Erborian BB Creme Au Ginseng Dore
Kolmar Korea Co Ltd
Kolmar Korea Co Ltd
Erborian BB Crème Au Ginseng Doré
FULL PRESCRIBING INFORMATION: CONTENTS*
FULL PRESCRIBING INFORMATION
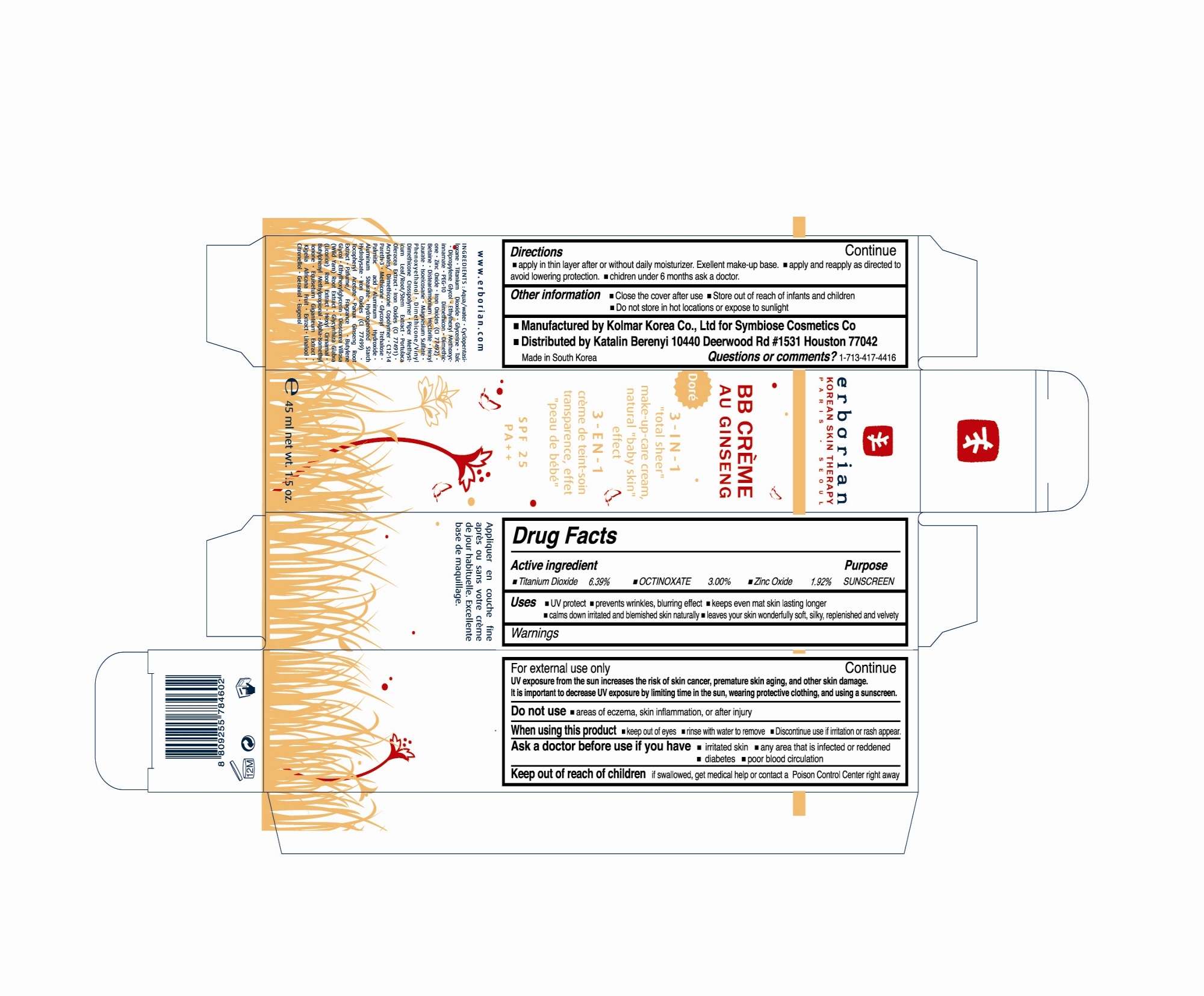
Drug Facts
Active Ingredient
Titanium Dioxide 6.39%
Octinoxate 3.00%
Zinc Oxide 1.92%
Uses
Helps prevent sunburn. Provides High protect from sunburn. Higher SPF gives more sunburn protection.
Directions
Apply after basic skin care product. Spread and appropriate amount entire face following the skin texture. Reapply as required.
Warnings
For external use only.
UV exposure from the sun increases the risk of skin cancer, premature skin aging, and other skin damage. It is important to decrease UV exposure by limiting time in the sun, wearing protective clothing, and using a sunscreen.
When using the product Keep out of eyes. Rinse with water to remove.
Keep out of reach of children if swallowed, get medical help or contact a Poison Control Center
Erborian BB Crème Au Ginseng Doré
SYMBIOSE COSMETICS
Net WT. 1.5 OZ (45mL)
Erborian BB Creme Au Ginseng DoreTitanium Dioxide CREAM
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||