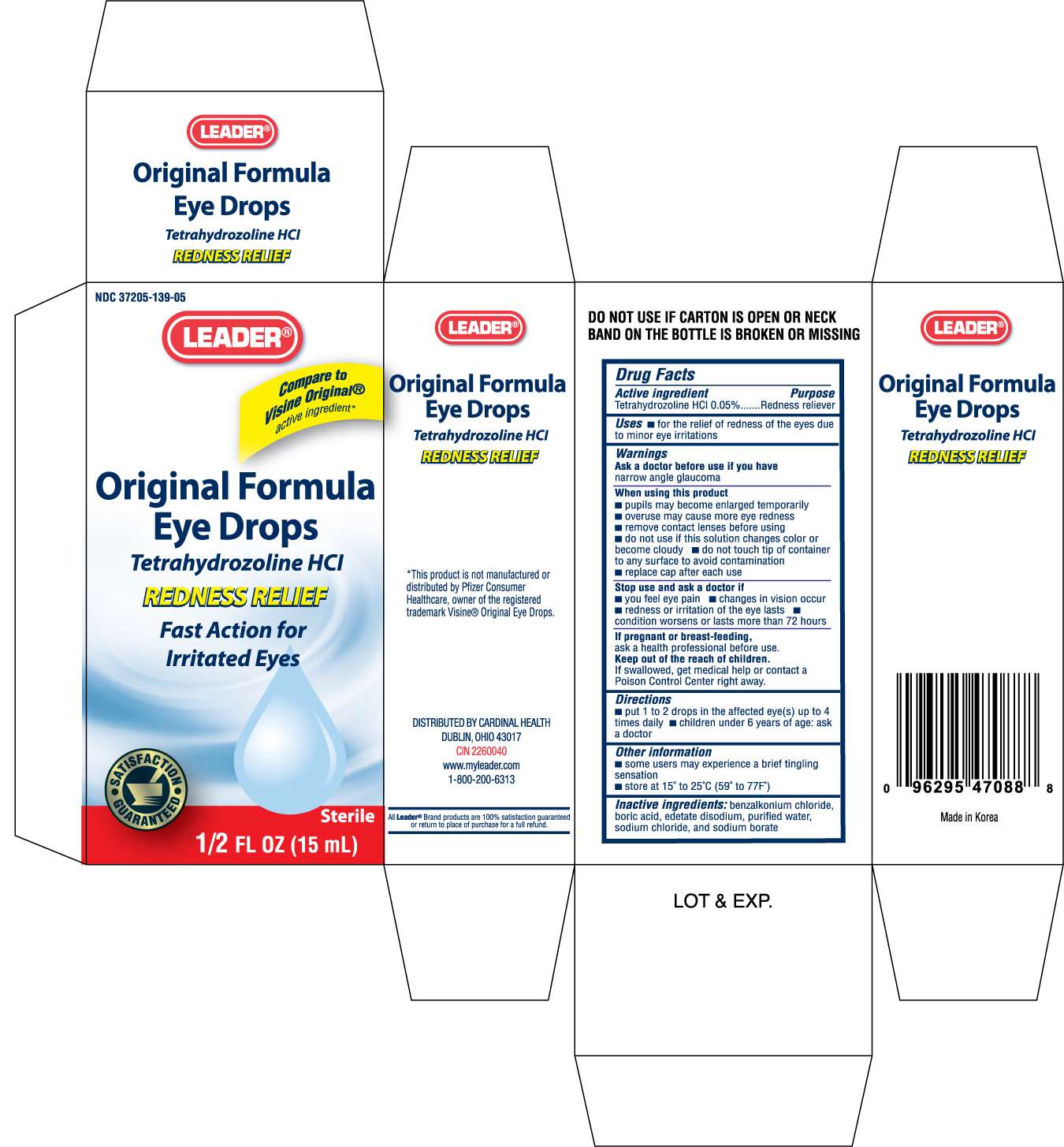
LEADER ORIGINAL FORMULA
Drug Facts
FULL PRESCRIBING INFORMATION
Active ingredient
Active ingredient
Purpose
Uses
For the relief of redness of the eyes due to minor eye irritations.
Warnings
Ask a doctor before use if you have narrow angle glaucoma
When Using this product
- pupils may become enlarged temporarily
- overuse may cause more eye redness
- remove contact lenses before using
- do not use if this solution changes color or become cloudy
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
Stop use and ask a doctor if
- you feel eye pain
- changes in vision occur
- redness or irritation of the eye lasts
- condition worsens or lasts more than 72 hours
If pregnant or breast-feeding
ask a health professional before use
Keep out of the reach of children
If swallowed, get medical help or contact a Poison Control Center right away
Uses
Directions
- put 1 to 2 drops in the affected eye(s) up to 4 times daily
- children under 6 years of age: ask a doctor
Other information
some users may experience a brief tingling sensation
store at 15 to 25C (59 to 77F)
Inactive ingredients:
benzalkonium chloride, boric acid, edetate disodium, purified
water, sodium chloride, and sodium borate
DISTRIBUTED BY CARDINAL HEALTH
DUBLIN, OHIO 43017
CIN2260040
www.myleader.com
1-800-200-6313
 Enter section text here
Enter section text here
LEADER ORIGINAL FORMULATETRAHYDROZOLINE HYDROCHLORIDE SOLUTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||