Watson Laboratories, Inc.
Revised: November 2013
173191-2
FULL PRESCRIBING INFORMATION
BOXED WARNING
Hepatotoxicity
Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed 4000 milligrams per day, and often involve more than one acetaminophen containing product.
Hydrocodone bitartrate and acetaminophen is supplied in tablet form for oral administration. Hydrocodone bitartrate is an opioid analgesic and antitussive and occurs as fine, white crystals or as a crystalline powder. It is affected by light. The chemical name is 4,5α-Epoxy-3-methoxy-17methylmorphinan-6-one tartrate (1:1) hydrate (2:5). It has the following structural formula:

Acetaminophen, 4´-Hydroxyacetanilide, a slightly bitter, white, odorless, crystalline powder, is a non-opiate, non-salicylate analgesic and antipyretic. It has the following structural formula:
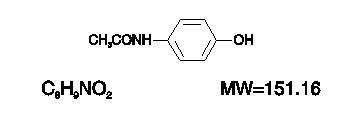
Hydrocodone Bitartrate and Acetaminophen Tablets USP for oral administration are available in a variety of strengths as described in the following table.
| Strength
|
Hydrocodone
Bitartrate
|
Acetaminophen
|
| 2.5 mg/500 mg |
2.5 mg |
500 mg |
| 5 mg/500 mg |
5 mg |
500 mg |
| 7.5 mg/325 mg |
7.5 mg |
325 mg |
| 7.5 mg/500 mg |
7.5 mg |
500 mg |
| 7.5 mg/650 mg |
7.5 mg |
650 mg |
| 7.5 mg/750 mg |
7.5 mg |
750 mg |
| 10 mg/325 mg |
10 mg |
325 mg |
| 10 mg/500 mg |
10 mg |
500 mg |
| 10 mg/650 mg |
10 mg |
650 mg |
| 10 mg/660 mg |
10 mg |
660 mg |
| 10 mg/750 mg |
10 mg |
750 mg |
In addition, each tablet contains the following inactive ingredients: anhydrous lactose, croscarmellose sodium, crospovidone, magnesium stearate, microcrystalline cellulose, povidone, starch and stearic acid; except the 7.5 mg/325 mg, 10 mg/325 mg and 10 mg/500 mg tablets do not contain anhydrous lactose.
Meets USP Dissolution Test 1.
Hydrocodone is a semisynthetic narcotic analgesic and antitussive with multiple actions qualitatively similar to those of codeine. Most of these involve the central nervous system and smooth muscle. The precise mechanism of action of hydrocodone and other opiates is not known, although it is believed to relate to the existence of opiate receptors in the central nervous system. In addition to analgesia, narcotics may produce drowsiness, changes in mood and mental clouding.
The analgesic action of acetaminophen involves peripheral influences, but the specific mechanism is as yet undetermined. Antipyretic activity is mediated through hypothalamic heat regulating centers. Acetaminophen inhibits prostaglandin synthetase. Therapeutic doses of acetaminophen have negligible effects on the cardiovascular or respiratory systems; however, toxic doses may cause circulatory failure and rapid, shallow breathing.
The behavior of the individual components is described below.
Hydrocodone: Following a 10 mg oral dose of hydrocodone administered to five adult male subjects, the mean peak concentration was 23.6 ± 5.2 ng/mL. Maximum serum levels were achieved at 1.3 ± 0.3 hours and the half-life was determined to be 3.8 ± 0.3 hours. Hydrocodone exhibits a complex pattern of metabolism including O-demethylation, N-demethylation and 6-keto reduction to the corresponding 6-α- and 6-β- hydroxy-metabolites. See
OVERDOSAGE
for toxicity information.
Acetaminophen: Acetaminophen is rapidly absorbed from the gastrointestinal tract and is distributed throughout most body tissues. The plasma half-life is 1.25 to 3 hours, but may be increased by liver damage and following overdosage. Elimination of acetaminophen is principally by liver metabolism (conjugation) and subsequent renal excretion of metabolites.
Approximately 85% of an oral dose appears in the urine within 24 hours of administration, most as the glucuronide conjugate, with small amounts of other conjugates and unchanged drug. See
OVERDOSAGE
for toxicity information.
Hydrocodone and acetaminophen tablets are indicated for the relief of moderate to moderately severe pain.
This product should not be administered to patients who have previously exhibited hypersensitivity to hydrocodone or acetaminophen. Patients known to be hypersensitive to other opioids may exhibit cross-sensitivity to hydrocodone.
Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed 4000 milligrams per day, and often involve more than one acetaminophen containing product. The excessive intake of acetaminophen may be intentional to cause self-harm or unintentional as patients attempt to obtain more pain relief or unknowingly take other acetaminophen-containing products.
The risk of acute liver failure is higher in individuals with underlying liver disease and in individuals who ingest alcohol while taking acetaminophen.
Instruct patients to look for acetaminophen or APAP on package labels and not to use more than one product that contains acetaminophen. Instruct patients to seek medical attention immediately upon ingestion of more than 4000 milligrams of acetaminophen per day, even if they feel well.
