FIRST Mouthwash BLM
FIRST - Mouthwash BLM
FULL PRESCRIBING INFORMATION
F IRST ® - Mouthwash BLM Rx
Diphenhydramine HCl , Lidocaine HCl , Aluminum Hydroxide, Magnesium Hydroxide, and Simethicone Compounding Kit
FOR PRESCRIPTION COMPOUNDING ONLY
Each FIRST®— Mouthwash BLM Compounding Kit is comprised of 0.2 grams of diphenhydramine hydrochloride powder USP and 1.6 grams of lidocaine hydrochloride powder USP for oral use.* FIRST®— Mouthwash BLM Compounding Kit also contains a 236 mL suspension containing 3.15 grams of aluminum hydroxide USP (equivalent to dried gel USP), 3.15 grams of magnesium hydroxide USP, and 0.315 grams of simethicone USP with benzyl alcohol, butylparaben, flavor, hydroxyethylcellulose, propylparaben, purified water, saccharin sodium, sorbitol solution, D&C red #28, and FD&C red #40.* When compounded, the final product provides an homogeneous suspension containing diphenhydramine hydrochloride, lidocaine hydrochloride, and aluminum hydroxide, magnesium hydroxide, and simethicone comparable to the active ingredients (Benadryl®, Lidocaine HCl 2% Viscous, Maalox® 1:1:1) contained in Magic Mouthwash.**
| Size | 8 FL OZ (237 mL) |
| NDC# | 65628-050-01 |
| Diphenhydramine HCl | 0.2g |
| Lidocaine HCl | 1.6g |
| FIRST®-Mouthwash Suspension | 236 mL |
Everything you need to make this is included…
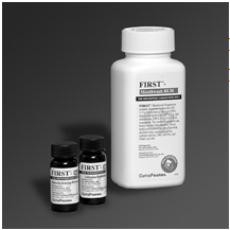
1. FIRST®— Mouthwash BLM Compounding Kit contains premeasured diphenhydramine hydrochloride powder, lidocaine hydrochloride powder and mouthwash suspension (aluminum hydroxide, magnesium hydroxide, simethicone plus inactive ingredients)

2. Important - Before dispensing, tap the top and bottom of the bottle containing diphenhydramine hydrochloride to loosen the powder and remove the cap. Empty the diphenhydramine hydrochloride powder into the bottle containing the mouthwash liquid suspension. Likewise, tap the top and bottom of the bottle containing lidocaine hydrochloride to loosen the powder and remove the cap. Empty the lidocaine hydrochloride powder into the bottle containing the mouthwash liquid suspension.
The appropriate quantities of diphenhydramine hydrochloride powder and lidocaine hydrochloride powder have been packaged in each bottle to deliver the required dosage of each drug. Residual quantities remaining in the bottles after emptying need not be rinsed out.

3. Close the bottle and shake for 20 to 30 seconds. Instruct the patient to shake bottle well before each use.
Prior to compounding, store FIRST®— Mouthwash BLM Compounding Kit at room temperature between 15°-30°C (59°-86°F) [see USP]. Also store final formulation at room temperature, 15°-30°C (59°-86°F).
FIRST®— Mouthwash BLM Compounding Kit components have a three-year expiration date.*** Based on real time controlled room temperature and humidity testing, compounded FIRST®— Mouthwash BLM Compounding Kit is stable for at least six months.***
FIRST®— Mouthwash suspension meets the requirements for total aerobic microbial count of not more than 100 cfu/mL, as well as for the absence of the specified microorganisms Escherichia coli, Pseudomonas aeruginosa , Staphylococcus aureus , and Salmonella spp. when tested as described in the current USP under <61> Microbial Enumeration Tests and <62> Tests for Specified Microorganisms. FIRST®— Mouthwash suspension also meets the requirements as described in the current USP under <51> Antimicrobial Effectiveness Testing for Category 4 products.
For oral use only. Avoid contact with eyes. Keep container tightly closed. Keep out of the reach of children. Protect from light. Protect from freezing. Compounded product, as dispensed, is stable for at least 180 days at room temperature.
* Certificate of analysis on file
** This product is not manufactured by Pfizer, Inc., manufacturer of Benadryl® or by Novartis Consumer Health, Inc. manufacturer of Maalox®
*** Data and documentation on file
RX ONLY
Revised: February 2010
U.S. Patent No. 6,708,822 B1
Additional U.S. Patent Pending
Distributed By:
CutisPharma, Inc.
SMART PRODUCTS FOR SMART PEOPLE®
Woburn, MA 01801, USA www.cutispharma.com
NDC 65628-050-01
FIRST® Mouthwash BLM
Each kit contains:
Active Ingredients:
Diphenhydramine hydrochloride 0.2g
Lidocaine hydrochloride 1.6g
Aluminum hydroxide 3.15g in 236mL suspension (equivalent to dried gel USP)
Magnesium hydroxide 3.15g in 236mL suspension
Simethicone 0.315g in 236mL suspension
8 FL OZ (237 mL) as dispensed

FIRST Mouthwash BLMDiphenhydramine Hydrochloride and Lidocaine Hydrochloride and Aluminum Hydroxide and Magnesium Hydro KIT
| ||||||||||||||||||||||||||||||||||||||||