples from ESS30009). The EC50 values of lamivudine against different HIV-1 clades (A-G) ranged from 0.001 to 0.120 µM, and against HIV-2 isolates from 0.003 to 0.120 μM in peripheral blood mononuclear cells. Ribavirin (50 μM) decreased the anti-HIV-1 activity of lamivudine by 3.5 fold in MT-4 cells. In HIV-1-infected MT-4 cells, lamivudine in combination with zidovudine at various ratios exhibited synergistic antiretroviral activity. Please see the full prescribing information for EPIVIR-HBV for information regarding the inhibitory activity of lamivudine against HBV.
Resistance: Lamivudine-resistant variants of HIV-1 have been selected in cell culture. Genotypic analysis showed that the resistance was due to a specific amino acid substitution in the HIV-1 reverse transcriptase at codon 184 changing the methionine to either isoleucine or valine (M184V/I).
HIV-1 strains resistant to both lamivudine and zidovudine have been isolated from patients. Susceptibility of clinical isolates to lamivudine and zidovudine was monitored in controlled clinical trials. In patients receiving lamivudine monotherapy or combination therapy with lamivudine plus zidovudine, HIV-1 isolates from most patients became phenotypically and genotypically resistant to lamivudine within 12 weeks. In some patients harboring zidovudine-resistant virus at baseline, phenotypic sensitivity to zidovudine was restored by 12 weeks of treatment with lamivudine and zidovudine. Combination therapy with lamivudine plus zidovudine delayed the emergence of mutations conferring resistance to zidovudine.
Lamivudine-resistant HBV isolates develop substitutions (rtM204V/I) in the YMDD motif of the catalytic domain of the viral reverse transcriptase. rtM204V/I substitutions are frequently accompanied by other substitutions (rtV173L, rtL180M) which enhance the level of lamivudine resistance or act as compensatory mutations improving replication efficiency. Other substitutions detected in lamivudine-resistant HBV isolates include: rtL80I and rtA181T. Similar HBV mutants have been reported in HIV-1-infected patients who received lamivudine-containing antiretroviral regimens in the presence of concurrent infection with hepatitis B virus [see Warnings and Precautions (5.2)].
Cross-Resistance: Lamivudine-resistant HIV-1 mutants were cross-resistant to didanosine (ddI) and zalcitabine (ddC). In some patients treated with zidovudine plus didanosine or zalcitabine, isolates resistant to multiple reverse transcriptase inhibitors, including lamivudine, have emerged.
Genotypic and Phenotypic Analysis of On-Therapy HIV-1 Isolates From Patients With Virologic Failure: Study EPV20001: Fifty-three of 554 (10%) patients enrolled in EPV20001 were identified as virological failures (plasma HIV-1 RNA level ≥400 copies/mL) by Week 48. Twenty-eight patients were randomized to the lamivudine once-daily treatment group and 25 to the lamivudine twice-daily treatment group. The median baseline plasma HIV-1 RNA levels of patients in the lamivudine once-daily group and lamivudine twice-daily group were 4.9 log10 copies/mL and 4.6 log10 copies/mL, respectively.
Genotypic analysis of on-therapy isolates from 22 patients identified as virologic failures in the lamivudine once-daily group showed that isolates from 0/22 patients contained treatment-emergent amino acid substitutions associated with zidovudine resistance (M41L, D67N, K70R, L210W, T215Y/F, or K219Q/E), isolates from 10/22 patients contained treatment-emergent amino acid substitutions associated with efavirenz resistance (L100I, K101E, K103N, V108I, or Y181C), and isolates from 8/22 patients contained a treatment-emergent lamivudine resistance-associated substitution (M184I or M184V).
Genotypic analysis of on-therapy isolates from patients (n = 22) in the lamivudine twice-daily treatment group showed that isolates from 1/22 patients contained treatment-emergent zidovudine resistance substitutions, isolates from 7/22 contained treatment-emergent efavirenz resistance substitutions, and isolates from 5/22 contained treatment-emergent lamivudine resistance substitutions.
