LEXIVA
These highlights do not include all the information needed to use LEXIVA safely and effectively. See full prescribing information for LEXIVA.LEXIVA (fosamprenavir calcium) Tablets, for oral useLEXIVA (fosamprenavir calcium) Oral SuspensionInitial U.S. Approval: 2003
FULL PRESCRIBING INFORMATION: CONTENTS*
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
- DOSAGE FORMS & STRENGTHS
- LEXIVA CONTRAINDICATIONS
- WARNINGS AND PRECAUTIONS
- LEXIVA ADVERSE REACTIONS
- DRUG INTERACTIONS
- USE IN SPECIFIC POPULATIONS
- OVERDOSAGE
- LEXIVA DESCRIPTION
- CLINICAL PHARMACOLOGY
- NONCLINICAL TOXICOLOGY
- CLINICAL STUDIES
- HOW SUPPLIED
- INFORMATION FOR PATIENTS
- SPL PATIENT PACKAGE INSERT
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
INDICATIONS & USAGE
LEXIVA® is indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) infection.
The following points should be considered when initiating therapy with LEXIVA plus ritonavir in protease inhibitor-experienced patients:
•The protease inhibitor-experienced patient trial was not large enough to reach a definitive conclusion that LEXIVA plus ritonavir and lopinavir plus ritonavir are clinically equivalent
[see Clinical Studies (14.2)]
.•Once-daily administration of LEXIVA plus ritonavir is not recommended for adult protease inhibitor-experienced patients or any pediatric patients
[see Dosage and Administration (2.1, 2.2), Clinical Studies (14.2, 14.3)]
.•Dosing of LEXIVA plus ritonavir is not recommended for protease inhibitor-experienced pediatric patients younger than 6 months
[see Clinical Pharmacology (12.3)].
DOSAGE & ADMINISTRATION
LEXIVA Tablets may be taken with or without food.
Adults should take LEXIVA Oral Suspension without food. Pediatric patients should take LEXIVA Oral Suspension with food [see Clinical Pharmacology (12.3)]. If emesis occurs within 30 minutes after dosing, re-dosing of LEXIVA Oral Suspension should occur.
Higher-than-approved dose combinations of LEXIVA plus ritonavir are not recommended due to an increased risk of transaminase elevations [see Overdosage (10)].
When LEXIVA is used in combination with ritonavir, prescribers should consult the full prescribing information for ritonavir.
Therapy-Naive Adults:
•LEXIVA 1,400 mg twice daily (without ritonavir).•LEXIVA 1,400 mg once daily plus ritonavir 200 mg once daily.•LEXIVA 1,400 mg once daily plus ritonavir 100 mg once daily.∘Dosing of LEXIVA 1,400 mg once daily plus ritonavir 100 mg once daily is supported by pharmacokinetic data
[see Clinical Pharmacology (12.3)]
.•LEXIVA 700 mg twice daily plus ritonavir 100 mg twice daily.∘Dosing of LEXIVA 700 mg twice daily plus 100 mg ritonavir twice daily is supported by pharmacokinetic and safety data
[see Clinical Pharmacology (12.3)]
.
Protease Inhibitor-Experienced Adults:
•LEXIVA 700 mg twice daily plus ritonavir 100 mg twice daily.
The recommended dosage of LEXIVA in patients aged at least 4 weeks to 18 years should be calculated based on body weight (kg) and should not exceed the recommended adult dose (Table 1).
Table 1. Twice-Daily Dosage Regimens by Weight for Protease Inhibitor-Naive Pediatric Patients (Greater Than or Equal to 4 Weeks of Age) and for Protease Inhibitor-Experienced Pediatric Patients (Greater Than or Equal to 6 Months of Age) Using LEXIVA Oral Suspension With Concurrent Ritonavir
|
Weight |
Twice-Daily Dosage Regimen |
|
<11 kg |
LEXIVA 45 mg/kg plus ritonavir 7 mg/kga |
|
11 kg - <15 kg |
LEXIVA 30 mg/kg plus ritonavir 3 mg/kga |
|
15 kg - <20 kg |
LEXIVA 23 mg/kg plus ritonavir 3 mg/kga |
|
≥20 kg |
LEXIVA 18 mg/kg plus ritonavir 3 mg/kga |
aWhen dosing with ritonavir, do not exceed the adult dose of LEXIVA 700 mg/
ritonavir 100 mg twice-daily dose.
Alternatively, protease inhibitor-naive children aged 2 years and older can be administered LEXIVA (without ritonavir) 30 mg per kg twice daily.
LEXIVA should only be administered to infants born at 38 weeks gestation or greater and who have attained a post-natal age of 28 days.
For pediatric patients, pharmacokinetic and clinical data:
•do not support once-daily dosing of LEXIVA alone or in combination with ritonavir
[see Clinical Studies (14.3)]
.•do not support administration of LEXIVA alone or in combination with ritonavir for protease inhibitor‑experienced children younger than 6 months
[see Clinical Pharmacology (12.3)]
.•do not support twice-daily dosing of LEXIVA without ritonavir in pediatric patients younger than 2 years
[see Clinical Pharmacology (12.3)].
Other Dosing Considerations:
•When administered without ritonavir, the adult regimen of LEXIVA Tablets 1,400 mg twice daily may be used for pediatric patients weighing at least 47 kg.•When administered in combination with ritonavir, LEXIVA Tablets may be used for pediatric patients weighing at least 39 kg; ritonavir capsules may be used for pediatric patients weighing at least 33 kg.
See Clinical Pharmacology (12.3).
Mild Hepatic Impairment (Child-Pugh Score Ranging From 5 to 6):LEXIVA should be used with caution at a reduced dosage of 700 mg twice daily without ritonavir (therapy-naive) or 700 mg twice daily plus ritonavir 100 mg once daily (therapy-naive or protease inhibitor-experienced).
Moderate Hepatic Impairment (Child-Pugh Score Ranging From 7 to 9):LEXIVA should be used with caution at a reduced dosage of 700 mg twice daily without ritonavir (therapy-naive), or 450 mg twice daily plus ritonavir 100 mg once daily (therapy-naive or protease inhibitor-experienced).
Severe Hepatic Impairment (Child-Pugh Score Ranging From 10 to 15):LEXIVA should be used with caution at a reduced dosage of 350 mg twice daily without ritonavir (therapy-naive) or 300 mg twice daily plus ritonavir 100 mg once daily (therapy-naive or protease inhibitor-experienced).
There are no data to support dosing recommendations for pediatric patients with hepatic impairment.
DOSAGE FORMS & STRENGTHS
LEXIVA Tablets, 700 mg, are pink, film-coated, capsule-shaped, biconvex tablets with “GX LL7” debossed on one face.
LEXIVA Oral Suspension, 50 mg per mL, is a white to off-white suspension that has a characteristic grape-bubblegum-peppermint flavor.
LEXIVA CONTRAINDICATIONS
LEXIVA is contraindicated:
•in patients with previously demonstrated clinically significant hypersensitivity (e.g., Stevens-Johnson syndrome) to any of the components of this product or to amprenavir.•when coadministered with drugs that are highly dependent on cytochrome P450 3A4 (CYP3A4) for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events (Table 2).
|
Drug Class/Drug Name |
Clinical Comment |
|
Alpha 1-adrenoreceptor antagonist: Alfuzosin |
Potentially increased alfuzosin concentrations can result in hypotension. |
|
Antiarrhythmics: Flecainide, propafenone |
POTENTIAL for serious and/or life-threatening reactions such as cardiac arrhythmias secondary to increases in plasma concentrations of antiarrhythmics if LEXIVA is co-prescribed with ritonavir. |
|
Antimycobacterials: Rifampina |
May lead to loss of virologic response and possible resistance to LEXIVA or to the class of protease inhibitors. |
|
Ergot derivatives: Dihydroergotamine, ergonovine, ergotamine, methylergonovine |
POTENTIAL for serious and/or life-threatening reactions such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. |
|
GI motility agents: Cisapride |
POTENTIAL for serious and/or life-threatening reactions such as cardiac arrhythmias. |
|
Herbal products: St. John’s wort (Hypericum perforatum) |
May lead to loss of virologic response and possible resistance to LEXIVA or to the class of protease inhibitors. |
|
HMG co-reductase inhibitors: Lovastatin, simvastatin |
POTENTIAL for serious reactions such as risk of myopathy including rhabdomyolysis. |
|
Neuroleptic: Pimozide |
POTENTIAL for serious and/or life-threatening reactions such as cardiac arrhythmias. |
|
Non-nucleoside reverse transcriptase inhibitor: Delavirdinea |
May lead to loss of virologic response and possible resistance to delavirdine. |
|
PDE5 inhibitor: Sildenafil (REVATIO®) (for treatment of pulmonary arterial hypertension) |
A safe and effective dose has not been established when used with LEXIVA. There is increased potential for sildenafil-associated adverse events (which include visual disturbances, hypotension, prolonged erection, and syncope). |
|
Sedative/hypnotics: Midazolam, triazolam |
POTENTIAL for serious and/or life-threatening reactions such as prolonged or increased sedation or respiratory depression. |
a See Clinical Pharmacology (12.3) Tables 10, 11, 12, or 13 for magnitude of interaction.
•when coadministered with ritonavir in patients receiving the antiarrhythmic agents, flecainide and propafenone. If LEXIVA is coadministered with ritonavir, reference should be made to the full prescribing information for ritonavir for additional contraindications.