Rarely, acetaminophen may cause serious skin reactions such as acute generalized exanthematous pustulosis (AGEP), Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. Patients should be informed about the signs of serious skin reactions, and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity.
There have been post-marketing reports of hypersensitivity and anaphylaxis associated with use of acetaminophen. Clinical signs included swelling of the face, mouth, and throat, respiratory distress, urticaria, rash, pruritus, and vomiting. There were infrequent reports of life-threatening anaphylaxis requiring emergency medical attention. Instruct patients to discontinue Hydrocodone Bitartrate and Acetaminophen Tablets, USP immediately and seek medical care if they experience these symptoms. Do not prescribe Hydrocodone Bitartrate and Acetaminophen Tablets, USP for patients with acetaminophen allergy.
Respiratory Depression: At high doses or in sensitive patients, hydrocodone may produce dose-related respiratory depression by acting directly on the brain stem respiratory center. Hydrocodone also affects the center that controls respiratory rhythm, and may produce irregular and periodic breathing.
Head Injury and Increased Intracranial Pressure: The respiratory depressant effects of narcotics and their capacity to elevate cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions or a pre-existing increase in intracranial pressure. Furthermore, narcotics produce adverse reactions which may obscure the clinical course of patients with head injuries.
Acute Abdominal Conditions: The administration of narcotics may obscure the diagnosis or clinical course of patients with acute abdominal conditions.
Special Risk Patients: As with any narcotic analgesic agent, hydrocodone bitartrate and acetaminophen tablets should be used with caution in elderly or debilitated patients, and those with severe impairment of hepatic or renal function, hypothyroidism, Addison's disease, prostatic hypertrophy or urethral stricture. The usual precautions should be observed and the possibility of respiratory depression should be kept in mind.
Cough Reflex: Hydrocodone suppresses the cough reflex; as with all narcotics, caution should be exercised when hydrocodone bitartrate and acetaminophen tablets are used postoperatively and in patients with pulmonary disease.
- Do not take Hydrocodone Bitartrate and Acetaminophen Tablets, USP if you are allergic to any of its ingredients.
- If you develop signs of allergy such as a rash or difficulty breathing stop taking Hydrocodone Bitartrate and Acetaminophen Tablets, USP and contact your healthcare provider immediately.
- Do not take more than 4000 milligrams of acetaminophen per day. Call your doctor if you took more than the recommended dose.
Hydrocodone, like all narcotics, may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks such as driving a car or operating machinery; patients should be cautioned accordingly.
Alcohol and other CNS depressants may produce an additive CNS depression, when taken with this combination product, and should be avoided.
Hydrocodone may be habit-forming. Patients should take the drug only for as long as it is prescribed, in the amounts prescribed, and no more frequently than prescribed.
In patients with severe hepatic or renal disease, effects of therapy should be monitored with serial liver and/or renal function tests.
Patients receiving narcotics, antihistamines, antipsychotics, antianxiety agents, or other CNS depressants (including alcohol) concomitantly with hydrocodone bitartrate and acetaminophen tablets may exhibit an additive CNS depression. When combined therapy is contemplated, the dose of one or both agents should be reduced.
The use of MAO inhibitors or tricyclic antidepressants with hydrocodone preparations may increase the effect of either the antidepressant or hydrocodone.
Acetaminophen may produce false-positive test results for urinary 5-hydroxyindoleacetic acid.
No adequate studies have been conducted in animals to determine whether hydrocodone or acetaminophen have a potential for carcinogenesis, mutagenesis, or impairment of fertility.
Teratogenic Effects: Pregnancy Category C: There are no adequate and well-controlled studies in pregnant women. Hydrocodone bitartrate and acetaminophen tablets should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects: Babies born to mothers who have been taking opioids regularly prior to delivery will be physically dependent. The withdrawal signs include irritability and excessive crying, tremors, hyperactive reflexes, increased respiratory rate, increased stools, sneezing, yawning, vomiting, and fever. The intensity of the syndrome does not always correlate with the duration of maternal opioid use or dose. There is no consensus on the best method of managing withdrawal.
As with all narcotics, administration of this product to the mother shortly before delivery may result in some degree of respiratory depression in the newborn, especially if higher doses are used.
