FLUZONE QUADRIVALENT
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use Fluzone Quadrivalent safely and effectively. See full prescribing information for Fluzone Quadrivalent. Fluzone Quadrivalent (Influenza Virus Vaccine)Suspension for Intramuscular Injection2013-2014 FormulaInitial U.S. Approval (Fluzone Quadrivalent): 2013INDICATIONS AND USAGE Fluzone Quadrivalent is a vaccine indicated for active immunization for the prevention of influenza disease caused by influenza A subtype viruses and type B viruses contained in the vaccine. (1) Fluzone Quadrivalent is approved for use in persons 6 months of age and older. (1)DOSAGE AND ADMINISTRATION For intramuscular use only (2) Age Dose Schedule "-" Indicates information is not applicable 6 months through 35 months One or two doses 1 or 2 doses depends on vaccination history as per Advisory Committee on Immunization Practices annual recommendations on prevention and control of influenza with vaccines, 0.25 mL each If 2 doses, administer at least 4 weeks apart 36 months through 8 years One or two doses , 0.5 mL each If 2 doses, administer at least 4 weeks apart 9 years and older One dose, 0.5 mL - DOSAGE FORMS AND STRENGTHSSuspension for injection supplied in 3 presentations: prefilled single-dose syringe (yellow plunger rod), 0.25 mL; prefilled single-dose syringe (purple plunger rod), 0.5 mL; single-dose vial, 0.5 mL. (3)CONTRAINDICATIONSSevere allergic reaction (e.g., anaphylaxis) to any component of the vaccine, including egg protein, or after previous dose of any influenza vaccine. (4)WARNINGS AND PRECAUTIONS If Guillain-Barré syndrome (GBS) has occurred within 6 weeks of previous influenza vaccination, the decision to give Fluzone Quadrivalent should be based on careful consideration of the potential benefits and risks. (5.1) Side Effects In children 6 months through 35 months of age, the most common (≥10%) injection-site reactions were pain (57%) or tenderness (54%), erythema (37%), and swelling (22%); the most common solicited systemic adverse reactions were irritability (54%), abnormal crying (41%), malaise (38%), drowsiness (38%), appetite loss (32%), myalgia (27%), vomiting (15%), and fever (14%). (6.1) In children 3 years through 8 years of age, the most common (≥10%) injection-site reactions were pain (67%), erythema (34%), and swelling (25%); the most common solicited systemic adverse reactions were myalgia (39%), malaise (32%), and headache (23%). (6.1) In adults 18 years and older, the most common (≥10%) injection-site reaction was pain (47%); the most common solicited systemic adverse reactions were myalgia (24%), headache (16%), and malaise (11%). (6.1) In adults 65 years of age and older, the most common (≥10%) injection-site reaction was pain (33%); the most common solicited systemic adverse reactions were myalgia (18%), headache (13%), and malaise (11%). (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Sanofi Pasteur Inc., at 1-800-822-2463 (1-800-VACCINE) or VAERS at 1-800-822-7967 or www.vaers.hhs.gov.USE IN SPECIFIC POPULATIONS Safety and effectiveness of Fluzone Quadrivalent have not been established in pregnant women or children less than 6 months of age. (8.1) (8.4) Pregnancy: Pregnancy registry available. Call Sanofi Pasteur Inc. at 1-800-822-2463. (8.1) Antibody responses to Fluzone Quadrivalent are lower in persons ≥65 years of age than in younger adults. (8.5)
FULL PRESCRIBING INFORMATION: CONTENTS*
- 1 FLUZONE QUADRIVALENT INDICATIONS AND USAGE
- 2 FLUZONE QUADRIVALENT DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORMS AND STRENGTHS
- 4 FLUZONE QUADRIVALENT CONTRAINDICATIONS
- 5 WARNINGS AND PRECAUTIONS
- 6 FLUZONE QUADRIVALENT ADVERSE REACTIONS
- 8 USE IN SPECIFIC POPULATIONS
- 11 FLUZONE QUADRIVALENT DESCRIPTION
- 12 CLINICAL PHARMACOLOGY
- 14 CLINICAL STUDIES
- 15 REFERENCES
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Fluzone® Quadrivalent is an inactivated quadrivalent influenza virus vaccine indicated for the prevention of influenza disease caused by influenza A subtype viruses and type B viruses contained in the vaccine.
Fluzone Quadrivalent is approved for use in persons 6 months of age and older.
2 DOSAGE AND ADMINISTRATION
- For intramuscular use only
2.1 Dose and Schedule
The dose and schedule for Fluzone Quadrivalent are presented in Table 1.
| Age | Dose | Schedule |
|---|---|---|
| "-" Indicates information is not applicable | ||
| 6 months through 35 months | One or two doses |
If 2 doses, administer at least 4 weeks apart |
| 36 months through 8 years | One or two doses |
If 2 doses, administer at least 4 weeks apart |
| 9 years and older | One dose, 0.5 mL | - |
2.2 Administration
Inspect Fluzone Quadrivalent visually for particulate matter and/or discoloration prior to administration. If any of these defects or conditions exist, the vaccine should not be administered.
Before administering a dose of vaccine, shake the prefilled syringe or single-dose vial. Withdraw the vaccine using a sterile needle and syringe.
The preferred sites for intramuscular injection are the anterolateral aspect of the thigh in infants 6 months through 11 months of age, the anterolateral aspect of the thigh (or the deltoid muscle if muscle mass is adequate) in persons 12 months through 35 months of age, or the deltoid muscle in persons ≥36 months of age. The vaccine should not be injected into the gluteal area or areas where there may be a major nerve trunk.
Do not administer this product intravenously, intradermally, or subcutaneously.
Fluzone Quadrivalent vaccine should not be combined through reconstitution or mixed with any other vaccine.