WARNINGS AND PRECAUTIONS
•Certain drugs should not be coadministered with LEXIVA due to risk of serious or life-threatening adverse reactions. (5.1) •LEXIVA should be discontinued for severe skin reactions including Stevens-Johnson syndrome. (5.2) •LEXIVA should be used with caution in patients with a known sulfonamide allergy. (5.3)•Use of higher than approved doses may lead to transaminase elevations. Patients with hepatitis B or C are at increased risk of transaminase elevations. (5.4)•Patients receiving LEXIVA may develop new onset or exacerbations of diabetes mellitus, hyperglycemia (5.5), immune reconstitution syndrome (5.6), redistribution/accumulation of body fat (5.7), and elevated triglyceride and cholesterol concentrations (5.8). Monitor cholesterol and triglycerides prior to therapy and periodically thereafter.•Acute hemolytic anemia has been reported with amprenavir. (5.9)•Hemophilia: Spontaneous bleeding may occur, and additional factor VIII may be required. (5.10)•Nephrolithiasis: Cases of nephrolithiasis have been reported with fosamprenavir. (5.11)
See Table 2 for listings of drugs that are contraindicated due to potentially life-threatening adverse events, significant drug interactions, or loss of virologic activity [see Contraindications (4), Drug Interactions (7.2)]. See Table 7 for a listing of established and other potentially significant drug interactions [see Drug Interactions (7.3)].
Severe and life-threatening skin reactions, including 1 case of Stevens-Johnson syndrome among 700 subjects treated with LEXIVA in clinical trials. Treatment with LEXIVA should be discontinued for severe or life-threatening rashes and for moderate rashes accompanied by systemic symptoms [see Adverse Reactions (6)].
LEXIVA should be used with caution in patients with a known sulfonamide allergy. Fosamprenavir contains a sulfonamide moiety. The potential for cross-sensitivity between drugs in the sulfonamide class and fosamprenavir is unknown. In a clinical trial of LEXIVA used as the sole protease inhibitor, rash occurred in 2 of 10 subjects (20%) with a history of sulfonamide allergy compared with 42 of 126 subjects (33%) with no history of sulfonamide allergy. In 2 clinical trials of LEXIVA plus low-dose ritonavir, rash occurred in 8 of 50 subjects (16%) with a history of sulfonamide allergy compared with 50 of 412 subjects (12%) with no history of sulfonamide allergy.
Use of LEXIVA with ritonavir at higher-than-recommended dosages may result in transaminase elevations and should not be used [see Dosage and Administration (2), Overdosage (10)]. Patients with underlying hepatitis B or C or marked elevations in transaminases prior to treatment may be at increased risk for developing or worsening of transaminase elevations. Appropriate laboratory testing should be conducted prior to initiating therapy with LEXIVA and patients should be monitored closely during treatment.
New onset diabetes mellitus, exacerbation of pre-existing diabetes mellitus, and hyperglycemia have been reported during postmarketing surveillance in HIV-1-infected patients receiving protease inhibitor therapy. Some patients required either initiation or dose adjustments of insulin or oral hypoglycemic agents for treatment of these events. In some cases, diabetic ketoacidosis has occurred. In those patients who discontinued protease inhibitor therapy, hyperglycemia persisted in some cases. Because these events have been reported voluntarily during clinical practice, estimates of frequency cannot be made and causal relationships between protease inhibitor therapy and these events have not been established.
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including LEXIVA. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves’ disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and “cushingoid appearance,” have been observed in patients receiving antiretroviral therapy, including LEXIVA. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
Treatment with LEXIVA plus ritonavir has resulted in increases in the concentration of triglycerides and cholesterol [see Adverse Reactions (6)]. Triglyceride and cholesterol testing should be performed prior to initiating therapy with LEXIVA and at periodic intervals during therapy. Lipid disorders should be managed as clinically appropriate [see Drug Interactions (7)].
Acute hemolytic anemia has been reported in a patient treated with amprenavir.
There have been reports of spontaneous bleeding in patients with hemophilia A and B treated with protease inhibitors. In some patients, additional factor VIII was required. In many of the reported cases, treatment with protease inhibitors was continued or restarted. A causal relationship between protease inhibitor therapy and these episodes has not been established.
Cases of nephrolithiasis were reported during postmarketing surveillance in HIV-1-infected patients receiving LEXIVA.Because these events were reported voluntarily during clinical practice, estimates of frequency cannot be made. If signs or symptoms of nephrolithiasis occur, temporary interruption or discontinuation of therapy may be considered.
Because the potential for HIV cross-resistance among protease inhibitors has not been fully explored, it is unknown what effect therapy with LEXIVA will have on the activity of subsequently administered protease inhibitors. LEXIVA has been studied in patients who have experienced treatment failure with protease inhibitors [see Clinical Studies (14.2)].
LEXIVA ADVERSE REACTIONS
•Severe or life-threatening skin reactions have been reported with the use of LEXIVA
[see Warnings and Precautions (5.2)]
.•The most common moderate to severe adverse reactions in clinical trials of LEXIVA were diarrhea, rash, nausea, vomiting, and headache.•Treatment discontinuation due to adverse events occurred in 6.4% of subjects receiving LEXIVA and in 5.9% of subjects receiving comparator treatments. The most common adverse reactions leading to discontinuation of LEXIVA (incidence less than or equal to 1% of subjects) included diarrhea, nausea, vomiting, AST increased, ALT increased, and rash.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Adult Trials: The data for the 3 active-controlled clinical trials described below reflect exposure of 700 HIV-1–infected subjects to LEXIVA Tablets, including 599 subjects exposed to LEXIVA for greater than 24 weeks, and 409 subjects exposed for greater than 48 weeks. The population age ranged from 17 to 72 years. Of these subjects, 26% were female, 51% Caucasian, 31% black, 16% American Hispanic, and 70% were antiretroviral-naive. Sixty-one percent received LEXIVA 1,400 mg once daily plus ritonavir 200 mg once daily; 24% received LEXIVA 1,400 mg twice daily; and 15% received LEXIVA 700 mg twice daily plus ritonavir 100 mg twice daily.
Selected adverse reactions reported during the clinical efficacy trials of LEXIVA are shown in Tables 3 and 4. Each table presents adverse reactions of moderate or severe intensity in subjects treated with combination therapy for up to 48 weeks.
Table 3. Selected Moderate/Severe Clinical Adverse Reactions Reported in Greater Than or Equal to 2% of Antiretroviral-Naive Adult Subjects
|
Adverse Reaction |
APV30001a |
APV30002a |
||
|
LEXIVA 1,400 mg b.i.d. (n = 166) |
Nelfinavir 1,250 mg b.i.d. (n = 83) |
LEXIVA 1,400 mg q.d./ Ritonavir 200 mg q.d. (n = 322) |
Nelfinavir 1,250 mg b.i.d. (n = 327) |
|
|
Gastrointestinal |
||||
|
Diarrhea |
5% |
18% |
10% |
18% |
|
Nausea |
7% |
4% |
7% |
5% |
|
Vomiting |
2% |
4% |
6% |
4% |
|
Abdominal pain |
1% |
0% |
2% |
2% |
|
Skin |
||||
|
Rash |
8% |
2% |
3% |
2% |
|
General disorders |
||||
|
Fatigue |
2% |
1% |
4% |
2% |
|
Nervous system |
||||
|
Headache |
2% |
4% |
3% |
3% |
aAll subjects also received abacavir and lamivudine twice daily.
|
Adverse Reaction |
LEXIVA 700 mg b.i.d./ Ritonavir 100 mg b.i.d.a (n = 106) |
Lopinavir 400 mg b.i.d./ Ritonavir 100 mg b.i.d.a (n = 103) |
|
Gastrointestinal |
||
|
Diarrhea |
13% |
11% |
|
Nausea |
3% |
9% |
|
Vomiting |
3% |
5% |
|
Abdominal pain |
<1% |
2% |
|
Skin |
||
|
Rash |
3% |
0% |
|
Nervous system |
||
|
Headache |
4% |
2% |
aAll subjects also received 2 reverse transcriptase inhibitors.
Skin rash (without regard to causality) occurred in approximately 19% of subjects treated with LEXIVA in the pivotal efficacy trials. Rashes were usually maculopapular and of mild or moderate intensity, some with pruritus. Rash had a median onset of 11 days after initiation of LEXIVA and had a median duration of 13 days. Skin rash led to discontinuation of LEXIVA in less than 1% of subjects. In some subjects with mild or moderate rash, dosing with LEXIVA was often continued without interruption; if interrupted, reintroduction of LEXIVA generally did not result in rash recurrence.
The percentages of subjects with Grade 3 or 4 laboratory abnormalities in the clinical efficacy trials of LEXIVA are presented in Tables 5 and 6.
Table 5. Grade 3/4 Laboratory Abnormalities Reported in Greater Than or Equal to 2% of Antiretroviral-Naive Adult Subjects in Trials APV30001 and APV30002
|
Laboratory Abnormality |
APV30001a |
APV30002a |
||
|
LEXIVA 1,400 mg b.i.d. (n = 166) |
Nelfinavir 1,250 mg b.i.d. (n = 83) |
LEXIVA 1,400 mg q.d./ Ritonavir 200 mg q.d. (n = 322) |
Nelfinavir 1,250 mg b.i.d. (n = 327) |
|
|
ALT (>5 x ULN) |
6% |
5% |
8% |
8% |
|
AST (>5 x ULN) |
6% |
6% |
6% |
7% |
|
Serum lipase (>2 x ULN) |
8% |
4% |
6% |
4% |
|
Triglyceridesb (>750 mg/dL) |
0% |
1% |
6% |
2% |
|
Neutrophil count, absolute (<750 cells/mm3) |
3% |
6% |
3% |
4% |
aAll subjects also received abacavir and lamivudine twice daily.
bFasting specimens.