Acetaminophen is excreted in breast milk in small amounts, but the significance of its effects on nursing infants is not known. It is not known whether hydrocodone is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from hydrocodone and acetaminophen, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Safety and effectiveness in pediatric patients have not been established.
Clinical studies of hydrocodone bitartrate 5 mg and acetaminophen 500 mg did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Hydrocodone and the major metabolites of acetaminophen are known to be substantially excreted by the kidney. Thus the risk of toxic reactions may be greater in patients with impaired renal function due to the accumulation of the parent compound and/or metabolites in the plasma. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Hydrocodone may cause confusion and over-sedation in the elderly; elderly patients generally should be started on low doses of hydrocodone bitartrate and acetaminophen tablets and observed closely.
The most frequently reported adverse reactions are lightheadedness, dizziness, sedation, nausea, and vomiting. These effects seem to be more prominent in ambulatory than in nonambulatory patients, and some of these adverse reactions may be alleviated if the patient lies down.
Other adverse reactions include:
Central Nervous System: Drowsiness, mental clouding, lethargy, impairment of mental and physical performance, anxiety, fear, dysphoria, psychic dependence, and mood changes.
Gastrointestinal System: Prolonged administration of hydrocodone bitartrate and acetaminophen tablets may produce constipation.
Genitourinary System: Ureteral spasm, spasm of vesical sphincters, and urinary retention have been reported with opiates.
Respiratory Depression: Hydrocodone bitartrate may produce dose-related respiratory depression by acting directly on the brain stem respiratory center (see
OVERDOSAGE
).
Special Senses: Cases of hearing impairment or permanent loss have been reported predominantly in patients with chronic overdose.
Dermatological: Skin rash, pruritus.
The following adverse drug events may be borne in mind as potential effects of acetaminophen: allergic reactions, rash, thrombocytopenia, agranulocytosis.Potential effects of high dosage are listed in the
OVERDOSAGE
section.
Controlled Substance: Hydrocodone Bitartrate and Acetaminophen Tablets are classified as a Schedule III controlled substance.
Abuse and Dependence: Psychic dependence, physical dependence, and tolerance may develop upon repeated administration of narcotics; therefore, this product should be prescribed and administered with caution. However, psychic dependence is unlikely to develop when hydrocodone bitartrate and acetaminophen tablets are used for a short time for the treatment of pain.
Physical dependence, the condition in which continued administration of the drug is required to prevent the appearance of a withdrawal syndrome, assumes clinically significant proportions only after several weeks of continued narcotic use, although some mild degree of physical dependence may develop after a few days of narcotic therapy. Tolerance, in which increasingly large doses are required in order to produce the same degree of analgesia, is manifested initially by a shortened duration of analgesic effect, and subsequently by decreases in the intensity of analgesia. The rate of development of tolerance varies among patients.
Following an acute overdosage, toxicity may result from hydrocodone or acetaminophen.
Hydrocodone: Serious overdose with hydrocodone is characterized by respiratory depression (a decrease in respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis), extreme somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, and sometimes bradycardia and hypotension. In severe overdosage, apnea, circulatory collapse, cardiac arrest and death may occur.
Acetaminophen: In acetaminophen overdosage: dose-dependent, potentially fatal hepatic necrosis is the most serious adverse effect. Renal tubular necrosis, hypoglycemic coma and coagulation defects may also occur. Early symptoms following a potentially hepatotoxic overdose may include: nausea, vomiting, diaphoresis and general malaise. Clinical and laboratory evidence of hepatic toxicity may not be apparent until 48 to 72 hours post-ingestion.
Treatment: A single or multiple drug overdose with hydrocodone and acetaminophen is a potentially lethal polydrug overdose, and consultation with a regional poison control center is recommended. Immediate treatment includes support of cardiorespiratory function and measures to reduce drug absorption. Oxygen, intravenous fluids, vasopressors, and other supportive measures should be employed as indicated. Assisted or controlled ventilation should also be considered.
For hydrocodone overdose, primary attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and the institution of assisted or controlled ventilation. The narcotic antagonist naloxone hydrochloride is a specific antidote against respiratory depression which may result from overdosage or unusual sensitivity to narcotics, including hydrocodone. Since the duration of action of hydrocodone may exceed that of the antagonist, the patient should be kept under continued surveillance, and repeated doses of the antagonist should be administered as needed to maintain adequate respiration. A narcotic antagonist should not be administered in the absence of clinically significant respiratory or cardiovascular depression.