3 DOSAGE FORMS AND STRENGTHS
Fluzone Quadrivalent is a suspension for injection.
Fluzone Quadrivalent is supplied in 3 presentations:
1) Prefilled single-dose syringe (yellow syringe plunger rod), 0.25 mL, for persons 6 months through 35 months of age.
2) Prefilled single-dose syringe (purple syringe plunger rod), 0.5 mL, for persons 36 months of age and older.
3) Single-dose vial, 0.5 mL, for persons 36 months of age and older.
4 CONTRAINDICATIONS
Do not administer Fluzone Quadrivalent to anyone with a history of a severe allergic reaction (e.g., anaphylaxis) to any component of the vaccine [see Description (11) ], including egg protein, or to a previous dose of any influenza vaccine.
5 WARNINGS AND PRECAUTIONS
5.1 Guillain-Barré Syndrome
The 1976 swine influenza vaccine was associated with an elevated risk of Guillain-Barré syndrome (GBS). Evidence for a causal relation of GBS with other influenza vaccines is inconclusive; if an excess risk exists, it is probably slightly more than 1 additional case per 1 million persons vaccinated. (see ref. 1) If GBS has occurred within 6 weeks of previous influenza vaccination, the decision to give Fluzone Quadrivalent should be based on careful consideration of the potential benefits and risks.
5.2 Preventing and Managing Allergic Reactions
Appropriate medical treatment and supervision must be available to manage possible anaphylactic reactions following administration of Fluzone Quadrivalent.
5.3 Altered Immunocompetence
If Fluzone Quadrivalent is administered to immunocompromised persons, including those receiving immunosuppressive therapy, the expected immune response may not be obtained.
5.4 Limitations of Vaccine Effectiveness
Vaccination with Fluzone Quadrivalent may not protect all recipients.
6 ADVERSE REACTIONS
In children 6 months through 35 months of age, the most common (≥10%) injection-site reactions were pain (57%)








6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse event rates observed in the clinical trials of a vaccine cannot be directly compared to rates in the clinical trial of another vaccine, and may not reflect the rates observed in practice.
Children 6 Months Through 8 Years of Age
Study 1 (NCT01240746, see http://clinicaltrials.gov) was a single-blind, randomized, active-controlled multi-center safety and immunogenicity study conducted in the US. In this study, children 6 months through 35 months of age received one or two 0.25 mL doses of either Fluzone Quadrivalent or one of two formulations of a comparator trivalent influenza vaccine (TIV-1 or TIV-2), and children 3 years through 8 years of age received one or two 0.5 mL doses of either Fluzone Quadrivalent, TIV-1, or TIV-2. Each of the trivalent formulations contained an influenza type B virus that corresponded to one of the two type B viruses in Fluzone Quadrivalent (a type B virus of the Victoria lineage or a type B virus of the Yamagata lineage). For participants who received two doses, the doses were administered approximately 4 weeks apart. The safety analysis set included 1841 children 6 months through 35 months of age and 2506 children 3 years through 8 years of age. Among participants 6 months through 8 years of age in the three vaccine groups combined, 49.3% were female (Fluzone Quadrivalent, 49.2%; TIV-1, 49.8%; TIV-2, 49.4%), 58.4% Caucasian (Fluzone Quadrivalent, 58.4%; TIV-1, 58.9%; TIV-2, 57.8%), 20.2% Black (Fluzone Quadrivalent, 20.5%; TIV-1, 19.9%; TIV-2, 19.1%), 14.1% Hispanic (Fluzone Quadrivalent, 14.3%; TIV-1, 13.2%; TIV-2, 14.7%), and 7.3% were of other racial/ethnic groups (Fluzone Quadrivalent, 6.8%; TIV-1, 8.0%; TIV-2, 8.5%). Table 2 and Table 3 summarize solicited injection-site and systemic adverse reactions reported within 7 days post-vaccination via diary cards. Participants were monitored for unsolicited adverse events for 28 days after each dose and serious adverse events (SAEs) during the 6 months following the last dose.
| Fluzone Quadrivalent (N  |
TIV-1 (N  |
TIV-2 (N  |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Any (%) |
Grade 2 (%) |
Grade 3 (%) |
Any (%) |
Grade 2 (%) |
Grade 3 (%) |
Any (%) |
Grade 2 (%) |
Grade 3 (%) |
|
| Injection-site adverse reactions | |||||||||
- Pain
 |