ULN = Upper limit of normal.
The incidence of Grade 3 or 4 hyperglycemia in antiretroviral-naive subjects who received LEXIVA in the pivotal trials was less than 1%.
Table 6. Grade 3/4 Laboratory Abnormalities Reported in Greater Than or Equal to 2% of Protease Inhibitor-Experienced Adult Subjects in Trial APV30003
|
Laboratory Abnormality |
LEXIVA 700 mg b.i.d./ Ritonavir 100 mg b.i.d.a (n = 104) |
Lopinavir 400 mg b.i.d./ Ritonavir 100 mg b.i.d.a (n = 103) |
|
Triglyceridesb (>750 mg/dL) |
11%c |
6%c |
|
Serum lipase (>2 x ULN) |
5% |
12% |
|
ALT (>5 x ULN) |
4% |
4% |
|
AST (>5 x ULN) |
4% |
2% |
|
Glucose (>251 mg/dL) |
2%c |
2%c |
aAll subjects also received 2 reverse transcriptase inhibitors.
bFasting specimens.
cn = 100 for LEXIVA plus ritonavir, n = 98 for lopinavir plus ritonavir.
ULN = Upper limit of normal.
Pediatric Trials: LEXIVA with and without ritonavir was studied in 237 HIV-1–infected pediatric subjects aged at least 4 weeks to 18 years in 3 open‑label trials, APV20002, APV20003, and APV29005 [see Clinical Studies (14.3)]. Vomiting and neutropenia occurred more frequently in pediatric subjects compared to adults. Other adverse events occurred with similar frequency in pediatric subjects compared with adults.
The frequency of vomiting among pediatric subjects receiving LEXIVA twice daily with ritonavir was 20% in subjects aged at least 4 weeks to less than 2 years and 36% in subjects aged 2 to 18 years compared with 10% in adults. The frequency of vomiting among pediatric subjects receiving LEXIVA twice daily without ritonavir was 60% in subjects aged 2 to 5 years compared with 16% in adults.
The median duration of drug‑related vomiting episodes in APV29005 was 1 day (range: 1 to 3 days), in APV20003 was 16 days (range: 1 to 38 days), and in APV20002 was 9 days (range: 4 to 13 days). Vomiting was treatment limiting in 4 pediatric subjects across all 3 trials.
The incidence of Grade 3 or 4 neutropenia (neutrophils less than 750 cells per mm3) seen in pediatric subjects treated with LEXIVA with and without ritonavir was higher (15%) than the incidence seen in adult subjects (3%). Grade 3/4 neutropenia occurred in 10% (5/51) of subjects aged at least 4 weeks to less than 2 years and 16% (28/170) of subjects aged 2 to 18 years.
In addition to adverse reactions reported from clinical trials, the following reactions have been identified during post-approval use of LEXIVA. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to LEXIVA.
Cardiac Disorders: Myocardial infarction.
Metabolism and Nutrition Disorders:Hypercholesterolemia.
Nervous System Disorders: Oral paresthesia.
Skin and Subcutaneous Tissue Disorders: Angioedema.
Urogenital: Nephrolithiasis.
DRUG INTERACTIONS
See also Contraindications (4), Clinical Pharmacology (12.3).
If LEXIVA is used in combination with ritonavir, see full prescribing information for ritonavir for additional information on drug interactions.
Amprenavir, the active metabolite of fosamprenavir, is an inhibitor of CYP3A4 metabolism and therefore should not be administered concurrently with medications with narrow therapeutic windows that are substrates of CYP3A4. Data also suggest that amprenavir induces CYP3A4.
Amprenavir is metabolized by CYP3A4. Coadministration of LEXIVA and drugs that induce CYP3A4, such as rifampin, may decrease amprenavir concentrations and reduce its therapeutic effect. Coadministration of LEXIVA and drugs that inhibit CYP3A4 may increase amprenavir concentrations and increase the incidence of adverse effects.
The potential for drug interactions with LEXIVA changes when LEXIVA is coadministered with the potent CYP3A4 inhibitor ritonavir. The magnitude of CYP3A4-mediated drug interactions (effect on amprenavir or effect on coadministered drug) may change when LEXIVA is coadministered with ritonavir. Because ritonavir is a CYP2D6 inhibitor, clinically significant interactions with drugs metabolized by CYP2D6 are possible when coadministered with LEXIVA plus ritonavir.
There are other agents that may result in serious and/or life-threatening drug interactions [see Contraindications (4)].
See Contraindications (4).
Table 7 provides a listing of established or potentially clinically significant drug interactions. Information in the table applies to LEXIVA with or without ritonavir, unless otherwise indicated.
|
Concomitant Drug Class: Drug Name |
Effect on Concentration of Amprenavir or Concomitant Drug |
Clinical Comment |
|
HCV/HIV-Antiviral Agents |
||
|
HCV protease inhibitor: Telaprevira |
LEXIVA/ritonavir: ↓Amprenavir ↓Telaprevir |
Coadministration of LEXIVA/ritonavir and telaprevir is not recommended. |
|
HCV protease inhibitor: Boceprevir |
LEXIVA/ritonavir: ↓Amprenavir (predicted) ↓Boceprevir (predicted) |
Coadministration of LEXIVA/ritonavir and boceprevir is not recommended. A pharmacokinetic interaction has been reported between boceprevir and some HIV protease inhibitors in combination with ritonavir, leading to decreased HIV protease inhibitor concentrations and, in some cases, decreased boceprevir concentrations. |
|
Non-nucleoside reverse transcriptase inhibitor: Efavirenza |
LEXIVA: ↓Amprenavir LEXIVA/ritonavir: ↓Amprenavir |
Appropriate doses of the combinations with respect to safety and efficacy have not been established. An additional 100 mg/day (300 mg total) of ritonavir is recommended when efavirenz is administered with LEXIVA/ritonavir once daily. No change in the ritonavir dose is required when efavirenz is administered with LEXIVA plus ritonavir twice daily. |
|
Non-nucleoside reverse transcriptase inhibitor: Nevirapinea |
LEXIVA: ↓Amprenavir ↑Nevirapine LEXIVA/ritonavir: ↓Amprenavir ↑Nevirapine |
Coadministration of nevirapine and LEXIVA without ritonavir is not recommended. No dosage adjustment required when nevirapine is administered with LEXIVA/ritonavir twice daily. The combination of nevirapine administered with LEXIVA/ritonavir once-daily regimen has not been studied. |
|
HIV protease inhibitor: Atazanavira |
LEXIVA: Interaction has not been evaluated. LEXIVA/ritonavir: ↓Atazanavir ↔Amprenavir |
Appropriate doses of the combinations with respect to safety and efficacy have not been established. |
|
HIV protease inhibitors: Indinavira, nelfinavira |
LEXIVA: ↑Amprenavir Effect on indinavir and nelfinavir is not well established. LEXIVA/ritonavir: Interaction has not been evaluated. |
Appropriate doses of the combinations with respect to safety and efficacy have not been established. |
|
HIV protease inhibitors: Lopinavir/ritonavira |
↓Amprenavir ↓Lopinavir |
An increased rate of adverse events has been observed. Appropriate doses of the combinations with respect to safety and efficacy have not been established. |
|
HIV protease inhibitor: Saquinavira |
LEXIVA: ↓Amprenavir Effect on saquinavir is not well established. LEXIVA/ritonavir: Interaction has not been evaluated. |
Appropriate doses of the combination with respect to safety and efficacy have not been established. |
|
HIV integrase inhibitor: Raltegravira |
LEXIVA: ↓Amprenavir ↓Raltegravir LEXIVA/ritonavir: ↓Amprenavir ↓Raltegravir |
Appropriate doses of the combination with respect to safety and efficacy have not been established. |
|
HIV CCR5 co-receptor antagonist: Maraviroca |
LEXIVA/ritonavir: ↓Amprenavir ↑Maraviroc |
No dosage adjustment required for LEXIVA/ritonavir. The recommended dose of maraviroc is 150 mg twice daily when coadministered with LEXIVA/ritonavir. LEXIVA should be given with ritonavir when coadministered with maraviroc. |
|
Other Agents |
||
|
Antiarrhythmics: Amiodarone, bepridil, lidocaine (systemic), and quinidine |
↑Antiarrhythmics |
Use with caution. Increased exposure may be associated with life-threatening reactions such as cardiac arrhythmias. Therapeutic concentration monitoring, if available, is recommended for antiarrhythmics. |
|
Anticoagulant: Warfarin |
Concentrations of warfarin may be affected. It is recommended that INR (international normalized ratio) be monitored. |
|
|
Anticonvulsants: Carbamazepine, phenobarbital, phenytoin Phenytoina |
LEXIVA: ↓Amprenavir LEXIVA/ritonavir: ↑Amprenavir ↓Phenytoin |
Use with caution. LEXIVA may be less effective due to decreased amprenavir plasma concentrations in patients taking these agents concomitantly. Plasma phenytoin concentrations should be monitored and phenytoin dose should be increased as appropriate. No change in LEXIVA/ritonavir dose is recommended. |
|
Antidepressant: Paroxetine, trazodone |
↓Paroxetine ↑Trazodone |
Coadministration of paroxetine with LEXIVA/ritonavir significantly decreased plasma levels of paroxetine. Any paroxetine dose adjustment should be guided by clinical effect (tolerability and efficacy). Concomitant use of trazodone and LEXIVA with or without ritonavir may increase plasma concentrations of trazodone. Adverse events of nausea, dizziness, hypotension, and syncope have been observed following coadministration of trazodone and ritonavir. If trazodone is used with a CYP3A4 inhibitor such as LEXIVA, the combination should be used with caution and a lower dose of trazodone should be considered. |