Gastric decontamination with activated charcoal should be administered just prior to N-acetylcysteine (NAC) to decrease systemic absorption if acetaminophen ingestion is known or suspected to have occurred within a few hours of presentation. Serum acetaminophen levels should be obtained immediately if the patient presents 4 hours or more after ingestion to assess potential risk of hepatotoxicity; acetaminophen levels drawn less than 4 hours post-ingestion may be misleading. To obtain the best possible outcome, NAC should be administered as soon as possible where impending or evolving liver injury is suspected. Intravenous NAC may be administered when circumstances preclude oral administration.
Vigorous supportive therapy is required in severe intoxication. Procedures to limit the continuing absorption of the drug must be readily performed since the hepatic injury is dose dependent and occurs early in the course of intoxication.
Dosage should be adjusted according to the severity of the pain and the response of the patient. However, it should be kept in mind that tolerance to hydrocodone can develop with continued use and that the incidence of untoward effects is dose related.
2.5 mg/500 mg
5 mg/500 mg
|
The usual adult dosage is one or two tablets every four to six hours as needed for pain. The total daily dosage should not exceed 8 tablets.
|
7.5 mg/325 mg
7.5 mg/500 mg
7.5 mg/650 mg
|
The usual adult dosage is one tablet every four to six hours as needed for pain. The total daily dosage should not exceed 6 tablets.
|
7.5 mg/750 mg
|
The usual adult dosage is one tablet every four to six hours as needed for pain. The total daily dosage should not exceed 5 tablets.
|
10 mg/325 mg
10 mg/500 mg
10 mg/650 mg
10 mg/660 mg
|
The usual adult dosage is one tablet every four to six hours as needed for pain. The total daily dosage should not exceed 6 tablets.
|
10 mg/750 mg
|
The usual adult dosage is one tablet every four to six hours as needed for pain. The total daily dosage should not exceed 5 tablets.
|
Hydrocodone Bitartrate and Acetaminophen Tablets USP are available in the following strengths:
2.5 mg/500 mg
|
2.5 mg hydrocodone bitartrate and 500 mg acetaminophen, oblong, white tablets bisected on one side and debossed with WATSON 388 on the other side, supplied in bottles of 100.
|
5 mg/500 mg
|
5 mg hydrocodone bitartrate and 500 mg acetaminophen, capsule-shaped, white tablets bisected on one side and debossed with WATSON 349 on the other side, supplied in bottles of 100 and 500.
|
7.5 mg/325 mg
|
7.5 mg hydrocodone bitartrate and 325 mg acetaminophen, capsule- shaped, white tablets bisected on one side and debossed with WATSON 3203 on the other side, supplied in bottles of 100.
|
7.5 mg/500 mg
|
7.5 mg hydrocodone bitartrate and 500 mg acetaminophen, capsule- shaped, white tablets bisected on one side and debossed with WATSON 385 on the other side, supplied in bottles of 100 and 500.
|
7.5 mg/650 mg
|
7.5 mg hydrocodone bitartrate and 650 mg acetaminophen, capsule- shaped, white tablets bisected on one side and debossed with WATSON 502 on the other side, supplied in bottles of 100 and 500.
|
7.5 mg/750 mg
|
7.5 mg hydrocodone bitartrate and 750 mg acetaminophen, oblong, white tablets bisected on one side and debossed with WATSON 387 on the other side, supplied in bottles of 100 and 500.
|
10 mg/325 mg
|
10 mg hydrocodone bitartrate and 325 mg acetaminophen, capsule-shaped, white tablets bisected on one side and debossed with WATSON 853 on the other side, supplied in bottles of 100 and 500.
|
10 mg/500 mg
|
10 mg hydrocodone bitartrate and 500 mg acetaminophen, capsule-shaped, white tablets bisected on one side and debossed with WATSON 540 on the other side, supplied in bottles of 100 and 500.
|
10 mg/650 mg
|
10 mg hydrocodone bitartrate and 650 mg acetaminophen, capsule-shaped, white tablets bisected on one side and debossed with WATSON 503 on the other side, supplied in bottles of 100 and 500.
|
10 mg/660 mg
|
10 mg hydrocodone bitartrate and 660 mg acetaminophen, oval-shaped, white tablets bisected on one side and debossed with WATSON 517 on the other side, supplied in bottles of 100 and 500.
|
10 mg/750 mg
|
10 mg hydrocodone bitartrate and 750 mg acetaminophen, capsule-shaped, white tablets bisected on one side and debossed with WATSON 3228 on the other side, supplied in bottles of 100. |
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container with a child-resistant closure.
Manufactured by:
Watson Laboratories, Inc.
Corona, CA 92880 USA
Distributed by:
Watson Pharma, Inc.
Parsippany, NJ 07054 USA
Revised: November 2013 173191-2
NDC 0591-0388-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 2.5 mg/500 mg
Watson 100 Tablets Rx only