57.0 | 10.2 | 1.0 | 52.3 | 11.5 | 0.8 | 50.3 | 5.4 | 2.7 |
- Tenderness
 |
54.1 | 11.3 | 1.9 | 48.4 | 8.2 | 1.9 | 49.7 | 10.3 | 0.0 |
| - Erythema | 37.3 | 1.5 | 0.2 | 32.9 | 1.0 | 0.0 | 33.3 | 1.0 | 0.0 |
| - Swelling | 21.6 | 0.8 | 0.2 | 19.7 | 1.0 | 0.0 | 17.3 | 0.0 | 0.0 |
| Systemic adverse reactions | |||||||||
|
- Fever (≥100.4°F)
|
14.3 | 5.5 | 2.1 | 16.0 | 6.6 | 1.7 | 13.0 | 4.1 | 2.0 |
- Malaise
 |
38.1 | 14.5 | 4.6 | 35.2 | 14.8 | 4.7 | 32.4 | 12.8 | 6.8 |
- Myalgia
 |
26.7 | 6.6 | 1.9 | 26.6 | 9.4 | 1.6 | 25.0 | 6.8 | 2.7 |
- Headache
 |
8.9 | 2.5 | 0.6 | 9.4 | 3.9 | 0.0 | 12.2 | 4.7 | 0.0 |
- Irritability
 |
54.0 | 26.4 | 3.2 | 52.8 | 20.1 | 3.1 | 53.5 | 22.9 | 2.8 |
- Crying abnormal
 |
41.2 | 12.3 | 3.3 | 36.5 | 8.2 | 1.9 | 29.9 | 10.4 | 2.1 |
- Drowsiness
 |
37.7 | 8.4 | 1.3 | 32.1 | 3.8 | 0.6 | 31.9 | 5.6 | 0.7 |
- Appetite loss
 |
32.3 | 9.1 | 1.8 | 33.3 | 5.7 | 1.9 | 25.0 | 8.3 | 0.7 |
- Vomiting
 |
14.8 | 6.2 | 1.0 | 11.3 | 4.4 | 0.6 | 13.9 | 6.3 | 0.0 |
| Fluzone Quadrivalent (N  |
TIV-1 (N  |
TIV-2 (N  |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Any (%) |
Grade 2 (%) |
Grade 3 (%) |
Any (%) |
Grade 2 (%) |
Grade 3 (%) |
Any (%) |
Grade 2 (%) |
Grade 3 (%) |
|||
| Injection-site adverse reactions | |||||||||||
| - Pain | 66.6 | 15.8 | 2.1 | 64.6 | 9.5 | 2.0 | 63.8 | 11.6 | 2.8 | ||
| - Erythema | 34.1 | 2.9 | 1.8 | 36.8 | 3.4 | 1.2 | 35.2 | 2.5 | 1.8 | ||
| - Swelling | 24.8 | 2.8 | 1.4 | 25.4 | 1.5 | 1.2 | 25.9 | 2.5 | 1.8 | ||
| Systemic adverse reactions | |||||||||||
|
- Fever (≥100.4°F)
|
7.0 | 2.1 | 2.1 | 7.1 | 2.2 | 1.2 | 7.6 | 2.8 | 0.8 | ||
| - Headache | 23.1 | 6.8 | 2.2 | 21.2 | 5.1 | 2.7 | 24.4 | 7.5 | 2.0 | ||
| - Malaise | 31.9 | 11.2 | 5.5 | 32.8 | 11.4 | 5.6 | 33.4 | 10.8 | 5.0 | ||
| - Myalgia | 38.6 | 12.2 | 3.3 | 34.1 | 9.0 | 2.7 | 38.4 | 11.1 | 2.8 | ||
Among children 6 months through 8 years of age, unsolicited non-serious adverse events were reported in 1360 (47.0%) recipients in the Fluzone Quadrivalent group, 352 (48.0%) recipients in the TIV-1 group, and 346 (48.0%) recipients in the TIV-2 group. The most commonly reported unsolicited non-serious adverse events were cough, vomiting, and pyrexia. During the 28 days following vaccination, a total of 16 (0.6%) recipients in the Fluzone Quadrivalent group, 4 (0.5%) recipients in the TIV-1 group, and 4 (0.6%) recipients in the TIV-2 group, experienced at least one SAE; no deaths occurred. Throughout the study period, a total of 41 (1.4%) recipients in the Fluzone Quadrivalent group, 7 (1.0%) recipients in the TIV-1 group, and 14 (1.9%) recipients in the TIV-2 group, experienced at least one SAE. Three SAEs were considered to be possibly related to vaccination: croup in a Fluzone Quadrivalent recipient and 2 episodes of febrile seizure, 1 each in a TIV-1 recipient and a TIV-2 recipient. One death occurred in the TIV-1 group (a drowning 43 days post-vaccination).
Adults
In study 2 (NCT00988143, see http://clinicaltrials.gov), a multi-centered randomized, open-label trial conducted in the US, adults 18 years of age and older received one dose of either Fluzone Quadrivalent or one of two formulations of comparator trivalent influenza vaccine (TIV-1 or TIV-2). Each of the trivalent formulations contained an influenza type B virus that corresponded to one of the two type B viruses in Fluzone Quadrivalent (a type B virus of the Victoria lineage or a type B virus of the Yamagata lineage). The safety analysis set included 570 recipients, half aged 18-60 years and half aged 61 years or older. Among participants in the three vaccine groups combined, 67.2% were female (Fluzone Quadrivalent, 68.4%; TIV-1, 67.9%; TIV-2, 65.3%), 88.4% Caucasian (Fluzone Quadrivalent, 91.1%; TIV-1, 86.8%; TIV-2, 87.4%), 9.6% Black (Fluzone Quadrivalent, 6.8%; TIV-1, 12.1%; TIV-2, 10.0%), 0.4% Hispanic (Fluzone Quadrivalent, 0.0%; TIV-1, 0.5%; TIV-2, 0.5%), and 1.7% were of other racial/ethnic groups (Fluzone Quadrivalent, 2.1%; TIV-1, 0.5%; TIV-2, 2.2%). Table 4 summarizes solicited injection-site and systemic adverse reactions reported within 3 days post-vaccination via diary cards. Participants were monitored for unsolicited adverse events and SAEs during the 21 days following vaccination.
