Felodipine
FULL PRESCRIBING INFORMATION: CONTENTS*
- FELODIPINE DESCRIPTION
- CLINICAL PHARMACOLOGY
- PHARMACOKINETICS
- INDICATIONS & USAGE
- FELODIPINE CONTRAINDICATIONS
- PRECAUTIONS
- INFORMATION FOR PATIENTS
- DRUG INTERACTIONS
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- PREGNANCY
- NURSING MOTHERS
- PEDIATRIC USE
- GERIATRIC USE
- FELODIPINE ADVERSE REACTIONS
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
FELODIPINE DESCRIPTION
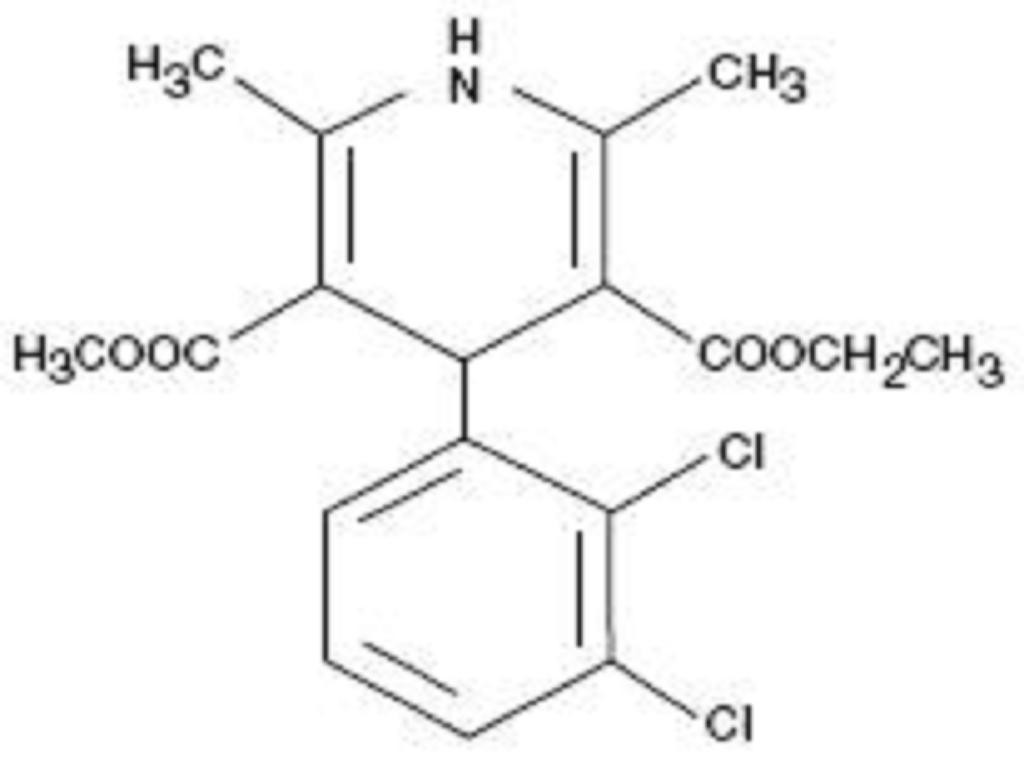
CLINICAL PHARMACOLOGY
Mechanism of ActionCardiovascular Effects
PHARMACOKINETICS
Cardiovascular EffectsDOSAGE AND ADMINISTRATION
Geriatric Use
Hepatic Dysfunction
Cardiovascular Effects
PRECAUTIONS
Renal/Endocrine Effects
Clinical Studies
*
DoseNSystolic/DiastolicMean PeakMean Trough Trough/PeakResponseResponseRatios (%s)*
INDICATIONS & USAGE
FELODIPINE CONTRAINDICATIONS
PRECAUTIONS
GeneralHypotension
ADVERSE REACTIONS
Heart Failure
Patients with Impaired Liver Function
CLINICAL PHARMACOLOGYDOSAGE AND ADMINISTRATION
Peripheral Edema
INFORMATION FOR PATIENTS
DRUG INTERACTIONS
CYP3A4 InhibitorsItraconazole
Erythromycin
Grapefruit juice
Cimetidine
Beta-Blocking Agents
Digoxin
Anticonvulsants
Tacrolimus
Other Concomitant Therapy
Interaction with Food
CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
111
1
1
PREGNANCY
Category C.Teratogenic Effects
1
Nonteratogenic Effects
1
1
NURSING MOTHERS
PEDIATRIC USE
GERIATRIC USE
CLINICAL PHARMACOLOGY, Geriatric UseFELODIPINE ADVERSE REACTIONS
DOSAGE AND ADMINISTRATION
*
Body SystemPlacebo2.5 mg5 mg10 mgAdverse EventsN=334N=255N=581N=408Body as a WholeCardiovascularDigestiveNervousRespiratorySkin*
Gingival Hyperplasia
PRECAUTIONS, Information for Patients
Clinical Laboratory Test Findings
Serum Electrolytes
CLINICAL PHARMACOLOGY, Renal/Endocrine Effects
Serum Glucose
Liver Enzymes
OVERDOSAGE
DOSAGE & ADMINISTRATION
ADVERSE REACTIONSCLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism
Geriatric Use
CLINICAL PHARMACOLOGY
Patients with Impaired Liver Function
CLINICAL PHARMACOLOGY
HOW SUPPLIED
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION


FelodipineFelodipine TABLET, EXTENDED RELEASE
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!