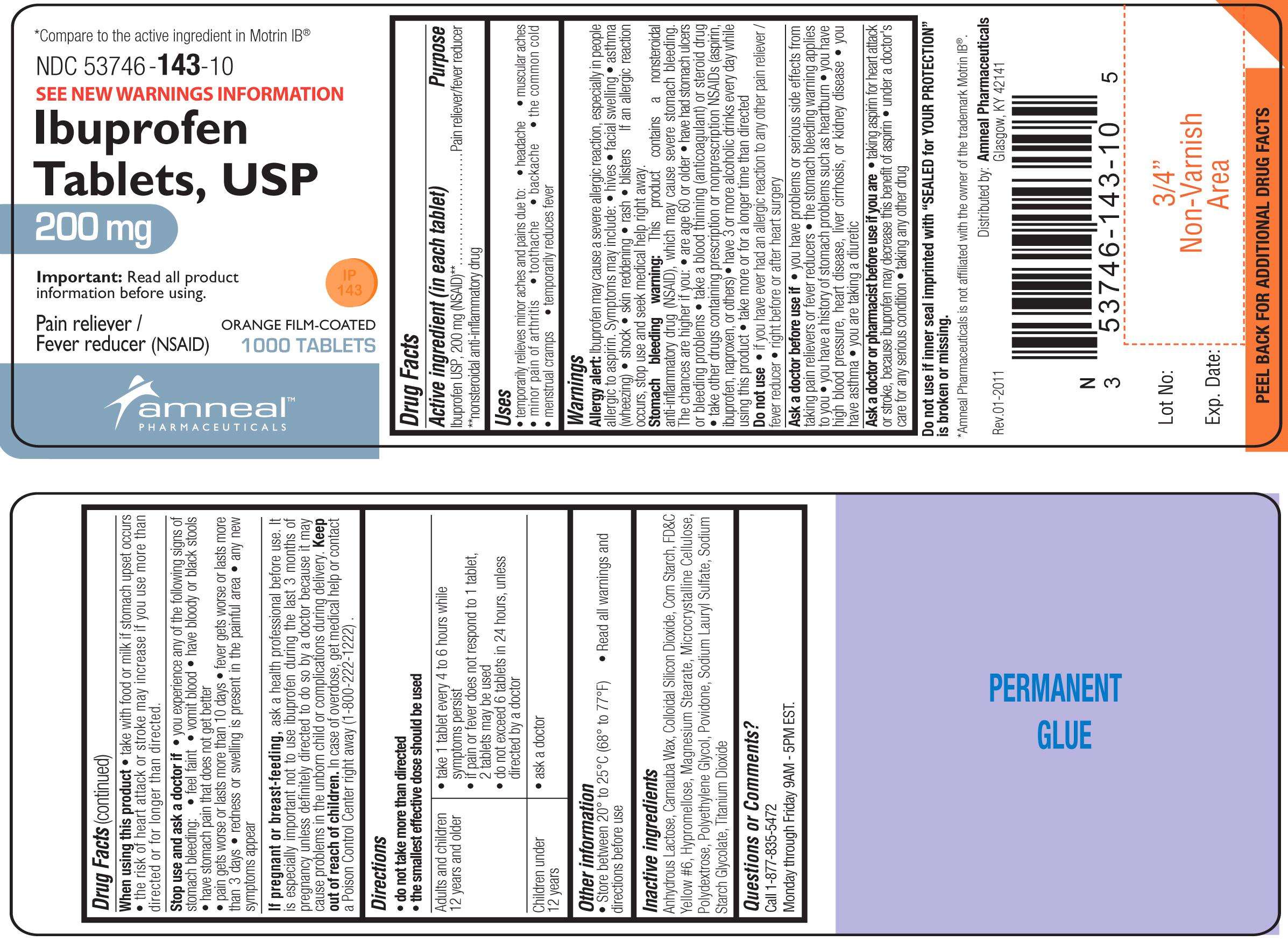
Ibuprofen (NSAID)
Amneal Pharmaceuticals
Amneal Pharmaceuticals
Ibuprofen Tablets, USP Fever reducer/ Pain Reliever (NSAID)
FULL PRESCRIBING INFORMATION: CONTENTS*
- OTC - DO NOT USE
- OTC - ASK DOCTOR
- OTC - ASK DOCTOR/PHARMACIST
- OTC - WHEN USING
- OTC - STOP USE
- PREGNANCY OR BREAST FEEDING
FULL PRESCRIBING INFORMATION
(in each tablet)
Ibuprofen USP, 200 mg (NSAID)**
**nonsteroidal anti-inflammatory drug
Pain reliever/fever reducer
- temporarily relieves minor aches and pains due to:
- headache
- muscular aches
- minor pain of arthritis
- toothache
- backache
- the common cold
- menstrual cramps
- temporarily reduces fever
Allergy alert: Ibuprofen may cause a severe allergic reaction, especially in people allergic to aspirin. Symptoms may include:
- hives
- facial swelling
- asthma(wheezing)
- shock
- skin reddening
- rash
- blisters
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning: This product contains a nonsteroidal anti-inflammatory drug (NSAID), which may cause severe stomach bleeding. The chances are higher if you:
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
OTC - DO NOT USE
Do not use
- if you have ever had an allergic reaction to any other pain reliever/fever reducer
- right before or after heart surgery
OTC - ASK DOCTOR
Ask a doctor before use if
- you have problems or serious side effects from taking pain relievers or fever reducers
- the stomach bleeding warning applies to you
- you have a history of stomach problems such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
- you have asthma
- you are taking a diuretic
OTC - ASK DOCTOR/PHARMACIST
Ask a doctor or pharmacist before use if you are
- taking aspirin for heart attack or stroke, because ibuprofen may decrease this benefit of aspirin
- under a doctor's care for any serious condition
- taking any other drug
OTC - WHEN USING
When using this product
- take with food or milk if stomach upset occurs
- the risk of heart attack or stroke may increase if you use more than directed or for longer than directed
OTC - STOP USE
Stop use and ask a doctor if
- you experience any of the following signs of stomach bleeding
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present in the painful area
- any new symptoms appear
PREGNANCY OR BREAST FEEDING
If pregnant or breast-feeding, ask a health professional before use. It is especially important not to use ibuprofen during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
- do not take more than directed
- the smallest effective dose should be used
| Adults and children 12 years and older |
|
| Children under 12 years |
|
- Store between 20° to 25°C (68° to 77°F)
- Read all warnings and directions before use
-
-
-
Brown Tablets: Anhydrous Lactose, Carnauba Wax, Colloidal Silicon Dioxide, Hypromellose, Iron Oxide Red, Magnesium Stearate, Microcrystalline Cellulose, Polydextrose, Polyethylene Glycol, Povidone, Sodium Lauryl Sulfate, Sodium Starch Glycolate, Corn Starch Corn, Titanium Dioxide
-
Orange Tablets: Anhydrous Lactose, Carnauba Wax, Colloidal Silicon Dioxide, FD&C Yellow #6, Hypromellose, Magnesium Stearate, Microcrystalline Cellulose, Polydextrose, Polyethylene Glycol, Povidone, Sodium Lauryl Sulfate, Sodium Starch Glycolate, Corn Starch, Titanium Dioxide
-
-
Call 1-877-835-5472
Monday through Friday 9AM - 5PM EST.
*Amneal Pharmaceuticals is not affiliated with the owner of the trademark Advil®
Distributed By:
Ameal Pharmaceuticals
Glasgow, KY 42141

Ibuprofen (NSAID)Ibuprofen (NSAID) TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ibuprofen (NSAID)Ibuprofen (NSAID) TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||