| Fluzone Quadrivalent (N  |
TIV-1 (N  |
TIV-2 (N  |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Any (%) | Grade 2 (%) |
Grade 3 (%) |
Any (%) | Grade 2 (%) |
Grade 3 (%) |
Any (%) | Grade 2 (%) |
Grade 3 (%) |
|
| Injection-site adverse reactions | |||||||||
| - Pain | 47.4 | 6.8 | 0.5 | 52.1 | 7.9 | 0.5 | 43.2 | 6.3 | 0.0 |
| - Erythema | 1.1 | 0.0 | 0.0 | 1.6 | 0.5 | 0.0 | 1.6 | 0.5 | 0.0 |
| - Swelling | 0.5 | 0.0 | 0.0 | 3.2 | 0.5 | 0.0 | 1.1 | 0.0 | 0.0 |
| - Induration | 0.5 | 0.0 | 0.0 | 1.6 | 0.5 | 0.0 | 0.5 | 0.0 | 0.0 |
| - Ecchymosis | 0.5 | 0.0 | 0.0 | 0.5 | 0.0 | 0.0 | 0.5 | 0.0 | 0.0 |
| Systemic adverse reactions | |||||||||
| - Myalgia | 23.7 | 5.8 | 0.0 | 25.3 | 5.8 | 0.0 | 16.8 | 5.8 | 0.0 |
| - Headache | 15.8 | 3.2 | 0.5 | 18.4 | 6.3 | 0.5 | 18.0 | 4.2 | 0.0 |
| - Malaise | 10.5 | 1.6 | 1.1 | 14.7 | 3.2 | 1.1 | 12.1 | 4.7 | 0.5 |
| - Shivering | 2.6 | 0.5 | 0.0 | 5.3 | 1.1 | 0.0 | 3.2 | 0.5 | 0.0 |
|
- Fever (≥100.4°F)
|
0.0 | 0.0 | 0.0 | 0.5 | 0.5 | 0.0 | 0.5 | 0.5 | 0.0 |
Unsolicited non-serious adverse events were reported in 33 (17.4%) recipients in the Fluzone Quadrivalent group, 45 (23.7%) recipients in the TIV-1 group, and 45 (23.7%) recipients in the TIV-2 group. The most commonly reported unsolicited non-serious adverse events were headache, cough, and oropharyngeal pain. In the follow-up period, there were two SAEs, 1 (0.5%) in the Fluzone Quadrivalent group and 1 (0.5%) in the TIV-2 group. No deaths were reported during the trial period.
Geriatric Adults
In Study 3 (NCT01218646, see http://clinicaltrials.gov), a multi-center, randomized, double-blind trial conducted in the US, adults 65 years of age and older received one dose of either Fluzone Quadrivalent, or one of two formulations of comparator trivalent influenza vaccine (TIV-1 or TIV-2). Each of the trivalent formulations contained an influenza type B virus that corresponded to one of the two type B viruses in Fluzone Quadrivalent (a type B virus of the Victoria lineage or a type B virus of the Yamagata lineage). The safety analysis set included 675 recipients. Among participants in the three vaccine groups combined, 55.7% were female (Fluzone Quadrivalent, 57.3%; TIV-1, 56.0%; TIV-2, 53.8%), 89.5% Caucasian (Fluzone Quadrivalent, 87.6%; TIV-1, 89.8%; TIV-2, 91.1%), 2.2% Black (Fluzone Quadrivalent, 4.0%; TIV-1, 1.8%; TIV-2, 0.9%), 7.4% Hispanic (Fluzone Quadrivalent, 8.4%; TIV-1, 7.6%; TIV-2, 6.2%) and 0.9% were of other racial/ethnic groups (Fluzone Quadrivalent, 0.0%; TIV-1, 0.9%; TIV-2, 1.8%).
Table 5 summarizes solicited injection-site and systemic adverse reactions reported within 7 days post-vaccination via diary cards. Participants were monitored for unsolicited adverse events and SAEs during the 21 days following vaccination.
| Fluzone Quadrivalent (N  |
TIV-1 (N  |
TIV-2 (N  |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Any (%) | Grade 2 (%) |
Grade 3 (%) |
Any (%) | Grade 2 (%) |
Grade 3 (%) |
Any (%) | Grade 2 (%) |
Grade 3 (%) |
|
| Injection-site adverse reactions | |||||||||
| - Pain | 32.6 | 1.3 | 0.9 | 28.6 | 2.7 | 0.0 | 23.1 | 0.9 | 0.0 |
| - Erythema | 2.7 | 0.9 | 0.0 | 1.3 | 0.0 | 0.0 | 1.3 | 0.4 | 0.0 |
| - Swelling | 1.8 | 0.4 | 0.0 | 1.3 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Systemic adverse reactions | |||||||||
| - Myalgia | 18.3 | 4.0 | 0.4 | 18.3 | 4.0 | 0.0 | 14.2 | 2.7 | 0.4 |
| - Headache | 13.4 | 1.3 | 0.4 | 11.6 | 1.3 | 0.0 | 11.6 | 1.8 | 0.4 |
| - Malaise | 10.7 | 4.5 | 0.4 | 6.3 | 0.4 | 0.0 | 11.6 | 2.7 | 0.9 |
|
- Fever (≥100.4°F) |
1.3 | 0.0 | 0.4 | 0.0 | 0.0 | 0.0 | 0.9 | 0.4 | 0.4 |
Unsolicited non-serious adverse events were reported in 28 (12.4%) recipients in the Fluzone Quadrivalent group, 22 (9.8%) recipients in the TIV-1 group, and 22 (9.8%) recipients in the TIV-2 group. The most commonly reported adverse events were oropharyngeal pain, rhinorrhea, injection-site induration, and headache. Three SAEs were reported during the follow-up period, 2 (0.9%) in the TIV-1 group and 1 (0.4%) in the TIV-2 group. No deaths were reported during the trial period.
6.2 Post-Marketing Experience
Currently, there are no post-marketing data available for Fluzone Quadrivalent vaccine.
The following events have been spontaneously reported during the post-approval use of the trivalent formulation of Fluzone. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to vaccine exposure. Adverse events were included based on one or more of the following factors: severity, frequency of reporting, or strength of evidence for a causal relationship to Fluzone.
