Guanidine hydrochloride
GUANIDINEHYDROCHLORIDETablets
FULL PRESCRIBING INFORMATION: CONTENTS*
- GUANIDINE HYDROCHLORIDE DESCRIPTION
- CLINICAL PHARMACOLOGY
- GUANIDINE HYDROCHLORIDE INDICATIONS AND USAGE
- GUANIDINE HYDROCHLORIDE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- GUANIDINE HYDROCHLORIDE ADVERSE REACTIONS
- GUANIDINE HYDROCHLORIDE DOSAGE AND ADMINISTRATION
- OVERDOSAGE
- HOW SUPPLIED
- PRINCIPAL DISPLAY PANEL - 125 mg Tablet Bottle Label
FULL PRESCRIBING INFORMATION
GUANIDINE HYDROCHLORIDE DESCRIPTION
Chemically, guanidine (aminomethanamidine) hydrochloride is a crystalline powder freely soluble in water and alcohol. The aqueous solution is neutral.
The structural formula is:
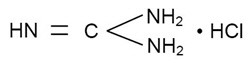
Each tablet contains 125 mg of guanidine hydrochloride with no color additive in the base. It also contains the following inactive ingredients: colloidal silicon dioxide, magnesium stearate, mannitol, and microcrystalline cellulose.
CLINICAL PHARMACOLOGY
Guanidine apparently acts by enhancing the release of acetylcholine following a nerve impulse. It also appears to slow the rates of depolarization and repolarization of muscle cell membranes.
GUANIDINE HYDROCHLORIDE INDICATIONS AND USAGE
Guanidine is indicated for the reduction of the symptoms of muscle weakness and easy fatigability associated with the myasthenic syndrome of Eaton-Lambert. It is not indicated for treating myasthenia gravis. The Eaton-Lambert syndrome is ordinarily differentiated from myasthenia gravis by the usual association of the syndrome with small cell carcinoma of the lung, but myography may be necessary to make the diagnosis.
GUANIDINE HYDROCHLORIDE CONTRAINDICATIONS
Guanidine is contraindicated in individuals with a history of intolerance or allergy to this drug.
WARNINGS
Fatal bone-marrow suppression, apparently dose related, can occur with guanidine.
Safe use of guanidine hydrochloride in pregnancy has not been established. Therefore, the benefits of therapy must be weighed against the potential hazards. Because guanidine is excreted in milk, patients on this drug should discontinue breastfeeding.
Since there is inadequate experience in children who have received this drug, safety and efficacy in children have not been established.
PRECAUTIONS
Baseline blood studies should be followed by frequent red and white blood cell and differential counts. The drug should be discontinued upon appearance of bone-marrow suppression. Concurrent therapy with other drugs that may cause bone-marrow suppression should be avoided.
Renal function may be affected in some patients receiving guanidine. Patients should therefore have regular urine examinations and serum creatinine determinations while taking this drug.
Physicians should be given adequate precautions pertaining to the gastrointestinal side effects and the possibility of induced behavior disorders.
Treatment should not be continued longer than necessary.
GUANIDINE HYDROCHLORIDE ADVERSE REACTIONS
Anemia, leukopenia, and thrombocytopenia resulting from bone-marrow suppression attributable to guanidine have been reported. Other adverse reactions that have been observed are:
General: sore throat, rash, fever.
Neurologic: paresthesia of lips, face, hands, feet; cold sensations in hands and feet; nervousness, lightheadedness, jitteriness, increased irritability; tremor, trembling sensation; ataxia; emotional lability; psychotic state; confusion; mood changes, and hallucinations.
Gastrointestinal: dry mouth; gastric irritation; anorexia; nausea; diarrhea; abdominal cramping. Gastrointestinal side effects may preclude the use of guanidine as a desired form of therapy.
Dermatologic: rash, flushing or pink complexion; folliculitis; petechiae, purpura, ecchymoses; sweating; skin eruptions; dryness and scaling of the skin.
Renal: elevation of blood creatinine, uremia; chronic interstitial nephritis, acute interstitial nephritis, and renal tubular necrosis.
Hepatic: abnormal liver function tests.
Cardiac: palpitation, tachycardia, atrial fibrillation, hypotension.
GUANIDINE HYDROCHLORIDE DOSAGE AND ADMINISTRATION
Initial dosage is usually between 10 and 15 mg/kg (5 to 7 mg/pound) of body weight per day in 3 or 4 divided doses. This dosage may be gradually increased to a total daily dosage of 35 mg/kg (16 mg/pound) of body weight per day or up to the development of side effects. As individual tolerance is highly variable, the dosage must be carefully titrated. Once a tolerable dose has been established, it should be continued. Occasionally removal of the primary neoplastic lesion may result in improvement of symptoms, permitting the discontinuance of guanidine.
OVERDOSAGE
Mild gastrointestinal disorders, such as anorexia, increased peristalsis, or diarrhea are early warnings that tolerance is being exceeded. These symptoms may be relieved by atropine, but nevertheless note should be taken of these symptoms and dosage reductions considered. Slight numbness or tingling of the lips and fingertips shortly after taking a dose of guanidine has been reported. This per se is not an indication to discontinue treatment and/or reduce dosage.
Severe guanidine intoxication is characterized by nervous hyperirritability, fibrillary tremors and convulsive contractions of muscle, salivation, vomiting, diarrhea, hypoglycemia, and circulatory disturbances. Administration of intravenous calcium gluconate may control the neuromuscular and convulsive symptoms and provide some relief of other toxic manifestations.
Atropine is more effective than calcium in relieving the G.I. symptoms, circulatory disturbances, and changes in blood sugar.
HOW SUPPLIED
Guanidine hydrochloride tablets: 125 mg, white, round tablet; impressed with the product identification number "KEY 74" on one side. Guanidine hydrochloride tablets are available in bottles of 100 (NDC 0085-0492-01).
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Manufactured for: Merck Sharp & Dohme Corp., a subsidiary of
MERCK & CO., INC., Whitehouse Station, NJ 08889, USA
Manufactured by: Schering Plough Canada Inc.
Pointe Claire, Quebec H9R 1B4, Canada
Copyright © 1987, 1992 Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc.
All rights reserved.
Revised 05/2012
O-012559-GU/HCL-TB-USPI.2
81-483544
PRINCIPAL DISPLAY PANEL - 125 mg Tablet Bottle Label
Guanidine
Hydrochloride
NDC 0085-0492-01
Each tablet contains
Guanidine Hydrochloride
125 mg
100 TABLETS
Rx only
Manufactured for: Merck Sharp & Dohme
Corp., a subsidiary of Merck & Co., Inc.,
Whitehouse Station, NJ 08889, USA
Manufactured by: Schering Plough Canada Inc.,
Pointe Claire, Quebec H9R 1B4, Canada
Formulated in Canada.

Guanidine hydrochlorideGuanidine hydrochloride TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||