Fluconazole
FULL PRESCRIBING INFORMATION: CONTENTS*
- FLUCONAZOLE DESCRIPTION
- CLINICAL PHARMACOLOGY
- INDICATIONS & USAGE
- CLINICAL STUDIES
- FLUCONAZOLE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- DRUG INTERACTIONS
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- PREGNANCY
- NURSING MOTHERS
- PEDIATRIC USE
- GERIATRIC USE
- FLUCONAZOLE ADVERSE REACTIONS
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- STORAGE AND HANDLING
- REFERENCES
- INFORMATION FOR PATIENTS
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
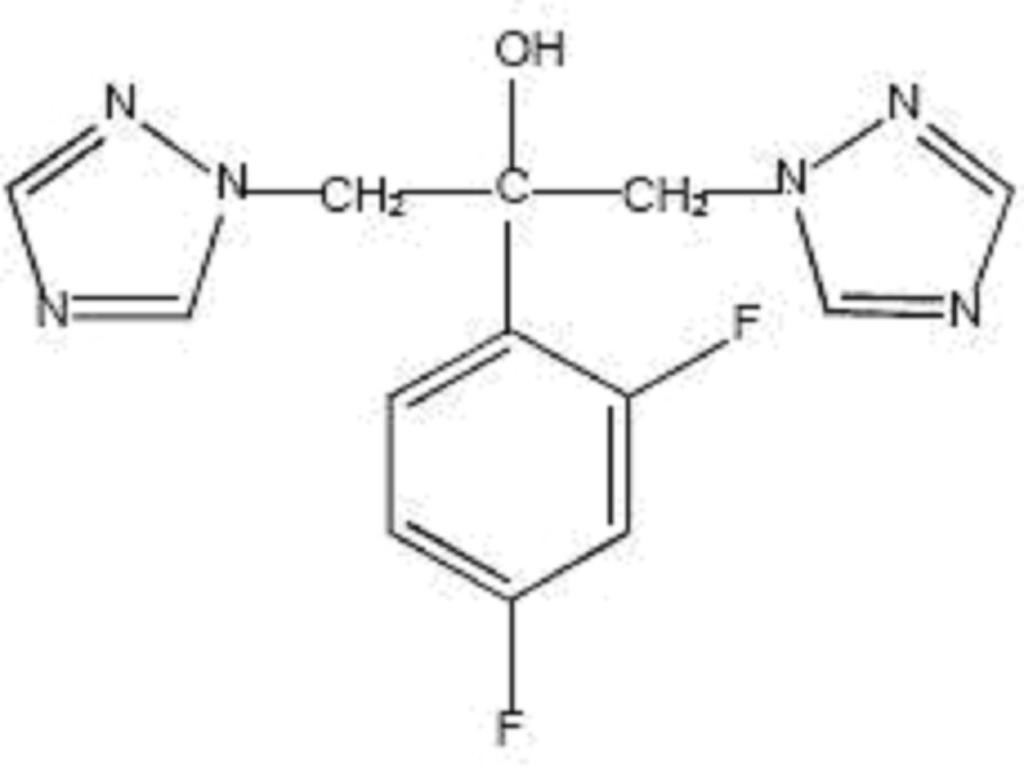
FLUCONAZOLE DESCRIPTION
CLINICAL PHARMACOLOGY
Pharmacokinetics and Metabolism**
Pharmacokinetics in Children
Pharmacokinetics in Elderly
Drug Interaction Studies
Oral contraceptives
Cimetidine
Antacid
Hydrochlorothiazide
Rifampin
Warfarin
Phenytoin
Cyclosporine
Zidovudine
Theophylline
Terfenadine
Oral Hypoglycemics
Rifabutin
Tacrolimus
Cisapride
Midazolam
Azithromycin
Microbiology
Mechanism of Action
Activity In Vitro and In Clinical Infections
Susceptibility Testing Methods
Table 1: Susceptibility Interpretive Criteria for Fluconazole
Quality Control
Table 2: Acceptable Quality Control Ranges for Fluconazole to be Used in Validation of Susceptibility Test Results
Activity In Vivo
Drug Resistance
INDICATIONS & USAGE
Prophylaxis
CLINICAL STUDIES
Cryptococcal MeningitisVaginal Candidiasis
Pediatric Studies
Oropharyngeal Candidiasis
**
FLUCONAZOLE CONTRAINDICATIONS
WARNINGS
1. Hepatic InjuryFluconazole has been associated with rare cases of serious hepatic toxicity, including fatalities primarily in patients with serious underlying medical conditions. In cases of fluconazole-associated hepatotoxicity, no obvious relationship to total daily dose, duration of therapy, sex or age of the patient has been observed.