|
Antifungals: Ketoconazolea, itraconazole |
↑Ketoconazole ↑Itraconazole |
Increase monitoring for adverse events. LEXIVA: Dose reduction of ketoconazole or itraconazole may be needed for patients receiving more than 400 mg ketoconazole or itraconazole per day. LEXIVA/ritonavir: High doses of ketoconazole or itraconazole (greater than 200 mg/day) are not recommended. |
|
Anti-gout: Colchicine |
↑Colchicine |
Patients with renal or hepatic impairment should not be given colchicine with LEXIVA/ritonavir. LEXIVA/ritonavir and coadministration of colchicine: Treatment of gout flares: 0.6 mg (1 tablet) x 1 dose, followed by 0.3 mg (half tablet) 1 hour later. Dose to be repeated no earlier than 3 days. Prophylaxis of gout flares: If the original regimen was 0.6 mg twice a day, the regimen should be adjusted to 0.3 mg once a day. Treatment of familial Mediterranean fever (FMF): Maximum daily dose of 0.6 mg (may be given as 0.3 mg twice a day). LEXIVA and coadministration of colchicine: Treatment of gout flares: 1.2 mg (2 tablets) x 1 dose. Dose to be repeated no earlier than 3 days. Prophylaxis of gout flares: If the original regimen was 0.6 mg twice a day, the regimen should be adjusted to 0.3 mg twice a day or 0.6 mg once a day. Treatment of FMF: Maximum daily dose of 1.2 mg (may be given as 0.6 mg twice a day). |
|
Antimycobacterial: Rifabutina |
↑Rifabutin and rifabutin metabolite |
A complete blood count should be performed weekly and as clinically indicated to monitor for neutropenia. LEXIVA: A dosage reduction of rifabutin by at least half the recommended dose is required. LEXIVA/ritonavir: Dosage reduction of rifabutin by at least 75% of the usual dose of 300 mg/day is recommended (a maximum dose of 150 mg every other day or 3 times per week). |
|
Benzodiazepines: Alprazolam, clorazepate, diazepam, flurazepam |
↑Benzodiazepines |
Clinical significance is unknown. A decrease in benzodiazepine dose may be needed. |
|
Calcium channel blockers: Diltiazem, felodipine, nifedipine, nicardipine, nimodipine, verapamil, amlodipine, nisoldipine, isradipine |
↑Calcium channel blockers |
Use with caution. Clinical monitoring of patients is recommended. |
|
Corticosteroid: Dexamethasone |
↓Amprenavir |
Use with caution. LEXIVA may be less effective due to decreased amprenavir plasma concentrations. |
|
Endothelin-receptor antagonists: Bosentan |
↑Bosentan |
Coadministration of bosentan in patients on LEXIVA: In patients who have been receiving LEXIVA for at least 10 days, start bosentan at 62.5 mg once daily or every other day based upon individual tolerability. Coadministration of LEXIVA in patients on bosentan: Discontinue use of bosentan at least 36 hours prior to initiation of LEXIVA. After at least 10 days following the initiation of LEXIVA, resume bosentan at 62.5 mg once daily or every other day based upon individual tolerability. |
|
Histamine H2-receptor antagonists: Cimetidine, famotidine, nizatidine, ranitidinea |
LEXIVA: ↓Amprenavir LEXIVA/ritonavir: Interaction not evaluated |
Use with caution. LEXIVA may be less effective due to decreased amprenavir plasma concentrations. |
|
HMG-CoA reductase inhibitors: Atorvastatina |
↑Atorvastatin |
Titrate atorvastatin dose carefully and use the lowest necessary dose; do not exceed atorvastatin 20 mg/day. |
|
Immunosuppressants: Cyclosporine, tacrolimus, rapamycin |
↑Immunosuppressants |
Therapeutic concentration monitoring is recommended for immunosuppressant agents. |
|
Inhaled beta-agonist: Salmeterol |
↑Salmeterol |
Concurrent administration of salmeterol with LEXIVA is not recommended. The combination may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations, and sinus tachycardia. |
|
Inhaled/nasal steroid: Fluticasone |
LEXIVA: ↑Fluticasone LEXIVA/ritonavir: ↑Fluticasone |
Use with caution. Consider alternatives to fluticasone, particularly for long-term use. May result in significantly reduced serum cortisol concentrations. Systemic corticosteroid effects including Cushing’s syndrome and adrenal suppression have been reported during postmarketing use in patients receiving ritonavir and inhaled or intranasally administered fluticasone. Coadministration of fluticasone and LEXIVA/ritonavir is not recommended unless the potential benefit to the patient outweighs the risk of systemic corticosteroid side effects. |
|
Narcotic analgesic: Methadone |
↓Methadone |
Data suggest that the interaction is not clinically relevant; however, patients should be monitored for opiate withdrawal symptoms. |
|
Oral contraceptives: Ethinyl estradiol/norethindronea |
LEXIVA: ↓Amprenavir ↓Ethinyl estradiol LEXIVA/ritonavir: ↓Ethinyl estradiol |
Alternative methods of non-hormonal contraception are recommended. May lead to loss of virologic response. a Increased risk of transaminase elevations. No data are available on the use of LEXIVA/ritonavir with other hormonal therapies, such as hormone replacement therapy (HRT) for postmenopausal women. |
|
PDE5 inhibitors: Sildenafil, tadalafil, vardenafil |
↑Sildenafil ↑Tadalafil ↑Vardenafil |
May result in an increase in PDE5 inhibitor-associated adverse events, including hypotension, syncope, visual disturbances, and priapism. Use of PDE5 inhibitors for pulmonary arterial hypertension (PAH): •Use of sildenafil (REVATIO) is contraindicated when used for the treatment of PAH [see Contraindications (4)]. •The following dose adjustments are recommended for use of tadalafil (ADCIRCA®) with LEXIVA:Coadministration of ADCIRCA in patients on LEXIVA: In patients receiving LEXIVA for at least one week, start ADCIRCA at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability. Coadministration of LEXIVA in patients on ADCIRCA: Avoid use of ADCIRCA during the initiation of LEXIVA. Stop ADCIRCA at least 24 hours prior to starting LEXIVA. After at least one week following the initiation of LEXIVA, resume ADCIRCA at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability. Use of PDE5 inhibitors for erectile dysfunction: LEXIVA: Sildenafil: 25 mg every 48 hours. Tadalafil: no more than 10 mg every 72 hours. Vardenafil: no more than 2.5 mg every 24 hours. LEXIVA/ritonavir: Sildenafil: 25 mg every 48 hours. Tadalafil: no more than 10 mg every 72 hours. Vardenafil: no more than 2.5 mg every 72 hours. Use with increased monitoring for adverse events. |
|
Proton pump inhibitors: Esomeprazolea, lansoprazole, omeprazole, pantoprazole, rabeprazole |
LEXIVA: ↔Amprenavir ↑Esomeprazole LEXIVA/ritonavir: ↔Amprenavir ↔Esomeprazole |
Proton pump inhibitors can be administered at the same time as a dose of LEXIVA with no change in plasma amprenavir concentrations. |
|
Tricyclic antidepressants: Amitriptyline, imipramine |
↑Tricyclics |
Therapeutic concentration monitoring is recommended for tricyclic antidepressants. |
a See Clinical Pharmacology (12.3) Tables 10, 11, 12, or 13 for magnitude of interaction.
USE IN SPECIFIC POPULATIONS
Pregnancy Category C. Embryo/fetal development studies were conducted in rats (dosed from day 6 to day 17 of gestation) and rabbits (dosed from day 7 to day 20 of gestation). Administration of fosamprenavir to pregnant rats and rabbits produced no major effects on embryo-fetal development; however, the incidence of abortion was increased in rabbits that were administered fosamprenavir. Systemic exposures (AUC0-24 hr) to amprenavir at these dosages were 0.8 (rabbits) to 2 (rats) times the exposures in humans following administration of the maximum recommended human dose (MRHD) of fosamprenavir alone or 0.3 (rabbits) to 0.7 (rats) times the exposures in humans following administration of the MRHD of fosamprenavir in combination with ritonavir. In contrast, administration of amprenavir was associated with abortions and an increased incidence of minor skeletal variations resulting from deficient ossification of the femur, humerus, and trochlea, in pregnant rabbits at the tested dose approximately one-twentieth the exposure seen at the recommended human dose.
The mating and fertility of the F1 generation born to female rats given fosamprenavir was not different from control animals; however, fosamprenavir did cause a reduction in both pup survival and body weights. Surviving F1 female rats showed an increased time to successful mating, an increased length of gestation, a reduced number of uterine implantation sites per litter, and reduced gestational body weights compared with control animals. Systemic exposure (AUC0-24 hr) to amprenavir in the F0 pregnant rats was approximately 2 times higher than exposures in humans following administration of the MRHD of fosamprenavir alone or approximately the same as those seen in humans following administration of the MRHD of fosamprenavir in combination with ritonavir.
There are no adequate and well-controlled studies in pregnant women. LEXIVA should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Antiretroviral Pregnancy Registry: To monitor maternal-fetal outcomes of pregnant women exposed to LEXIVA, an Antiretroviral Pregnancy Registry has been established. Physicians are encouraged to register patients by calling 1-800-258-4263.
