Fludeoxyglucose
The University of Texas MD Anderson Cancer Center
University of Texas MD Anderson Cancer Center
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use Fludeoxyglucose F18 Injection safely and effectively. See full prescribing information for Fludeoxyglucose F18 Injection. Fludeoxyglucose F18 InjectionInitial U.S. Approval: 2005RECENT MAJOR CHANGESWarnings and Precautions: (5.1,5.2) 7/2010Adverse Reactions (6)7/2010DOSAGE FORMS AND STRENGTHSMultiple-dose vial containing: 0.74 - 11.1 GBq/mL (20 - 300 mCi/mL) of Fludeoxyglucose F18 Injection and a 4.5 mg of sodium chloride in citrate buffer (approximately 27 mL volume), for intravenous administration or 0.74 - 5.55 GBq/mL (20 - 150 mCi/mL) of Fludeoxyglucose F18 Injection and a 4.5 mg of sodium chloride in phosphate buffer (approximately 15 mL volume), for intravenous administration Side EffectsHypersensitivity reactions have occurred; have emergency resuscitation equipment and personnel immediately available (6). To report SUSPECTED ADVERSE REACTIONS, contact MDACC CRF at 713-563-5455 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
FULL PRESCRIBING INFORMATION: CONTENTS*
- 3 DOSAGE FORMS AND STRENGTHS
- 6 FLUDEOXYGLUCOSE ADVERSE REACTIONS
- Final Product Labels
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- 11.0 FLUDEOXYGLUCOSE DESCRIPTION
FULL PRESCRIBING INFORMATION
3 DOSAGE FORMS AND STRENGTHS
Multiple-dose vial containing:
- 0.74 - 11.1 GBq/mL (20 - 300 mCi/mL) of Fludeoxyglucose F18 Injection and a 4.5 mg of sodium chloride in citrate buffer (approximately 27 mL volume), for intravenous administration
or
- 0.74 - 5.55 GBq/mL (20 - 150 mCi/mL) of Fludeoxyglucose F18 Injection and a 4.5 mg of sodium chloride in phosphate buffer (approximately 15 mL volume), for intravenous administration
6 ADVERSE REACTIONS
Hypersensitivity reactions with pruritus, edema and rash have been reported in the post-marketing setting. Have emergency resuscitation equipment and personnel immediately available.
Manufactured and Distributed by:
The University of Texas MD Anderson Cancer Center
Center for Advanced Biomedical Imaging Research
Cyclotron Radiochemistry Facility
1881 East Road
Houston, TX 77054
Final Product Labels

FDG Citrate Formulation

FDG Phosphate Formulation
16 HOW SUPPLIED/STORAGE AND HANDLING
Fludeoxyglucose F 18 Injection is supplied in a multi-dose, 30 mL glass vial containing:
- between 0.740 - 11.1 GBq/mL (20 - 300 mCi/mL), of no carrier added 2-deoxy-2-[F 18] fluoro-D-glucose, at end of synthesis, in approximately 27 mL. Citrate Buffer Formulation: NDC 60215-411-30
or
- between 0.740 - 5.55 GBq/mL (20 - 150 mCi/mL), of no carrier added 2-deoxy-2-[F 18] fluoro-D-glucose, at end of synthesis, in approximately 15 mL. Phosphate Buffer Formulation: NDC 60215-411-15
The total volume and total radioactivity per vial are variable. The contents of each vial are sterile, pyrogen-free and preservative free. Each vial is enclosed in a shielding container of appropriate thickness.
Storage
Store the Fludeoxyglucose F 18 Injection vial upright in a shielded container at 10 - 25°C (68 - 77°F), excursions permitted to 15 - 30°C (59 - 86°F) [See USP Controlled Room Temperature].
Handling
Receipt, transfer, handling, possession, or use of this product is subject to the radioactive material regulations and licensing requirements of the U.S. Nuclear Regulatory Commission, Agreement States or Licensing States as appropriate.
11.0 DESCRIPTION
11.1 Chemical Characteristics
Fludeoxyglucose F 18 Injection is a positron emitting radiopharmaceutical that is used for diagnostic purposes in conjunction with positron emission tomography (PET) imaging. The active ingredient 2-deoxy-2-[18F]fluoro-D-glucose has the molecular formula of C6H11 18FO5 with a molecular weight of 181.26, and has the following chemical structure:
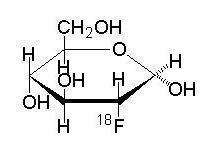
Fludeoxyglucose F 18 Injection is provided as a ready to use sterile, pyrogen free, clear, colorless solution. Each mL contains between 0.740 to 11.1 GBq (20.0 - 300 mCi) of 2-deoxy-2-[18F]fluoro-D-glucose at the EOS, 4.5 mg of sodium chloride in citrate buffer or 0.740 to 5.55 GBq (20.0 – 150 mCi) of 2-deoxy-2-[18F]fluoro-D-glucose at the EOS, 4.5 mg of sodium chloride in phosphate buffer. The pH of the solution is between 4.5 and 7.5. The solution is packaged in a multiple-dose glass vial and does not contain any preservative.
11.2 Physical Characteristics
Fluorine F 18 has a physical half-life of 109.7 minutes and decays to Oxygen O 18 (stable) by positron decay. The principal photons useful for imaging are the dual 511 keV “annihilation” gamma photons that are produced and emitted simultaneously in opposite directions when the positron interacts with an electron (Table 2).
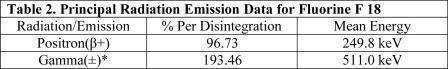
*Produced by positron annihilation
From: Kocher, D.C. Radioactive Decay Tables DOE/TIC-I 1026, 89 (1981)
The specific gamma ray constant (point source air kerma coefficient) for fluorine F 18 is 5.7 R/hr/mCi (1.35 x 10 -6 Gy/hr/kBq) at 1 cm. The half-value layer (HVL) for the 511keV photons is 4 mm lead (Pb). The range of attenuation coefficients for this radionuclide as a function of lead shield thickness is shown in Table 3. For example, the interposition of an 8 mm thickness of Pb, with a coefficient of attenuation of 0.25, will decrease the external radiation by 75%.
_43954e40.jpg)
For use in correcting for physical decay of this radionuclide, the fractions remaining at selected intervals after calibration are shown in Table 4.
_43954e40.jpg)
*Calibration Time
FludeoxyglucoseFludeoxyglucose F18 INJECTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||