Fluconazole hepatotoxicity has usually, but not always, been reversible on discontinuation of therapy. Patients who develop abnormal liver function tests during fluconazole therapy should be monitored for the development of more severe hepatic injury. Fluconazole should be discontinued if clinical signs and symptoms consistent with liver disease develop that may be attributable to fluconazole.
2. Anaphylaxis
3. Dermatologic
PRECAUTIONS
GeneralSingle Dose
DRUG INTERACTIONS
Oral Hypoglycemics
Coumarin-Type Anticoagulants
Phenytoin
Cyclosporine
Rifampin
Theophylline
Terfenadine
Cisapride
Astemizole
Rifabutin
Tacrolimus
Short-acting Benzodiazepines
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
PREGNANCY
Teratogenic EffectsPregnancy Category C
NURSING MOTHERS
PEDIATRIC USE
GERIATRIC USE
FLUCONAZOLE ADVERSE REACTIONS
In Patients Receiving a Single Dose for Vaginal CandidiasisIn Patients Receiving Multiple Doses for Other Infections
Hepatobiliary
Postmarketing Experience
Immunologic
Cardiovascular
Central Nervous System
Dermatologic
Hematopoietic and Lymphatic
Metabolic
Other Senses
Adverse Reactions in Children
OVERDOSAGE
OVERDOSAGE
DOSAGE & ADMINISTRATION
Dosage and Administration in AdultsSingle Dose
Multiple Dose
Dosage and Administration in Children
**
Oropharyngeal candidiasis
Esophageal candidiasis
Systemic candida infections
Cryptococcal meningitis
Dosage in Patients With Impaired Renal Function
HOW SUPPLIED
STORAGE AND HANDLING
REFERENCES
INFORMATION FOR PATIENTS
Fluconazole TabletsWhat is Fluconazole?
What is a Vaginal Yeast Infection?
-
● itching
-
● a burning feeling when you urinate
-
● redness
-
● soreness
-
● a thick white vaginal discharge that looks like cottage cheese
-
● diabetes medicines you take by mouth such as glyburide, tolbutamide, glipizide
-
● blood thinners such as warfarin
-
● cyclosporine (used to prevent rejection of organ transplants)
-
● rifampin or rifabutin (used for tuberculosis)
-
● astemizole (used for allergies)
-
● tacrolimus (used to prevent rejection of organ transplants)
-
● phenytoin (used for seizures)
-
● theophylline (used for asthma)
-
● cisapride (Propulsid
-
● terfenadine (Seldane
-
● are taking any over-the-counter medicines you can buy without a prescription, including natural or herbal remedies
-
● have any liver problems.
-
● have any other medical conditions
-
● are pregnant, plan to become pregnant, or think you might be pregnant. Your doctor will discuss whether fluconazole is right for you.
-
● are breast-feeding. Fluconazole can pass through breast milk to the baby.
-
● are allergic to any other medicines including those used to treat yeast and other fungal infections.
-
● are allergic to any of the ingredients in fluconazole. If you need to know the inactive ingredients, ask your doctor or pharmacist.
How Should I Take Fluconazole?
Just swallow 1 fluconazole tablet to treat your vaginal yeast infection.
What Should I Avoid While Taking Fluconazole?
What Are The Possible Side Effects of Fluconazole?
-
● headache
-
● diarrhea
-
● nausea or upset stomach
-
● dizziness
-
● stomach pain
-
● changes in the way food tastes
What To Do For An Overdose
How To Store Fluconazole
General Advice About Prescription Medicines
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION


FluconazoleFluconazole TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!