The Centers for Disease Control and Prevention recommend that HIV-infected mothers not breastfeed their infants to avoid risking postnatal transmission of HIV. Although it is not known if amprenavir is excreted in human milk, amprenavir is secreted into the milk of lactating rats. Because of both the potential for HIV transmission and the potential for serious adverse reactions in nursing infants, mothers should be instructed not to breastfeed if they are receiving LEXIVA.
The safety, pharmacokinetic profile, virologic, and immunologic responses of LEXIVA with and without ritonavir were evaluated in protease inhibitor-naive and –experienced HIV-1–infected pediatric subjects aged at least 4 weeks to less than 18 years and weighing at least 3 kg in 3 open-label trials [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.3)]. Vomiting and neutropenia, were more frequent in pediatrics than in adults [see Adverse Reactions (6.1)]. Other adverse events occurred with similar frequency in pediatric subjects compared with adults.
Treatment with LEXIVA is not recommended in protease inhibitor-experienced pediatric patients younger than 6 months. The pharmacokinetics, safety, tolerability, and efficacy of LEXIVA in pediatric patients younger than 4 weeks have not been established [see Clinical Pharmacology (12.3)]. Available pharmacokinetic and clinical data do not support once-daily dosing of LEXIVA alone or in combination with ritonavir for any pediatrics or twice-daily dosing without ritonavir in pediatric patients younger than 2 years[see Clinical Pharmacology (12.3), Clinical Studies (14.3)]. See Dosage and Administration (2.2) for dosing recommendations for pediatric patients.
Clinical studies of LEXIVA did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger adults. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
Amprenavir is principally metabolized by the liver; therefore, caution should be exercised when administering LEXIVA to patients with hepatic impairment because amprenavir concentrations may be increased [see Clinical Pharmacology (12.3)]. Patients with impaired hepatic function receiving LEXIVA with or without concurrent ritonavir require dose reduction [see Dosage and Administration (2.3)].
There are no data to support dosing recommendations for pediatric subjects with hepatic impairment.
OVERDOSAGE
In a healthy volunteer repeat-dose pharmacokinetic trial evaluating high-dose combinations of LEXIVA plus ritonavir, an increased frequency of Grade 2/3 ALT elevations (greater than 2.5 x ULN) was observed with LEXIVA 1,400 mg twice daily plus ritonavir 200 mg twice daily (4 of 25 subjects). Concurrent Grade 1/2 elevations in AST (greater than 1.25 x ULN) were noted in 3 of these 4 subjects. These transaminase elevations resolved following discontinuation of dosing.
There is no known antidote for LEXIVA. It is not known whether amprenavir can be removed by peritoneal dialysis or hemodialysis. If overdosage occurs, the patient should be monitored for evidence of toxicity and standard supportive treatment applied as necessary.
LEXIVA DESCRIPTION
LEXIVA (fosamprenavir calcium) is a prodrug of amprenavir, an inhibitor of HIV protease. The chemical name of fosamprenavir calcium is (3S)-tetrahydrofuran-3-yl (1S,2R)-3-[[(4-aminophenyl) sulfonyl](isobutyl)amino]-1-benzyl-2-(phosphonooxy) propylcarbamate monocalcium salt. Fosamprenavir calcium is a single stereoisomer with the (3S)(1S,2R) configuration. It has a molecular formula of C25H34CaN3O9PS and a molecular weight of 623.7. It has the following structural formula:
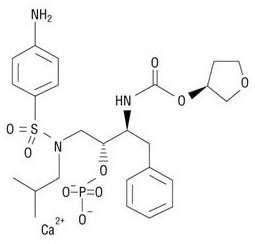
Fosamprenavir calcium is a white to cream-colored solid with a solubility of approximately 0.31 mg per mL in water at 25°C.
LEXIVA Tablets are available for oral administration in a strength of 700 mg of fosamprenavir as fosamprenavir calcium (equivalent to approximately 600 mg of amprenavir). Each 700 mg tablet contains the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and povidone K30. The tablet film-coating contains the inactive ingredients hypromellose, iron oxide red, titanium dioxide, and triacetin.
LEXIVA Oral Suspension is available in a strength of 50 mg per mL of fosamprenavir as fosamprenavir calcium equivalent to approximately 43 mg of amprenavir. LEXIVA Oral Suspension is a white to off-white suspension with a grape-bubblegum-peppermint flavor. Each one milliliter (1 mL) contains the inactive ingredients artificial grape-bubblegum flavor, calcium chloride dihydrate, hypromellose, methylparaben, natural peppermint flavor, polysorbate 80, propylene glycol, propylparaben, purified water, and sucralose.
CLINICAL PHARMACOLOGY
Fosamprenavir is an antiviral agent [see Microbiology (12.4)].
The pharmacokinetic properties of amprenavir after administration of LEXIVA, with or without ritonavir, have been evaluated in both healthy adult volunteers and in HIV-1-infected subjects; no substantial differences in steady-state amprenavir concentrations were observed between the 2 populations.
The pharmacokinetic parameters of amprenavir after administration of LEXIVA (with and without concomitant ritonavir) are shown in Table 8.
Table 8. Geometric Mean (95% CI) Steady-State Plasma Amprenavir Pharmacokinetic Parameters in Adults
|
Regimen |
Cmax (mcg/mL) |
Tmax (hours)a |
AUC24 (mcg•hr/mL) |
Cmin (mcg/mL) |
|
LEXIVA 1,400 mg b.i.d. |
4.82 (4.06-5.72) |
1.3 (0.8-4.0) |
33.0 (27.6-39.2) |
0.35 (0.27-0.46) |
|
LEXIVA 1,400 mg q.d. plus Ritonavir 200 mg q.d. |
7.24 (6.32-8.28) |
2.1 (0.8-5.0) |
69.4 (59.7-80.8) |
1.45 (1.16-1.81) |
|
LEXIVA 1,400 mg q.d. plus Ritonavir 100 mg q.d. |
7.93 (7.25-8.68) |
1.5 (0.75-5.0) |
66.4 (61.1-72.1) |
0.86 (0.74-1.01) |
|
LEXIVA 700 mg b.i.d. plus Ritonavir 100 mg b.i.d. |
6.08 (5.38-6.86) |
1.5 (0.75-5.0) |
79.2 (69.0-90.6) |
2.12 (1.77-2.54) |
aData shown are median (range).
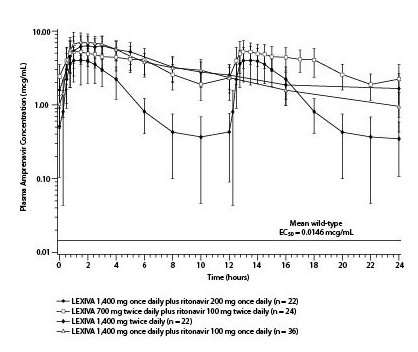
The mean plasma amprenavir concentrations of the dosing regimens over the dosing intervals are displayed in Figure 1.
Figure 1. Mean (SD) Steady-State Plasma Amprenavir Concentrations and Mean EC50 Values Against HIV from Protease Inhibitor-Naive Subjects (in the Absence of Human Serum)
Absorption and Bioavailability: After administration of a single dose of LEXIVA to HIV-1–infected subjects, the time to peak amprenavir concentration (Tmax) occurred between 1.5 and 4 hours (median 2.5 hours). The absolute oral bioavailability of amprenavir after administration of LEXIVA in humans has not been established.
After administration of a single 1,400-mg dose in the fasted state, LEXIVA Oral Suspension (50 mg per mL) and LEXIVA Tablets (700 mg) provided similar amprenavir exposures (AUC); however, the Cmax of amprenavir after administration of the suspension formulation was 14.5% higher compared with the tablet.
Effects of Food on Oral Absorption: Administration of a single 1,400-mg dose of LEXIVA Tablets in the fed state (standardized high-fat meal: 967 kcal, 67 grams fat, 33 grams protein, 58 grams carbohydrate) compared with the fasted state was associated with no significant changes in amprenavir Cmax, Tmax, or AUC0-∞ [see Dosage and Administration (2)].
Administration of a single 1,400-mg dose of LEXIVA Oral Suspension in the fed state (standardized high-fat meal: 967 kcal, 67 grams fat, 33 grams protein, 58 grams carbohydrate) compared with the fasted state was associated with a 46% reduction in Cmax, a 0.72-hour delay in Tmax, and a 28% reduction in amprenavir AUC0-∞.
Distribution: In vitro, amprenavir is approximately 90% bound to plasma proteins, primarily to alpha1-acid glycoprotein. In vitro, concentration-dependent binding was observed over the concentration range of 1 to 10 mcg per mL, with decreased binding at higher concentrations. The partitioning of amprenavir into erythrocytes is low, but increases as amprenavir concentrations increase, reflecting the higher amount of unbound drug at higher concentrations.
Metabolism: After oral administration, fosamprenavir is rapidly and almost completely hydrolyzed to amprenavir and inorganic phosphate prior to reaching the systemic circulation. This occurs in the gut epithelium during absorption. Amprenavir is metabolized in the liver by the CYP3A4 enzyme system. The 2 major metabolites result from oxidation of the tetrahydrofuran and aniline moieties. Glucuronide conjugates of oxidized metabolites have been identified as minor metabolites in urine and feces.
Amprenavir is both a substrate for and inducer of P-glycoprotein.
Elimination: Excretion of unchanged amprenavir in urine and feces is minimal. Unchanged amprenavir in urine accounts for approximately 1% of the dose; unchanged amprenavir was not detectable in feces. Approximately 14% and 75% of an administered single dose of 14C-amprenavir can be accounted for as metabolites in urine and feces, respectively. Two metabolites accounted for greater than 90% of the radiocarbon in fecal samples. The plasma elimination half-life of amprenavir is approximately 7.7 hours.