- Blood and Lymphatic System Disorders: Thrombocytopenia, lymphadenopathy
- Immune System Disorders: Anaphylaxis, other allergic/hypersensitivity reactions (including urticaria, angioedema)
- Eye disorders: Ocular hyperemia
- Nervous System Disorders: Guillain-Barré syndrome (GBS), convulsions, febrile convulsions, myelitis (including encephalomyelitis and transverse myelitis), facial palsy (Bell's palsy), optic neuritis/neuropathy, brachial neuritis, syncope (shortly after vaccination), dizziness, paresthesia
- Vascular Disorders: Vasculitis, vasodilation/flushing
- Respiratory, Thoracic and Mediastinal Disorders: Dyspnea, pharyngitis, rhinitis, cough, wheezing, throat tightness
- Skin and Subcutaneous Tissue Disorders: Stevens-Johnson syndrome
- General Disorders and Administration Site Conditions: Pruritus, asthenia/fatigue, pain in extremities, chest pain
- Gastrointestinal Disorders: Vomiting
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C: Animal reproduction studies have not been conducted with Fluzone Quadrivalent. It is also not known whether Fluzone Quadrivalent can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Fluzone Quadrivalent should be given to a pregnant woman only if clearly needed.
Sanofi Pasteur Inc. is conducting a prospective pregnancy exposure registry to collect data on pregnancy outcomes and newborn health status following vaccination with Fluzone Quadrivalent during pregnancy. Healthcare providers are encouraged to enroll women who receive Fluzone Quadrivalent during pregnancy in Sanofi Pasteur Inc.'s vaccination pregnancy registry by calling 1-800-822-2463.
8.3 Nursing Mothers
It is not known whether Fluzone Quadrivalent is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Fluzone Quadrivalent is administered to a nursing woman.
8.4 Pediatric Use
Safety and effectiveness of Fluzone Quadrivalent in children below the age of 6 months have not been established. Safety and immunogenicity of Fluzone Quadrivalent was evaluated in children 6 months through 8 years of age. [See Adverse Reactions (6.1) and Clinical Studies (14.1) ]
8.5 Geriatric Use
Safety and immunogenicity of Fluzone Quadrivalent was evaluated in adults 65 years of age and older. [See Clinical Studies (14.3) ] Antibody responses to Fluzone Quadrivalent are lower in persons ≥65 years of age than in younger adults.
11 DESCRIPTION
Fluzone Quadrivalent (Influenza Virus Vaccine) for intramuscular injection is an inactivated influenza virus vaccine, prepared from influenza viruses propagated in embryonated chicken eggs. The virus-containing allantoic fluid is harvested and inactivated with formaldehyde. Influenza virus is concentrated and purified in a linear sucrose density gradient solution using a continuous flow centrifuge. The virus is then chemically disrupted using a non-ionic surfactant, octylphenol ethoxylate (Triton® X-100), producing a "split virus". The split virus is further purified and then suspended in sodium phosphate-buffered isotonic sodium chloride solution. The Fluzone Quadrivalent process uses an additional concentration factor after the ultrafiltration step in order to obtain a higher hemagglutinin (HA) antigen concentration. Antigens from the four strains included in the vaccine are produced separately and then combined to make the quadrivalent formulation.
Fluzone Quadrivalent suspension for injection is clear and slightly opalescent in color.
Neither antibiotics nor preservative are used in the manufacture of Fluzone Quadrivalent.
The Fluzone Quadrivalent prefilled syringe and vial presentations are not made with natural rubber latex.
Fluzone Quadrivalent is standardized according to United States Public Health Service requirements and is formulated to contain 60 micrograms (mcg) HA per 0.5 mL dose in the recommended ratio of 15 mcg HA of each of the following four influenza strains recommended for the 2013-2014 influenza season: A/California/07/2009 X-179A (H1N1), A/Texas/50/2012 X-223A (H3N2) (an A/Victoria/361/2011-like virus), B/Massachusetts/02/2012 (B Yamagata lineage), and B/Brisbane/60/2008 (B Victoria lineage). The amounts of HA and other ingredients per dose of vaccine are listed in Table 6.
| Ingredient | Quantity (per dose) |
|
|---|---|---|
| Fluzone Quadrivalent 0.25 mL Dose |
Fluzone Quadrivalent 0.5 mL Dose |
|
|
Active Substance: Split influenza virus, inactivated strains |
30 mcg HA total | 60 mcg HA total |
| A (H1N1) | 7.5 mcg HA | 15 mcg HA |
| A (H3N2) | 7.5 mcg HA | 15 mcg HA |
| B/(Victoria lineage) | 7.5 mcg HA | 15 mcg HA |
| B/(Yamagata lineage) | 7.5 mcg HA | 15 mcg HA |
| Other: | ||
| Sodium phosphate-buffered isotonic sodium chloride solution | QS |
QS |
| Formaldehyde | ≤50 mcg | ≤100 mcg |
| Octylphenol ethoxylate | ≤125 mcg | ≤250 mcg |
| Preservative | None | None |
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Influenza illness and its complications follow infection with influenza viruses. Global surveillance of influenza identifies yearly antigenic variants. Since 1977, antigenic variants of influenza A (H1N1 and H3N2) viruses and influenza B viruses have been in global circulation. Since 2001, two distinct lineages of influenza B (Victoria and Yamagata lineages) have co-circulated worldwide. Protection from influenza virus infection has not been correlated with a specific level of hemagglutination inhibition (HI) antibody titer post-vaccination. However, in some human studies, antibody titers ≥1:40 have been associated with protection from influenza illness in up to 50% of subjects. (see ref. 2) (see ref. 3)
Antibodies against one influenza virus type or subtype confer limited or no protection against another. Furthermore, antibodies to one antigenic variant of influenza virus might not protect against a new antigenic variant of the same type or subtype. Frequent development of antigenic variants through antigenic drift is the virologic basis for seasonal epidemics and the reason for the usual change of one or more new strains in each year's influenza vaccine. Therefore, influenza vaccines are standardized to contain the hemagglutinins of influenza virus strains, representing the influenza viruses likely to be circulating in the US in the upcoming winter.