NDC 0591-0349-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 5 mg/500 mg
Watson 100 Tablets Rx only

NDC 0591-2605-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 7.5 mg/325 mg
Watson 100 Tablets Rx only

NDC 0591-0385-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 7.5 mg/500 mg
Watson 100 Tablets Rx only

NDC 0591-2611-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 7.5 mg/650 mg
Watson 100 Tablets Rx only

NDC 0591-0387-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 7.5 mg/750 mg
Watson 100 Tablets Rx only

NDC 0591-2612-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 10 mg/ 325 mg
Watson 100 Tablets Rx only

NDC 0591-2609-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 10 mg/ 500 mg
Watson 100 Tablets Rx only

NDC 0591-2610-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 10 mg/650 mg
Watson 100 Tablets Rx only

NDC 0591-0517-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 10 mg/660 mg
Watson 100 Tablets Rx only

NDC 0591-2607-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 10 mg/750 mg
Watson 100 Tablets Rx only

NDC 0591-3203-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 7.5 mg/325 mg
Watson 100 Tablets Rx only

NDC 0591-0502-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 7.5 mg/650 mg
Watson 100 Tablets Rx only

NDC 0591-0853-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 10 mg/ 325 mg
Watson 100 Tablets Rx only

NDC 0591-0540-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 10 mg/ 500 mg
Watson 100 Tablets Rx only

NDC 0591-0503-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 10 mg/650 mg
Watson 100 Tablets Rx only

NDC 0591-3228-01
Hydrocodone Bitartrate and Acetaminophen Tablets USP
CIII 10 mg/750 mg
Watson 100 Tablets Rx only

Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-2609 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
15 mm |
WATSON;540 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-2609-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-2609-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040148 |
2013-01-04 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-2612 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
14 mm |
WATSON;853 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-2612-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-2612-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040148 |
2013-03-07 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-2610 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
15 mm |
WATSON;503 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-2610-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-2610-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040094 |
2013-03-14 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-2605 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
14 mm |
WATSON;3203 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-2605-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-2605-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040148 |
2013-03-14 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-2611 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
16 mm |
WATSON;502 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-2611-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-2611-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040094 |
2013-04-15 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-2607 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
19 mm |
WATSON;3228 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-2607-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040094 |
2013-04-23 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-0388 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
15 mm |
WATSON;388 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-0388-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA081079 |
1991-08-30 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-0349 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
18 mm |
WATSON;349 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-0349-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-0349-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA089883 |
1988-12-01 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-3203 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
ORANGE (light orange) |
14 mm |
WATSON;3203 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-3203-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040148 |
2000-09-12 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-0385 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
14 mm |
WATSON;385 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-0385-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-0385-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA081080 |
1991-08-30 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-0502 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
PINK |
16 mm |
WATSON;502 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-0502-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-0502-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040094 |
1995-09-29 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-0387 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
18 mm |
WATSON;387 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-0387-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-0387-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA081083 |
1991-08-30 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-0853 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
YELLOW |
14 mm |
WATSON;853 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-0853-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-0853-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040148 |
1997-02-14 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-0540 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
BLUE |
15 mm |
WATSON;540 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-0540-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-0540-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040148 |
1997-02-14 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-0503 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
GREEN (light green) |
15 mm |
WATSON;503 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-0503-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-0503-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040094 |
1995-09-29 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-0517 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
WHITE |
18 mm |
WATSON;517 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-0517-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
|
2 |
NDC:0591-0517-05 |
500 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040094 |
2000-08-08 |
|
|
Hydrocodone Bitartrate and Acetaminophen
Hydrocodone Bitartrate and Acetaminophen TABLET
Product Information
|
|
Product Type
|
Human prescription drug label |
Item Code (Source)
|
NDC:0591-3228 |
|
Route of Administration
|
ORAL |
DEA Schedule
|
|
Product Characteristics
|
|
Color
|
Size
|
Imprint Code
|
Shape
|
|
YELLOW |
19 mm |
WATSON;3228 |
OVAL |
Packaging
|
|
#
|
Item Code
|
Package Description
|
Marketing Start Date
|
Marketing End Date
|
|
1 |
NDC:0591-3228-01 |
100 in 1 BOTTLE, PLASTIC |
|
|
Marketing Information
|
|
Marketing Category
|
Application Number or Monograph Citation
|
Marketing Start Date
|
Marketing End Date
|
|
ANDA |
ANDA040094 |
1999-03-22 |
|
|


