Special Populations: Hepatic Impairment: The pharmacokinetics of amprenavir have been studied after the administration of LEXIVA in combination with ritonavir to adult HIV-1–infected subjects with mild, moderate, and severe hepatic impairment. Following 2 weeks of dosing with LEXIVA plus ritonavir, the AUC of amprenavir was increased by approximately 22% in subjects with mild hepatic impairment, by approximately 70% in subjects with moderate hepatic impairment, and by approximately 80% in subjects with severe hepatic impairment compared with HIV-1–infected subjects with normal hepatic function. Protein binding of amprenavir was decreased in subjects with hepatic impairment. The unbound fraction at 2 hours (approximate Cmax) ranged between a decrease of -7% to an increase of 57% while the unbound fraction at the end of the dosing interval (Cmin) increased from 50% to 102% [see Dosage and Administration (2.3)].
The pharmacokinetics of amprenavir have been studied after administration of amprenavir given as AGENERASE® Capsules to adult subjects with hepatic impairment. Following administration of a single 600-mg oral dose, the AUC of amprenavir was increased by approximately 2.5-fold in subjects with moderate cirrhosis and by approximately 4.5-fold in subjects with severe cirrhosis compared with healthy volunteers [see Dosage and Administration (2.3)].
Renal Impairment: The impact of renal impairment on amprenavir elimination in adults has not been studied. The renal elimination of unchanged amprenavir represents approximately 1% of the administered dose; therefore, renal impairment is not expected to significantly impact the elimination of amprenavir.
Pediatric Patients: The pharmacokinetics of amprenavir following administration of LEXIVA Oral Suspension and LEXIVA Tablets, with or without ritonavir, have been studied in a total of 212 HIV-1–infected pediatric subjects enrolled in 3 trials. LEXIVA without ritonavir was administered as 30 or 40 mg per kg twice daily to children aged 2 to 5 years. LEXIVA with ritonavir was administered as LEXIVA 30 mg per kg plus ritonavir 6 mg per kg once daily to children aged 2 to 18 years and as LEXIVA 18 to 60 mg per kg plus ritonavir 3 to 10 mg per kg twice daily to children aged at least 4 weeks to 18 years; body weights ranged from 3 to 103 kg.
Amprenavir apparent clearance decreased with increasing weight. Weight-adjusted apparent clearance was higher in children younger than 4 years, suggesting that younger children require higher mg per kg dosing of LEXIVA.
The pharmacokinetics of LEXIVA Oral Suspension in protease inhibitor-naive infants younger than 6 months (n = 9) receiving LEXIVA 45 mg per kg plus ritonavir 10 mg per kg twice daily generally demonstrated lower AUC12 and Cmin than adults receiving twice-daily LEXIVA 700 mg plus ritonavir 100 mg, the dose recommended for protease-experienced adults. The mean steady-state amprenavir AUC12, Cmax, and Cmin were 26.6 mcg•hour per mL, 6.25 mcg per mL, and 0.86 mcg per mL, respectively. These data do not support twice-daily dosing of LEXIVA alone or in combination with ritonavir in protease inhibitor-experienced patients younger than 6 months. Because of expected low amprenavir exposure and a requirement for large volume of drug, twice-daily dosing of LEXIVA alone (without ritonavir) in pediatric subjects younger than 2 years was not studied.
Pharmacokinetic parameters for LEXIVA administered with food and with ritonavir in this patient population at the recommended weight-band–based dosage regimens are provided in Table 9.
Table 9. Geometric Mean (95% CI) Steady-State Plasma Amprenavir Pharmacokinetic Parameters by Weight in Pediatric and Adolescent Subjects Aged at Least 4 Weeks to 18 Years Receiving LEXIVA With Ritonavir
|
Weight |
Recommended Dosage Regimen |
Cmax |
AUC24 |
Cmin |
|||
|
n |
(mcg/mL) |
n |
(mcg•hr/mL) |
n |
(mcg/mL) |
||
|
<11 kg |
LEXIVA 45 mg/kg plus Ritonavir 7 mg/kg b.i.d |
12 |
6.00 (3.88, 9.29) |
12 |
57.3 (34.1, 96.2) |
27 |
1.65 (1.22, 2.24) |
|
11 kg - <15 kg |
LEXIVA 30 mg/kg plus Ritonavir 3 mg/kg b.i.d |
Not studieda |
|||||
|
15 kg - <20 kg |
LEXIVA 23 mg/kg plus Ritonavir 3 mg/kg b.i.d. |
5 |
9.54 (4.63, 19.7) |
5 |
121 (54.2, 269) |
9 |
3.56 (2.33, 5.43) |
|
>20 kg - <39 kg |
LEXIVA 18 mg/kg plus Ritonavir 3 mg/kg b.i.d. |
13 |
6.24 (5.01, 7.77) |
12 |
97.9 (77.0, 124) |
23 |
2.54 (2.11, 3.06) |
|
≥39 kg |
LEXIVA 700 mg plus Ritonavir 100 mg b.i.d. |
15 |
5.03 (4.04, 6.26) |
15 |
72.3 (59.6, 87.6) |
42 |
1.98 (1.72, 2.29) |
aRecommended dose for pediatric subjects weighing 11 kg to less than 15 kg is based on population pharmacokinetic analysis.
Subjects aged 2 to less than 6 years receiving LEXIVA 30 mg per kg twice daily without ritonavir achieved geometric mean (95% CI) amprenavir Cmax (n = 9), AUC12 (n = 9), and Cmin (n = 19) of 7.15 (5.05, 10.1), 22.3 (15.3, 32.6), and 0.513 (0.384, 0.686), respectively.
Geriatric Patients: The pharmacokinetics of amprenavir after administration of LEXIVA to patients older than 65 years have not been studied [see Use in Specific Populations (8.5)].
Gender: The pharmacokinetics of amprenavir after administration of LEXIVA do not differ between males and females.
Race: The pharmacokinetics of amprenavir after administration of LEXIVA do not differ between blacks and non-blacks.
Drug Interactions: [See Contraindications (4), Warnings and Precautions (5.1), Drug Interactions (7).]
Amprenavir, the active metabolite of fosamprenavir, is metabolized in the liver by the cytochrome P450 enzyme system. Amprenavir inhibits CYP3A4. Data also suggest that amprenavir induces CYP3A4. Caution should be used when coadministering medications that are substrates, inhibitors, or inducers of CYP3A4, or potentially toxic medications that are metabolized by CYP3A4. Amprenavir does not inhibit CYP2D6, CYP1A2, CYP2C9, CYP2C19, CYP2E1, or uridine glucuronosyltransferase (UDPGT).
Drug interaction trials were performed with LEXIVA and other drugs likely to be coadministered or drugs commonly used as probes for pharmacokinetic interactions. The effects of coadministration on AUC, Cmax, and Cmin values are summarized in Table 10 (effect of other drugs on amprenavir) and Table 12 (effect of LEXIVA on other drugs). In addition, since LEXIVA delivers comparable amprenavir plasma concentrations as AGENERASE, drug interaction data derived from trials with AGENERASE are provided in Tables 11 and 13. For information regarding clinical recommendations, [see Drug Interactions (7)].