Annual vaccination with the current vaccine is recommended because immunity during the year after vaccination declines, and because circulating strains of influenza virus change from year to year.
14 CLINICAL STUDIES
14.1 Immunogenicity of Fluzone Quadrivalent in Children 6 Months through 8 Years of Age
In Study 1 (NCT01240746) (see Section 6.1), 1419 children 6 months through 35 months of age and 2101 children 3 years through 8 years of age were included in the per-protocol immunogenicity analysis. Participants received one or two 0.25 mL doses or one or two 0.5 mL doses, respectively of Fluzone Quadrivalent, TIV-1, or TIV-2. For participants who received two doses, the doses were administered approximately 4 weeks apart. The distribution of demographic characteristics was similar to that of the safety analysis (see Section 6.1).
HI antibody geometric mean titers (GMTs) and seroconversion rates 28 days following vaccination with Fluzone Quadrivalent were non-inferior to those following each TIV for all four strains, based on pre-specified criteria (see Table 7 and Table 8).
| Antigen Strain | Fluzone Quadrivalent |
Pooled TIV
|
GMT Ratio (95%CI) |
|
N
 |
N
 |
|||
| GMT | GMT | |||
| A (H1N1) | 1124 | 1096 | 1.03 (0.93; 1.14) | |
| A (H3N2) | 822 | 828 | 0.99 (0.91; 1.08) | |
| Fluzone Quadrivalent |
TIV-1 |
TIV-2 |
GMT Ratio (95%CI) |
|
N
 |
N
 |
N
 |
||
| GMT | GMT | GMT | ||
|
B/Brisbane/60/2008 (B Victoria) |
86.1 | 64.3 | (19.5) |
1.34 (1.20; 1.50) |
|
B/Florida/04/2006 (B Yamagata) |
61.5 | (16.3) |
58.3 | 1.06 (0.94; 1.18) |
| Antigen Strain | Fluzone Quadrivalent |
Pooled TIV |
Difference of Seroconversion Rates (95% CI)  |
|
N
 |
N
 |
|||
Seroconversion
 |
||||
| A (H1N1) | 92.4 | 91.4 | 0.9 (-0.9; 3.0) | |
| A (H3N2) | 88.0 | 84.2 | 3.8 (1.4; 6.3) | |
| Fluzone Quadrivalent |
TIV-1 (B Victoria) |
TIV-2 (B Yamagata) |
Difference of Seroconversion Rates (95% CI)  |
|
N
 |
N
 |
N
 |
||
Seroconversion
 |
||||
|
B/Brisbane/60/2008 (B Victoria) |
71.8 | 61.1 | (20.0) |
10.7 (6.4; 15.1) |
|
B/Florida/04/2006 (B Yamagata) |
66.1 | (17.9) |
64.0 | 2.0 (-2.2; 6.4) |
Non-inferiority immunogenicity criteria based on HI antibody GMTs and seroconversion rates were also met when age subgroups (6 months to <36 months and 3 years to <9 years) were examined. In addition, HI antibody GMTs and seroconversion rates following Fluzone Quadrivalent were higher than those following TIV for the B strain not contained in each respective TIV based on pre-specified criteria (the lower limit of the 2-sided 95% CI of the ratio of the GMTs [Fluzone Quadrivalent divided by TIV] >1.5 for each B strain in Fluzone Quadrivalent compared with the corresponding B strain not contained in each TIV and the lower limit of the two 2-sided 95% CI of the difference of the seroconversion rates [Fluzone Quadrivalent minus TIV] >10% for each B strain in Fluzone Quadrivalent compared with the corresponding B strain not contained in each TIV).
14.2 Immunogenicity of Fluzone Quadrivalent in Adults ≥18 Years of Age
In Study 2 (NCT00988143) (see Section 6.1), 565 adults 18 years of age and older who had received one dose of Fluzone Quadrivalent, TIV-1, or TIV-2 were included in the per-protocol immunogenicity analysis. The distribution of demographic characteristics was similar to that of the safety analysis (see Section 6.1).
HI antibody GMTs 21 days following vaccination with Fluzone Quadrivalent were non-inferior to those following each TIV for all four strains, based on pre-specified criteria (see Table 9).
| Antigen Strain | Fluzone Quadrivalent |
Pooled TIV
|
GMT Ratio (95%CI) |
|
N
 |
N
 |
|||
| GMT | GMT | |||
| A (H1N1) | 161 | 151 | 1.06 (0.87; 1.31) | |
| A (H3N2) | 304 | 339 | 0.90 (0.70; 1.15) | |
| Fluzone Quadrivalent |
TIV-1 (B Victoria) |
TIV-2 (B Yamagata) |
GMT Ratio (95%CI) |
|
N
 |
N
 |
N
 |
||
| GMT | GMT | GMT | ||
|
B/Brisbane/60/2008 (B Victoria) |
101 | 114 | (44.0) |
0.89 (0.70; 1.12) |
|
B/Florida/04/2006 (B Yamagata) |
155 | (78.1) |
135 | 1.15 (0.93; 1.42) |
14.3 Immunogenicity of Fluzone Quadrivalent in Geriatric Adults ≥65 Years of Age
In Study 3 (NCT01218646) (see Section 6.1), 660 adults 65 years of age and older were included in the per-protocol immunogenicity analysis. The distribution of demographic characteristics was similar to that of the safety analysis (see Section 6.1).
