LAMISIL AT
Novartis Consumer Health, Inc.
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient
- Purpose
- LAMISIL AT Uses
- Warnings
- Do not use
- Ask Doctor before use if you have
- Ask a doctor or pharmacist
- When using this product
- Stop use and ask a doctor
- Keep Out of Reach of Children
- Directions
- LAMISIL AT Other information
- Inactive ingredients
- Questions
- Principal Display Panel
FULL PRESCRIBING INFORMATION
Active ingredient
Terbinafine1%
Purpose
Antifungal
LAMISIL AT Uses
- cures most athlete’s foot (tinea pedis) between the toes.
Effectiveness on the bottom or sides of foot is unknown.
- cures most jock itch (tinea cruris) and ringworm (tinea corporis)
- relieves itching, burning, cracking and scaling which accompany these conditions
Warnings
For external use only
Do not use
- on nails or scalp
- in or near the mouth or eyes
- for vaginal yeast infections
Ask Doctor before use if you have
Ask a doctor or pharmacist
When using this product
do not get into eyes. If eye contact occurs, rinse thoroughly with water.
Stop use and ask a doctor
do not get into eyes. If eye contact occurs, rinse thoroughly with water.
Keep Out of Reach of Children
If swallowed, get medical help or contact a poison control center right away.
Directions
- adults and children 12 years and over
- use the tip of the cap to break the seal and open the tube
- wash the affected skin with soap and water and dry completely before applying
- for athlete’s foot between the toes: apply once a day at bedtime for 1 week or as directed by a doctor. Wear well-fitting, ventilated shoes. Change shoes and socks at least once daily
-
 1 week between the toes
1 week between the toes - for jock itch and ringworm: apply once a day (morning or night) for 1 week or as directed by a doctor.
- wash hands after each use
- children under 12 years: ask a doctor
LAMISIL AT Other information
- do not use if seal on tube is broken or is not visible
- store at or below 30°C (86°F)
Inactive ingredients
benzyl alcohol, butylated hydroxytoluene, carbomer 974 P, ethanol, isopropyl myristate, polysorbate 20, purified water, sodium hydroxide, sorbitan monolaurate
Questions
Questions? call 1-800-452-0051
Distributed by:
Novartis Consumer Health, Inc.
Parsippany, NJ 07054-0622
Principal Display Panel
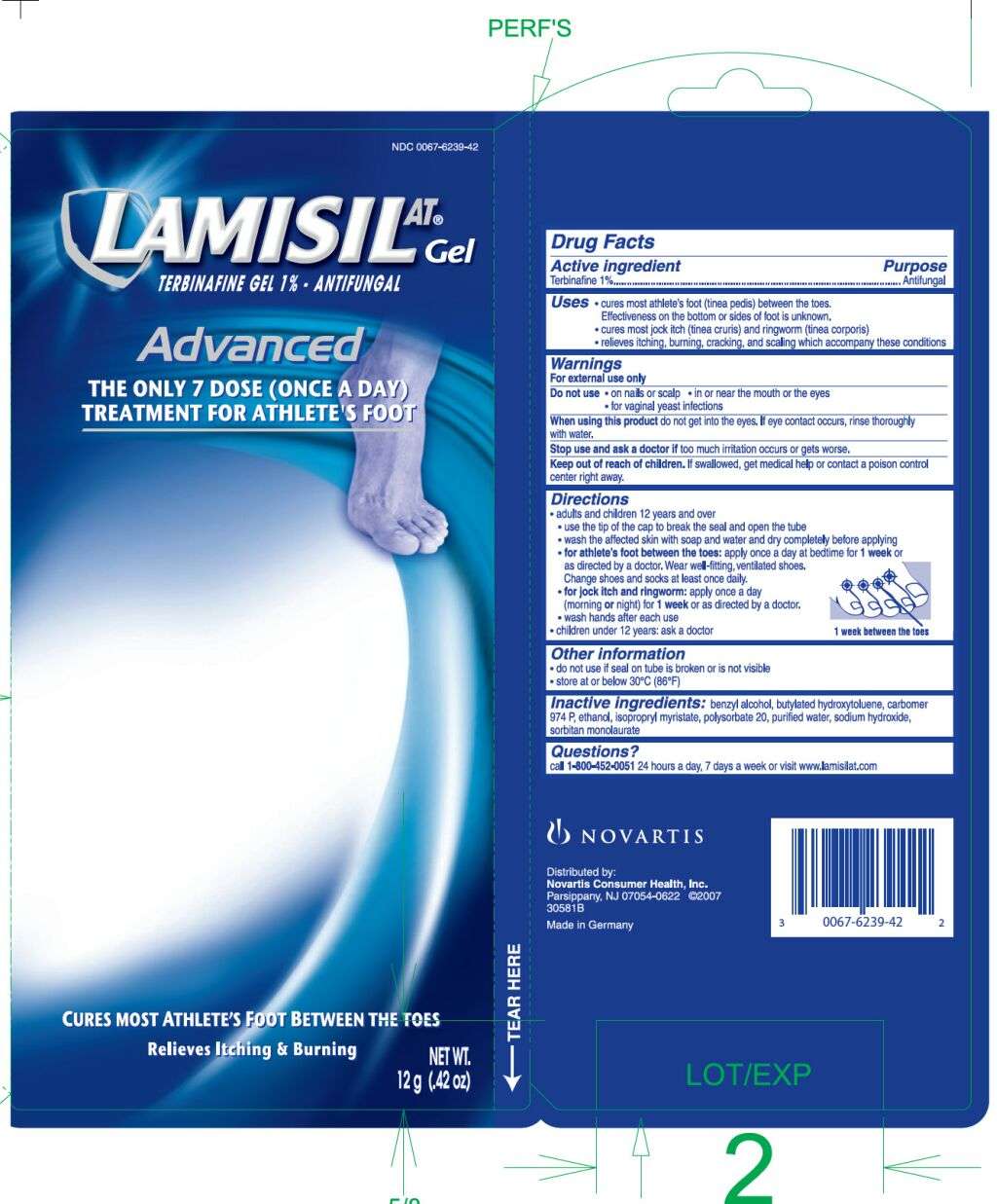
LAMISIL ATTERBINAFINE GEL
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||