Phenotypic analysis of baseline-matched on-therapy HIV-1 isolates from patients (n = 13) receiving lamivudine once daily showed that isolates from 12/13 patients were susceptible to zidovudine; isolates from 8/13 patients exhibited a 25- to 295-fold decrease in susceptibility to efavirenz, and isolates from 7/13 patients showed an 85- to 299-fold decrease in susceptibility to lamivudine.
Phenotypic analysis of baseline-matched on-therapy HIV-1 isolates from patients (n = 13) receiving lamivudine twice daily showed that isolates from all 13 patients were susceptible to zidovudine; isolates from 3/13 patients exhibited a 21- to 342-fold decrease in susceptibility to efavirenz, and isolates from 4/13 patients exhibited a 29- to 159-fold decrease in susceptibility to lamivudine.
Study EPV40001: Fifty patients received zidovudine 300 mg twice daily plus abacavir 300 mg twice daily plus lamivudine 300 mg once daily and 50 patients received zidovudine 300 mg plus abacavir 300 mg plus lamivudine 150 mg all twice daily. The median baseline plasma HIV-1 RNA levels for patients in the 2 groups were 4.79 log10 copies/mL and 4.83 log10 copies/mL, respectively. Fourteen of 50 patients in the lamivudine once-daily treatment group and 9 of 50 patients in the lamivudine twice-daily group were identified as virologic failures.
Genotypic analysis of on-therapy HIV-1 isolates from patients (n = 9) in the lamivudine once-daily treatment group showed that isolates from 6 patients had an abacavir and/or lamivudine resistance-associated substitution M184V alone. On-therapy isolates from patients (n = 6) receiving lamivudine twice daily showed that isolates from 2 patients had M184V alone, and isolates from 2 patients harbored the M184V substitution in combination with zidovudine resistance-associated amino acid substitutions.
Phenotypic analysis of on-therapy isolates from patients (n = 6) receiving lamivudine once daily showed that HIV-1 isolates from 4 patients exhibited a 32- to 53-fold decrease in susceptibility to lamivudine. HIV-1 isolates from these 6 patients were susceptible to zidovudine.
Phenotypic analysis of on-therapy isolates from patients (n = 4) receiving lamivudine twice daily showed that HIV-1 isolates from 1 patient exhibited a 45-fold decrease in susceptibility to lamivudine and a 4.5-fold decrease in susceptibility to zidovudine.
NONCLINICAL TOXICOLOGY
Long-term carcinogenicity studies with lamivudine in mice and rats showed no evidence of carcinogenic potential at exposures up to 10 times (mice) and 58 times (rats) those observed in humans at the recommended therapeutic dose for HIV-1 infection. Lamivudine was not active in a microbial mutagenicity screen or an in vitro cell transformation assay, but showed weak in vitro mutagenic activity in a cytogenetic assay using cultured human lymphocytes and in the mouse lymphoma assay. However, lamivudine showed no evidence of in vivo genotoxic activity in the rat at oral doses of up to 2,000 mg/kg, producing plasma levels of 35 to 45 times those in humans at the recommended dose for HIV-1 infection. In a study of reproductive performance, lamivudine administered to rats at doses up to 4,000 mg/kg/day, producing plasma levels 47 to 70 times those in humans, revealed no evidence of impaired fertility and no effect on the survival, growth, and development to weaning of the offspring.
Reproduction studies have been performed in rats and rabbits at orally administered doses up to 4,000 mg/kg/day and 1,000 mg/kg/day, respectively, producing plasma levels up to approximately 35 times that for the adult HIV dose. No evidence of teratogenicity due to lamivudine was observed. Evidence of early embryolethality was seen in the rabbit at exposure levels similar to those observed in humans, but there was no indication of this effect in the rat at exposure levels up to 35 times those in humans. Studies in pregnant rats and rabbits showed that lamivudine is transferred to the fetus through the placenta.