HI antibody GMTs 21 days following vaccination with Fluzone Quadrivalent were non-inferior to those following TIV for all four strains, based on pre-specified criteria (see Table 10). Seroconversion rates 21 days following Fluzone Quadrivalent were non-inferior to those following TIV for H3N2, B/Brisbane, and B/Florida, but not for H1N1 (see Table 11). The HI antibody GMT following Fluzone Quadrivalent was higher than that following TIV-1 for B/Florida but not higher than that following TIV-2 for B/Brisbane, based on pre-specified criteria (the lower limit of the 2-sided 95% CI of the ratio of the GMTs [Fluzone Quadrivalent divided by TIV] >1.5 for each B strain in Fluzone Quadrivalent compared with the corresponding B strain not contained in each TIV). Seroconversion rates following Fluzone Quadrivalent were higher than those following TIV for the B strain not contained in each respective TIV, based on pre-specified criteria (the lower limit of the two 2-sided 95% CI of the difference of the seroconversion rates [Fluzone Quadrivalent minus TIV] >10% for each B strain in Fluzone Quadrivalent compared with the corresponding B strain not contained in each TIV).
| Antigen Strain | Fluzone Quadrivalent |
Pooled TIV
|
GMT Ratio (95%CI) |
|
N
 |
N
 |
|||
| GMT | GMT | |||
| A (H1N1) | 231 | 270 | 0.85 (0.67; 1.09) | |
| A (H3N2) | 501 | 324 | 1.55 (1.25; 1.92) | |
| Fluzone Quadrivalent |
TIV-1 (B Victoria) |
TIV-2 (B Yamagata) |
GMT Ratio (95%CI) |
|
N
 |
N
 |
N
 |
||
| GMT | GMT | GMT | ||
|
B/Brisbane/60/2008 (B Victoria) |
73.8 | 57.9 | (42.2) |
1.27 (1.05; 1.55) |
|
B/Florida/04/2006 (B Yamagata) |
61.1 | (28.5) |
54.8 | 1.11 (0.90; 1.37) |
| Antigen Strain | Fluzone Quadrivalent |
Pooled TIV
|
Difference of Seroconversion Rate (95% CI)  |
|
N
 |
N
 |
|||
Seroconversion
 |
||||
| A (H1N1) | 65.91 | 69.77 | -3.86 (-11.50; 3.56) | |
| A (H3N2) | 69.09 | 59.32 | 9.77 (1.96; 17.20) | |
| Fluzone Quadrivalent |
TIV-1 (B Victoria) |
TIV-2 (B Yamagata) |
Difference of Seroconversion Rate (95% CI)  |
|
N
 |
N
 |
N
 |
||
Seroconversion
 |
||||
|
B/Brisbane/60/2008 (B Victoria) |
28.64 | 18.72 | (8.60) |
9.91 (1.96; 17.70) |
|
B/Florida/04/2006 (B Yamagata) |
33.18 | (9.13) |
31.22 | 1.96 (-6.73; 10.60) |
15 REFERENCES
-
1 Lasky T, Terracciano GJ, Magder L, et al. The Guillain-Barré syndrome and the 1992-1993 and 1993-1994 influenza vaccines. N Engl J Med 1998;339:1797-802. -
2 Hannoun C, Megas F, Piercy J. Immunogenicity and protective efficacy of influenza vaccination. Virus Res 2004;103:133-138. -
3 Hobson D, Curry RL, Beare AS, Ward-Gardner A. The role of serum haemagglutination-inhibiting antibody in protection against challenge infection with influenza A2 and B viruses. J Hyg Camb 1972;70:767-777.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Single-dose, prefilled syringe (yellow plunger rod), without needle, 0.25 mL (NDC 49281-513-00) (not made with natural rubber latex). Supplied as package of 10 (NDC 49281-513-25).
Single-dose, prefilled syringe (purple plunger rod), without needle, 0.5 mL (NDC 49281-413-88) (not made with natural rubber latex). Supplied as package of 10 (NDC 49281-413-50).
Single-dose vial, 0.5 mL (NDC 49281-413-58) (not made with natural rubber latex). Supplied as package of 10 (NDC 49281-413-10).
16.2 Storage and Handling
Store all Fluzone Quadrivalent presentations refrigerated at 2° to 8°C (35° to 46°F). DO NOT FREEZE. Discard if vaccine has been frozen.
Do not use after the expiration date shown on the label.
17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling (Patient Information). Inform the vaccine recipient or guardian:
- Fluzone Quadrivalent contains killed viruses and cannot cause influenza.
- Fluzone Quadrivalent stimulates the immune system to protect against influenza, but does not prevent other respiratory infections.
- Annual influenza vaccination is recommended.
- Report adverse reactions to their healthcare provider and/or to the Vaccine Adverse Event Reporting System (VAERS) at 1-800-822-7967.
- Sanofi Pasteur Inc. is conducting a prospective pregnancy exposure registry to collect data on pregnancy outcomes and newborn health status following vaccination with Fluzone Quadrivalent during pregnancy. Women who receive Fluzone Quadrivalent during pregnancy are encouraged to contact Sanofi Pasteur Inc. directly or have their healthcare provider contact Sanofi Pasteur Inc. at 1-800-822-2463.
Vaccine Information Statements must be provided to vaccine recipients or their guardians, as required by the National Childhood Vaccine Injury Act of 1986 prior to immunization. These materials are available free of charge at the Centers for Disease Control and Prevention (CDC) website (www.cdc.gov/vaccines).
Fluzone is a registered trademark of Sanofi Pasteur Inc.
Manufactured by:
Sanofi Pasteur Inc.
Swiftwater PA 18370 USA
Patient Information Sheet
Fluzone® Quadrivalent
Influenza Virus Vaccine
Please read this information sheet before getting Fluzone® Quadrivalent vaccine. This summary is not intended to take the place of talking with your healthcare provider. If you have questions or would like more information, please talk with your healthcare provider.
What is Fluzone Quadrivalent vaccine?
Fluzone Quadrivalent is a vaccine that helps protect against influenza illness (flu).
Fluzone Quadrivalent vaccine is for people who are 6 months of age and older.