Table 10. Drug Interactions: Pharmacokinetic Parameters for Amprenavir After Administration of LEXIVA in the Presence of the Coadministered Drug(s)
|
Coadministered Drug(s) and Dose(s) |
Dose of LEXIVA a |
n |
% Change in Amprenavir Pharmacokinetic Parameters (90% CI) |
||
|
Cmax |
AUC |
Cmin |
|||
|
Antacid (MAALOX TC®) 30 mL single dose |
1,400 mg single dose |
30 |
↓35 (↓24 to ↓42) |
↓18 (↓9 to ↓26) |
↑14 (↓7 to ↑39) |
|
Atazanavir 300 mg q.d. for 10 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 10 days |
22 |
↔ |
↔ |
↔ |
|
Atorvastatin 10 mg q.d. for 4 days |
1,400 mg b.i.d. for 2 weeks |
16 |
↓18 (↓34 to ↑1) |
↓27 (↓41 to ↓12) |
↓12 (↓27 to ↓6) |
|
Atorvastatin 10 mg q.d. for 4 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
16 |
↔ |
↔ |
↔ |
|
Efavirenz 600 mg q.d. for 2 weeks |
1,400 mg q.d. plus ritonavir 200 mg q.d. for 2 weeks |
16 |
↔ |
↓13 (↓30 to 7) |
↓36 (↓8 to ↓56) |
|
Efavirenz 600 mg q.d. plus additional ritonavir 100 mg q.d. for 2 weeks |
1,400 mg q.d. plus ritonavir 200 mg q.d. for 2 weeks |
16 |
↑18 (↑1 to ↑38) |
↑11 (0 to ↑24) |
↔ |
|
Efavirenz 600 mg q.d. for 2 weeks |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
16 |
↔ |
↔ |
↓17 (↓4 to ↓29) |
|
Esomeprazole 20 mg q.d. for 2 weeks |
1,400 mg b.i.d. for 2 weeks |
25 |
↔ |
↔ |
↔ |
|
Esomeprazole 20 mg q.d. for 2 weeks |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
23 |
↔ |
↔ |
↔ |
|
Ethinyl estradiol/ 0.035 mg/0.5 mg q.d. for 21 days |
700 mg b.i.d. plus ritonavirb 100 mg b.i.d. for 21 days |
25 |
↔c |
↔c |
↔c |
|
Ketoconazoled 200 mg q.d. for 4 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 4 days |
15 |
↔ |
↔ |
↔ |
|
Lopinavir/ritonavir 533 mg/133 mg b.i.d. |
1,400 mg b.i.d. for 2 weeks |
18 |
↓13e |
↓26e |
↓42e |
|
Lopinavir/ritonavir 400 mg/100 mg b.i.d. for 2 weeks |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
18 |
↓58 (↓42 to ↓70) |
↓63 (↓51 to ↓72) |
↓65 (↓54 to ↓73) |
|
Maraviroc 300 mg b.i.d. for 10 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 20 days |
14 |
↓34 (↓25 to ↓41) |
↓35 (↓29 to ↓41) |
↓36 (↓27 to ↓43) |
|
Maraviroc 300 mg q.d. for 10 days |
1,400 mg q.d. plus ritonavir 100 mg q.d. for 20 days |
14 |
↓29 (↓20 to ↓38) |
↓30 (↓23 to ↓36) |
↓15 (↓3 to ↓25) |
|
Methadone 70 to 120 mg q.d. for 2 weeks |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
19 |
↔c |
↔c |
↔c |
|
Nevirapine 200 mg b.i.d. for 2 weeksf |
1,400 mg b.i.d. for 2 weeks |
17 |
↓25 (↓37 to ↓10) |
↓33 (↓45 to ↓20) |
↓35 (↓50 to ↓15) |
|
Nevirapine 200 mg b.i.d. for 2 weeksf |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
17 |
↔ |
↓11 (↓23 to ↑3) |
↓19 (↓32 to ↓4) |
|
Phenytoin 300 mg q.d. for 10 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 10 days |
13 |
↔ |
↑20 (↑8 to ↑34) |
↑19 (↑6 to ↑33) |
|
Raltegravir 400 mg b.i.d. for 14 days |
1,400 mg b.i.d. for 14 days (fasted) |
14 |
↓27 (↓46 to ↔) |
↓36 (↓53 to ↓13) |
↓43g (↓59 to ↓21) |
|
1,400 mg b.i.d. for 14 daysh |
14 |
↓15 (↓27 to ↓1) |
↓17 (↓27 to ↓6) |
↓32g (↓53 to ↓1) |
|
|
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for |
14 |
↓14 (↓39 to ↑20) |
↓17 (↓38 to ↑12) |
↓20g (↓45 to ↑17) |
|
|
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for |
12 |
↓25 (↓42 to ↓2) |
↓25 (↓44 to ↔) |
↓33g (↓52 to ↓7) |
|
|
Raltegravir 400 mg b.i.d. for 14 days |
1,400 mg q.d. plus ritonavir 100 mg q.d. for |
13 |
↓18 (↓34 to ↔) |
↓24 (↓41 to ↔) |
↓50g (↓64 to ↓31) |
|
1,400 mg q.d. plus ritonavir 100 mg q.d. for 14 daysh |
14 |
↑27 (↓1 to ↑62) |
↑13 (↓7 to ↑38) |
17g (↓45 to ↑26) |
|
|
Ranitidine 300 mg single dose (administered 1 hour before fosamprenavir) |
1,400 mg single dose |
30 |
51 (↓43 to ↓58) |
30 (↓22 to ↓37) |
↔ (↓19 to ↑21) |
|
Rifabutin 150 mg q.o.d. for 2 weeks |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
15 |
↑36c (↑18 to ↑55) |
↑35c (↑17 to ↑56) |
↑17c (↓1 to ↑39) |
|
Telaprevir 750 mg q. 8 hr for 10 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 20 days |
18 |
↓35 (↓30 to ↓41) |
↓47 (↓42 to ↓51) |
↓56 (↓50 to ↓60) |
|
Telaprevir 1,125 mg q. 12 hr for 4 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 24 days |
17 |
↓40i (↓33 to ↓45) |
↓49i (↓45 to ↓53) |
↓58i (↓53 to ↓63) |
|
Tenofovir 300 mg q.d. for 4 to 48 weeks |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 4 to 48 weeks |
45 |
NA |
NA |
↔j |
|
Tenofovir 300 mg q.d. for 4 to 48 weeks |
1,400 mg q.d. plus ritonavir 200 mg q.d. for 4 to 48 weeks |
60 |
NA |
NA |
↔j |
aConcomitant medication is also shown in this column where appropriate.
bRitonavir Cmax, AUC, and Cmin increased by 63%, 45%, and 13%, respectively, compared with historical control.
cCompared with historical control.
dSubjects were receiving LEXIVA/ritonavir for 10 days prior to the 4-day treatment period with both ketoconazole and LEXIVA/ritonavir.
eCompared with LEXIVA 700 mg/ritonavir 100 mg b.i.d. for 2 weeks.
fSubjects were receiving nevirapine for at least 12 weeks prior to trial.
gClast (C12 hr or C24 hr).
hDoses of LEXIVA and raltegravir were given with food on pharmacokinetic sampling days and without regard to food all other days.
iN = 18 for Cmin.
jCompared with parallel control group.
↑ = Increase; ↓= Decrease; ↔ = No change (↑or ↓ less than or equal to 10%), NA = Not applicable.
Table 11. Drug Interactions: Pharmacokinetic Parameters for Amprenavir After Administration of AGENERASE in the Presence of the Coadministered Drug(s)
|
Coadministered Drug(s) and Dose(s) |
Dose of AGENERASE a |
n |
% Change in Amprenavir Pharmacokinetic Parameters (90% CI) |
||
|
Cmax |
AUC |
Cmin |
|||
|
Abacavir 300 mg b.i.d. for 2 to 3 weeks |
900 mg b.i.d. for 2 to 3 weeks |
4 |
↔a |
↔a |
↔a |
|
Clarithromycin 500 mg b.i.d. for 4 days |
1,200 mg b.i.d. for 4 days |
12 |
↑15 (↑1 to ↑31) |
↑18 (↑8 to ↑29) |
↑39 (↑31 to ↑47) |
|
Delavirdine 600 mg b.i.d. for 10 days |
600 mg b.i.d. for 10 days |
9 |
↑40b |
↑130b |
↑125b |
|
Ethinyl estradiol/norethindrone 0.035 mg/1 mg for 1 cycle |
1,200 mg b.i.d. for 28 days |
10 |
↔ |
↓22 (↓35 to ↓8) |
↓20 (↓41 to ↑8) |
|
Indinavir 800 mg t.i.d. for 2 weeks (fasted) |
750 or 800 mg t.i.d. for 2 weeks (fasted) |
9 |
↑18 (↑13 to ↑58) |
↑33 (↑2 to ↑73) |
↑25 (↓27 to ↑116) |
|
Ketoconazole 400 mg single dose |
1,200 mg single dose |
12 |
↓16 (↓25 to ↓6) |
↑31 (↑20 to ↑42) |
NA |
|
Lamivudine 150 mg single dose |
600 mg single dose |
11 |
↔ |
↔ |
NA |
|
Methadone 44 to 100 mg q.d. for >30 days |
1,200 mg b.i.d. for 10 days |
16 |
↓27c |
↓30c |
↓25c |
|
Nelfinavir 750 mg t.i.d. for 2 weeks (fed) |
750 or 800 mg t.i.d. for 2 weeks (fed) |
6 |
↓14 (↓38 to ↑20) |
↔ |
↑189 (↑52 to ↑448) |
|
Rifabutin 300 mg q.d. for 10 days |
1,200 mg b.i.d. for 10 days |
5 |
↔ |
↓15 (↓28 to 0) |
↓15 (↓38 to ↑17) |
|
Rifampin 300 mg q.d. for 4 days |
1,200 mg b.i.d. for 4 days |
11 |
↓70 (↓76 to ↓62) |
↓82 (↓84 to ↓78) |
↓92 (↓95 to ↓89) |
|
Saquinavir 800 mg t.i.d. for 2 weeks (fed) |
750 or 800 mg t.i.d. for 2 weeks (fed) |
7 |
↓37 (↓54 to ↓14) |
↓32 (↓49 to ↓9) |
↓14 (↓52 to ↑54) |
|
Zidovudine 300 mg single dose |
600 mg single dose |
12 |
↔ |
↑13 (↓2 to ↑31) |
NA |
aCompared with parallel control group.
bMedian percent change; confidence interval not reported.
cCompared with historical data.