CLINICAL STUDIES
The use of EPIVIR is based on the results of clinical studies in HIV-1-infected patients in combination regimens with other antiretroviral agents. Information from trials with clinical endpoints or a combination of CD4+ cell counts and HIV-1 RNA measurements is included below as documentation of the contribution of lamivudine to a combination regimen in controlled trials.
Clinical Endpoint Study: NUCB3007 (CAESAR) was a multi-center, double-blind, placebo-controlled study comparing continued current therapy (zidovudine alone [62% of patients] or zidovudine with didanosine or zalcitabine [38% of patients]) to the addition of EPIVIR or EPIVIR plus an investigational non-nucleoside reverse transcriptase inhibitor (NNRTI), randomized 1:2:1. A total of 1,816 HIV-1-infected adults with 25 to 250 CD4+ cells/mm3 (median = 122 cells/mm3) at baseline were enrolled: median age was 36 years, 87% were male, 84% were nucleoside-experienced, and 16% were therapy-naive. The median duration on study was 12 months. Results are summarized in Table 8.
|
Endpoint |
Current Therapy (n = 460) |
EPIVIR plus Current Therapy (n = 896) |
EPIVIR plus an NNRTIa plus Current Therapy (n = 460) |
|
HIV progression or death |
90 (19.6%) |
86 (9.6%) |
41 (8.9%) |
|
Death |
27 (5.9%) |
23 (2.6%) |
14 (3.0%) |
|
a An investigational non-nucleoside reverse transcriptase inhibitor not approved in the United States. |
Surrogate Endpoint Studies: Dual Nucleoside Analogue Studies: Principal clinical trials in the initial development of lamivudine compared lamivudine/zidovudine combinations with zidovudine monotherapy or with zidovudine plus zalcitabine. These studies demonstrated the antiviral effect of lamivudine in a 2-drug combination. More recent uses of lamivudine in treatment of HIV-1 infection incorporate it into multiple-drug regimens containing at least 3 antiretroviral drugs for enhanced viral suppression.
Dose Regimen Comparison Surrogate Endpoint Studies in Therapy-Naive Adults: EPV20001 was a multi-center, double-blind, controlled study in which patients were randomized 1:1 to receive EPIVIR 300 mg once daily or EPIVIR 150 mg twice daily, in combination with zidovudine 300 mg twice daily and efavirenz 600 mg once daily. A total of 554 antiretroviral treatment-naive HIV-1-infected adults enrolled: male (79%), Caucasian (50%), median age of 35 years, baseline CD4+ cell counts of 69 to 1,089 cells/mm3 (median = 362 cells/mm3), and median baseline plasma HIV-1 RNA of 4.66 log10 copies/mL. Outcomes of treatment through 48 weeks are summarized in Figure 2 and Table 9.
Figure 2. Virologic Response Through Week 48, EPV20001ab(Intent-to-Treat)
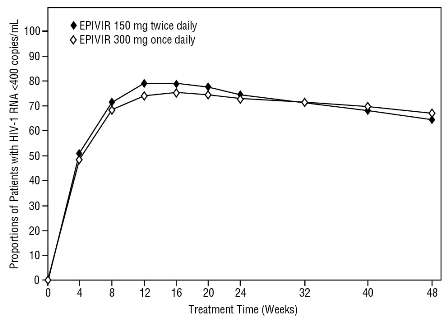
a Roche AMPLICOR HIV-1 MONITOR.
bResponders at each visit are patients who had achieved and maintained HIV-1 RNA <400 copies/mL without discontinuation by that visit.