Vaccination with Fluzone Quadrivalent vaccine may not protect all people who receive the vaccine.
Who should not get Fluzone Quadrivalent vaccine?
You should not get Fluzone Quadrivalent vaccine if you:
- ever had a severe allergic reaction to eggs or egg products.
- ever had a severe allergic reaction after getting any flu vaccine.
- are younger than 6 months of age.
Tell your healthcare provider if you or your child have or have had:
- Guillain-Barré syndrome (severe muscle weakness) after getting a flu vaccine.
- problems with your immune system as the immune response may be diminished.
How is the Fluzone Quadrivalent vaccine given?
Fluzone Quadrivalent vaccine is a shot given into the muscle of the arm.
For infants, Fluzone Quadrivalent vaccine is a shot given into the muscle of the thigh.
What are the possible side effects of Fluzone Quadrivalent vaccine?
The most common side effects of Fluzone Quadrivalent vaccine are:
- pain, redness, swelling, bruising and hardness where you got the shot
- muscle aches
- tiredness
- headache
- fever
These are not all of the possible side effects of Fluzone Quadrivalent vaccine. You can ask your healthcare provider for a list of other side effects that is available to healthcare professionals.
Call your healthcare provider for advice about any side effects that concern you. You may report side effects to the Vaccine Adverse Event Reporting System (VAERS) at 1-800-822-7967 or http://vaers.hhs.gov. Sanofi Pasteur Inc. is collecting information on pregnancy outcomes and the health of newborns following vaccination with Fluzone Quadrivalent during pregnancy. Women who receive Fluzone Quadrivalent during pregnancy are encouraged to contact Sanofi Pasteur Inc. directly or have their healthcare provider contact Sanofi Pasteur Inc. at 1-800-822-2463.
What are the ingredients in Fluzone Quadrivalent vaccine?
Fluzone Quadrivalent vaccine contains 4 killed flu virus strains.
Inactive ingredients include formaldehyde and octylphenol ethoxylate.
Manufactured by:
Sanofi Pasteur Inc.
Swiftwater, PA 18370 USA
6243, 6244
PRINCIPAL DISPLAY PANEL - 0.5 mL Vial Label
NDC 49281-413-58
Influenza Virus
Vaccine
Fluzone
® Quadrivalent
No Preservative
2013-2014 Formula
Mfd by: Sanofi Pasteur Inc.
Rx only
1 Dose (0.5 mL)
IM only

PRINCIPAL DISPLAY PANEL - 0.5 mL Vial Package
NDC 49281-413-10
2013 – 2014 Formula
Rx only
10 Vials
0.5 mL each
4
STRAINS
Influenza
Virus Vaccine
Fluzone ® Quadrivalent
For 3 years of age and older
For intramuscular injection only
SANOFI PASTEUR

PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Label
NDC 49281-413-88
0.5 mL Dose
Influenza Virus Vaccine
Fluzone
® Quadrivalent
2013-2014 Formula
No Preservative
Mfd by: Sanofi Pasteur Inc.
IM only
Rx only

PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Package
NDC 49281-413-50
2013 – 2014 Formula
10 Prefilled Syringes
0.5 mL each
Rx only
4
STRAINS
Influenza
Virus Vaccine
Fluzone ® Quadrivalent
For 3 years of age and older
For intramuscular injection only
SANOFI PASTEUR

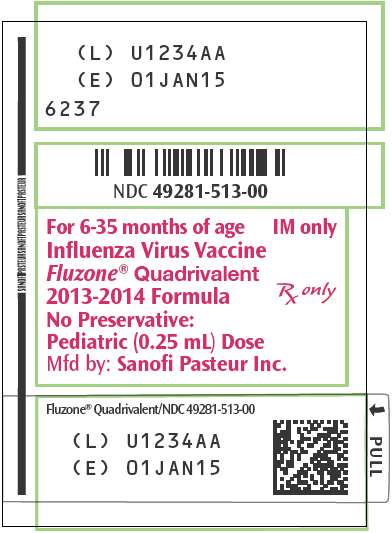
PRINCIPAL DISPLAY PANEL - 0.25 mL Syringe Label
NDC 49281-513-00
For 6-35 months of age
Influenza Virus Vaccine
Fluzone
®
Quadrivalent
2013-2014 Formula
No Preservative:
Pediatric (0.25 mL) Dose
Mfd by: Sanofi Pasteur Inc.
IM only
Rx only
PRINCIPAL DISPLAY PANEL - 0.25 mL Syringe Package
NDC 49281-513-25
2013 – 2014 Formula
PEDIATRIC DOSE
FOR 6 - 35 MONTHS OF AGE
10 Prefilled Syringes
0.25 mL each
Rx only
4
STRAINS
Influenza
Virus Vaccine
Fluzone ® Quadrivalent
For 6-35 months of age
For intramuscular injection only
SANOFI PASTEUR

FLUZONE QUADRIVALENTINFLUENZA A VIRUS A/CALIFORNIA/7/2009 X-179A (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/TEXAS/50/2012 X-223A (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/MASSACHUSETTS/2/2012 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/BRISBANE/60/2008 ANTIGEN (FORMALDEHYDE INACTIVATED) INJECTION, SUSPENSION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
FLUZONE QUADRIVALENTINFLUENZA A VIRUS A/CALIFORNIA/7/2009 X-179A (H1N1) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA A VIRUS A/TEXAS/50/2012 X-223A (H3N2) ANTIGEN (FORMALDEHYDE INACTIVATED), INFLUENZA B VIRUS B/MASSACHUSETTS/2/2012 ANTIGEN (FORMALDEHYDE INACTIVATED), and INFLUENZA B VIRUS B/BRISBANE/60/2008 ANTIGEN (FORMALDEHYDE INACTIVATED) INJECTION, SUSPENSION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||