|
Outcome |
EPIVIR 300 mg Once Daily plus RETROVIR plus Efavirenz (n = 278) |
EPIVIR 150 mg Twice Daily plus RETROVIR plus Efavirenz (n = 276) |
|
Respondera |
67% |
65% |
|
Virologic failureb |
8% |
8% |
|
Discontinued due to clinical progression |
<1% |
0% |
|
Discontinued due to adverse events |
6% |
12% |
|
Discontinued due to other reasonsc |
18% |
14% |
| a Achieved confirmed plasma HIV-1 RNA <400 copies/mL and maintained through 48 weeks. | ||
| b Achieved suppression but rebounded by Week 48, discontinued due to virologic failure, insufficient viral response according to the investigator, or never suppressed through Week 48. | ||
| c Includes consent withdrawn, lost to followup, protocol violation, data outside the study-defined schedule, and randomized but never initiated treatment. |
The proportions of patients with HIV-1 RNA <50 copies/mL (via Roche Ultrasensitive assay) through Week 48 were 61% for patients receiving EPIVIR 300 mg once daily and 63% for patients receiving EPIVIR 150 mg twice daily. Median increases in CD4+ cell counts were 144 cells/mm3 at Week 48 in patients receiving EPIVIR 300 mg once daily and 146 cells/mm3 for patients receiving EPIVIR 150 mg twice daily.
A small, randomized, open-label pilot study, EPV40001, was conducted in Thailand. A total of 159 treatment-naive adult patients (male 32%, Asian 100%, median age 30 years, baseline median CD4+ cell count 380 cells/mm3, median plasma HIV-1 RNA 4.8 log10 copies/mL) were enrolled. Two of the treatment arms in this study provided a comparison between lamivudine 300 mg once daily (n = 54) and lamivudine 150 mg twice daily (n = 52), each in combination with zidovudine 300 mg twice daily and abacavir 300 mg twice daily. In intent-to-treat analyses of 48-week data, the proportions of patients with HIV-1 RNA below 400 copies/mL were 61% (33/54) in the group randomized to once-daily lamivudine and 75% (39/52) in the group randomized to receive all 3 drugs twice daily; the proportions with HIV-1 RNA below 50 copies/mL were 54% (29/54) in the once-daily lamivudine group and 67% (35/52) in the all-twice-daily group; and the median increases in CD4+ cell counts were 166 cells/mm3 in the once-daily lamivudine group and 216 cells/mm3 in the all-twice-daily group.
Clinical Endpoint Study: ACTG300 was a multi-center, randomized, double-blind study that provided for comparison of EPIVIR plus RETROVIR (zidovudine) with didanosine monotherapy. A total of 471 symptomatic, HIV-1-infected therapy-naive (≤56 days of antiretroviral therapy) pediatric patients were enrolled in these 2 treatment arms. The median age was 2.7 years (range: 6 weeks to 14 years), 58% were female, and 86% were non-Caucasian. The mean baseline CD4+ cell count was 868 cells/mm3 (mean: 1,060 cells/mm3 and range: 0 to 4,650 cells/mm3 for patients ≤5 years of age; mean: 419 cells/mm3 and range: 0 to 1,555 cells/mm3 for patients >5 years of age) and the mean baseline plasma HIV-1 RNA was 5.0 log10 copies/mL. The median duration on study was 10.1 months for the patients receiving EPIVIR plus RETROVIR and 9.2 months for patients receiving didanosine monotherapy. Results are summarized in Table 10.
|
Endpoint |
EPIVIR plus RETROVIR (n = 236) |
Didanosine (n = 235) |
|
HIV disease progression or death (total) |
15 (6.4%) |
37 (15.7%) |
|
Physical growth failure |
7 (3.0%) |
6 (2.6%) |
|
Central nervous system deterioration |
4 (1.7%) |
12 (5.1%) |
|
CDC Clinical Category C |
2 (0.8%) |
8 (3.4%) |
|
Death |
2 (0.8%) |
11 (4.7%) |
HOW SUPPLIED
EPIVIR Scored Tablets, 150 mg
White, diamond-shaped, scored, film-coated tablets debossed with “GX CJ7” on both sides.
Bottle of 60 tablets (NDC 0173-0470-01) with child-resistant closure.
EPIVIR Tablets, 300 mg
Gray, modified diamond-shaped, film-coated tablets engraved with “GX EJ7” on one side and plain on the reverse side.
Bottle of 30 tablets (NDC 0173-0714-00) with child-resistant closure.
Recommended Storage:
Store EPIVIR Tablets at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
EPIVIR Oral Solution, 10 mg/mL
A clear, colorless to pale yellow, strawberry-banana-flavored liquid, contains 10 mg of lamivudine in each 1 mL.
Plastic bottle of 240 mL (NDC 0173-0471-00) with child-resistant closure. This product does not require reconstitution.
Recommended Storage:
Store in tightly closed bottles at 25°C (77°F) [see USP Controlled Room Temperature].
INFORMATION FOR PATIENTS
Information About Therapy With EPIVIR: EPIVIR is not a cure for HIV-1 infection and patients may continue to experience illnesses associated with HIV-1 infection, including opportunistic infections. Patients should remain under the care of a physician when using EPIVIR. Patients should be advised that the use of EPIVIR has not been shown to reduce the risk of transmission of HIV-1 to others through sexual contact or blood contamination.
Patients should be advised that the long-term effects of EPIVIR are unknown at this time.
Patients should be advised of the importance of taking EPIVIR with combination therapy on a regular dosing schedule and to avoid missing doses.
EPIVIR should not be coadministered with drugs containing lamivudine or emtricitabine, including COMBIVIR (lamivudine/zidovudine) Tablets, EPZICOM (abacavir sulfate and lamivudine) Tablets, TRIZIVIR (abacavir sulfate, lamivudine, and zidovudine), ATRIPLA (efavirenz, emtricitabine, and tenofovir), EMTRIVA (emtricitabine) or TRUVADA (emtricitabine and tenofovir) [see Warnings and Precautions (5.3)].
Redistribution/Accumulation of Body Fat: Patients should be informed that redistribution or accumulation of body fat may occur in patients receiving antiretroviral therapy, including EPIVIR, and that the cause and long-term health effects of these conditions are not known at this time [see Warnings and Precautions (5.7)].
Differences in Formulations of EPIVIR: Patients should be advised that EPIVIR Tablets and Oral Solution contain a higher dose of the same active ingredient (lamivudine) as EPIVIR-HBV Tablets and Oral Solution. If a decision is made to include lamivudine in the HIV-1 treatment regimen of a patient co-infected with HIV-1 and HBV, the formulation and dosage of lamivudine in EPIVIR (not EPIVIR-HBV) should be used [see Warnings and Precautions (5.2)].
Co-infection With HIV-1 and HBV: Patients co-infected with HIV-1 and HBV should be informed that deterioration of liver disease has occurred in some cases when treatment with lamivudine was discontinued. Patients should be advised to discuss any changes in regimen with their physician [see Warnings and Precautions (5.2)].
Risk of Pancreatitis: Parents or guardians should be advised to monitor pediatric patients for signs and symptoms of pancreatitis [see Warnings and Precautions (5.5)].
Sucrose Content of EPIVIR Oral Solution: Diabetic patients should be advised that each 15-mL dose of EPIVIR Oral Solution contains 3 grams of sucrose [see Description (11)].
COMBIVIR, EPIVIR, EPIVIR-HBV, EPZICOM, and TRIZIVIR are registered trademarks of GlaxoSmithKline. ATRIPLA, EMTRIVA, and TRUVADA are trademarks of their respective owners and are not trademarks of GlaxoSmithKline. The makers of these brands are not affiliated with and do not endorse GlaxoSmithKline or its products.
GlaxoSmithKline
Research Triangle Park, NC 27709
Manufactured under agreement from
Shire Pharmaceuticals Group plc
Basingstoke, UK
©2009, GlaxoSmithKline. All rights reserved.
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION


EPIVIRlamivudine TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||