↑ = Increase; ↓ = Decrease; ↔ = No change (↑or ↓ less than 10%); NA = Cmin not calculated for single‑dose trial.
|
Coadministered Drug(s) and Dose(s) |
Dose of LEXIVA a |
n |
% Change in Pharmacokinetic Parameters |
||
|
Cmax |
AUC |
Cmin |
|||
|
Atazanavir 300 mg q.d. for 10 daysb |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 10 days |
21 |
↓24 (↓39 to ↓6) |
↓22 (↓34 to ↓9) |
↔ |
|
Atorvastatin 10 mg q.d. for 4 days |
1,400 mg b.i.d. for 2 weeks |
16 |
↑304 (↑205 to ↑437) |
↑130 (↑100 to ↑164) |
↓10 (↓27 to ↑12) |
|
Atorvastatin 10 mg q.d. for 4 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
16 |
↑184 (↑126 to ↑257) |
↑153 (↑115 to ↑199) |
↑73 (↑45 to ↑108) |
|
Esomeprazole 20 mg q.d. for 2 weeks |
1,400 mg b.i.d. for 2 weeks |
25 |
↔ |
↑55 (↑39 to ↑73) |
ND |
|
Esomeprazole 20 mg q.d. for 2 weeks |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for |
23 |
↔ |
↔ |
ND |
|
Ethinyl estradiolc 0.035 mg q.d. for 21 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 21 days |
25 |
↓28 (↓21 to ↓35) |
↓37 (↓30 to ↓42) |
ND |
|
Ketoconazoled 200 mg q.d. for 4 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 4 days |
15 |
↑25 (↑0 to ↑56) |
↑169 (↑108 to ↑248) |
ND |
|
Lopinavir/ritonavire 533 mg/133 mg b.i.d. for 2 weeks |
1,400 mg b.i.d. for 2 weeks |
18 |
↔f |
↔f |
↔f |
|
Lopinavir/ritonavire 400 mg/100 mg b.i.d. for 2 weeks |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
18 |
↑30 (↓15 to ↑47) |
↑37 (↓20 to ↑55) |
↑52 (↓28 to ↑82) |
|
Maraviroc 300 mg b.i.d. for 10 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 20 days |
14 |
↑52 (↑27 to ↑82) |
↑149 (↑119 to ↑182) |
↑374 (↑303 to ↑457) |
|
Maraviroc 300 mg q.d. for 10 days |
1,400 mg q.d. plus ritonavir 100 mg q.d. for 20 days |
14 |
↑45 (↑20 to ↑74) |
↑126 (↑99 to ↑158) |
↑80 (↑53 to ↑113) |
|
Methadone 70 to 120 mg q.d. for 2 weeks |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
19 |
R-Methadone (active) |
||
|
↓21g (↓30 to ↓12) |
↓18g (↓27 to ↓8) |
↓11g (↓21 to ↑1) |
|||
|
S-Methadone (inactive) |
|||||
|
↓43g (↓49 to ↓37) |
↓43g (↓50 to ↓36) |
↓41g (↓49 to ↓31) |
|||
|
Nevirapine 200 mg b.i.d. for 2 weeksh |
1,400 mg b.i.d. for 2 weeks |
17 |
↑25 (↑14 to ↑37) |
↑29 (↑19 to ↑40) |
↑34 (↑20 to ↑49) |
|
Nevirapine 200 mg b.i.d. for 2 weeksh |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
17 |
↑13 (↑3 to ↑24) |
↑14 (↑5 to ↑24) |
↑22 (↑9 to ↑35) |
|
Norethindronec 0.5 mg q.d. for 21 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 21 days |
25 |
↓38 (↓32 to ↓44) |
↓34 (↓30 to ↓37) |
↓26 (↓20 to ↓32) |
|
Phenytoin 300 mg q.d. for 10 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 10 days |
14 |
↓20 (↓12 to ↓27) |
↓22 (↓17 to ↓27) |
↓29 (↓23 to ↓34) |
|
Rifabutin 150 mg every other day for 2 weeks i |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 2 weeks |
15 |
↓14 (↓28 to ↑4) |
↔ |
↑28 (↑12 to ↑46) |
|
(25-O-desacetylrifabutin metabolite) |
↑579 (↑479 to ↑698) |
↑1,120 (↑965 to ↑1,300) |
↑2,510 (↑1,910 to ↑3,300) |
||
|
Rifabutin + 25-O- desacetylrifabutin metabolite |
NA |
↑64 (↑46 to ↑84) |
NA |
||
|
Rosuvastatin 10 mg single dose |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 7 days |
↑45 |
↑8 |
NA |
|
|
Telaprevir 750 mg q. 8 hr for 10 days |
700 mg b.i.d. plus ritonavir 100 mg b.i.d. for 20 days |
18 |
↓33 (↓29 to ↓37) |
↓32 (↓28 to ↓37) |
↓30 (↓23 to ↓36) |
aConcomitant medication is also shown in this column where appropriate.
bComparison arm of atazanavir 300 mg q.d. plus ritonavir 100 mg q.d. for 10 days.
cAdministered as a combination oral contraceptive tablet: ethinyl estradiol 0.035 mg/norethindrone 0.5 mg.
dSubjects were receiving LEXIVA/ritonavir for 10 days prior to the 4-day treatment period with both ketoconazole and LEXIVA/ritonavir.
eData represent lopinavir concentrations.
fCompared with lopinavir 400 mg/ritonavir 100 mg b.i.d. for 2 weeks.
gDose normalized to methadone 100 mg. The unbound concentration of the active moiety, R‑methadone, was unchanged.
hSubjects were receiving nevirapine for at least 12 weeks prior to trial.
iComparison arm of rifabutin 300 mg q.d. for 2 weeks. AUC is AUC(0-48 hr).
↑ = Increase; ↓= Decrease; ↔ = No change (↑or ↓less than 10%); ND = Interaction cannot be determined as Cmin was below the lower limit of quantitation.
|
Coadministered |
Dose of AGENERASE |
% Change in Pharmacokinetic Parameters of Coadministered Drug (90% CI) |
|||
|
n |
Cmax |
AUC |
Cmin |
||
|
Abacavir 300 mg b.i.d. for 2 to 3 weeks |
900 mg b.i.d for 2 to 3 weeks |
4 |
↔a |
↔a |
↔a |
|
Clarithromycin 500 mg b.i.d. for 4 days |
1,200 mg b.i.d. for 4 days |
12 |
↓10 (↓24 to ↑7) |
↔ |
↔ |
|
Delavirdine 600 mg b.i.d. for 10 days |
600 mg b.i.d. for 10 days |
9 |
↓47b |
↓61b |
↓88b |
|
Ethinyl estradiol 0.035 mg for 1 cycle |
1,200 mg b.i.d. for 28 days |
10 |
↔ |
↔ |
↑32 (↓3 to ↑79) |
|
Indinavir 800 mg t.i.d. for 2 weeks (fasted) |
750 mg or 800 mg t.i.d. for 2 weeks (fasted) |
9 |
↓22a |
↓38a |
↓27a |
|
Ketoconazole 400 mg single dose |
1,200 mg single dose |
12 |
↑19 (↑8 to ↑33) |
↑44 (↑31 to ↑59) |
NA |
|
Lamivudine 150 mg single dose |
600 mg single dose |
11 |
↔ |
↔ |
NA |
|
Methadone 44 to 100 mg q.d. for >30 days |
1,200 mg b.i.d. for 10 days |
16 |
R-Methadone (active) |
||
|
↓25 (↓32 to ↓18) |
↓13 (↓21 to ↓5) |
↓21 (↓32 to ↓9) |
|||
|
S-Methadone (inactive) |
|||||
|
↓48 (↓55 to ↓40) |
↓40 (↓46 to ↓32) |
↓53 (↓60 to ↓43) |
|||
|
Nelfinavir 750 mg t.i.d. for 2 weeks (fed) |
750 mg or 800 mg t.i.d. for 2 weeks (fed) |
6 |
↑12a |
↑15a |
↑14a |
|
Norethindrone 1 mg for 1 cycle |
1,200 mg b.i.d. for 28 days |
10 |
↔ |
↑18 (↑1 to ↑38) |
↑45 (↑13 to ↑88) |
|
Rifabutin 300 mg q.d. for 10 days |
1,200 mg b.i.d. for 10 days |
5 |
↑119 (↑82 to ↑164) |
↑193 (↑156 to ↑235) |
↑271 (↑171 to ↑409) |
|
Rifampin 300 mg q.d. for 4 days |
1,200 mg b.i.d. for 4 days |
11 |
↔ |
↔ |
ND |
|
Saquinavir 800 mg t.i.d. for 2 weeks (fed) |
750 mg or 800 mg t.i.d. for 2 weeks (fed) |
7 |
↑21a |
↓19a |
↓48a |
|
Zidovudine 300 mg single dose |
600 mg single dose |
12 |
↑40 (↑14 to ↑71) |
↑31 (↑19 to ↑45) |
NA |
aCompared with historical data.
bMedian percent change; confidence interval not reported.
↑ = Increase; ↓ = Decrease; ↔= No change (↑or ↓ less than 10%); NA = Cmin not calculated for single-dose trial; ND = Interaction cannot be determined as Cmin was below the lower limit of quantitation.
Mechanism of Action: Fosamprenavir is a prodrug that is rapidly hydrolyzed to amprenavir by cellular phosphatases in the gut epithelium as it is absorbed. Amprenavir is an inhibitor of HIV-1 protease. Amprenavir binds to the active site of HIV-1 protease and thereby prevents the processing of viral Gag and Gag-Pol polyprotein precursors, resulting in the formation of immature non-infectious viral particles.
Antiviral Activity: Fosamprenavir has little or no antiviral activity in cell culture. The antiviral activity of amprenavir was evaluated against HIV-1 IIIB in both acutely and chronically infected lymphoblastic cell lines (MT-4, CEM-CCRF, H9) and in peripheral blood lymphocytes in cell culture. The 50% effective concentration (EC50) of amprenavir ranged from 0.012 to 0.08 microM in acutely infected cells and was 0.41 microM in chronically infected cells (1 microM = 0.50 mcg per mL). The median EC50 value of amprenavir against HIV-1 isolates from clades A to G was 0.00095 microM in peripheral blood mononuclear cells (PBMCs). Similarly, the EC50 values for amprenavir against monocytes/macrophage tropic HIV-1 isolates (clade B) ranged from 0.003 to 0.075 microM in monocyte/macrophage cultures. The EC50 values of amprenavir against HIV-2 isolates grown in PBMCs were higher than those for HIV-1 isolates, and ranged from 0.003 to 0.11 microM. Amprenavir exhibited synergistic anti–HIV–1 activity in combination with the nucleoside reverse transcriptase inhibitors (NRTIs) abacavir, didanosine, lamivudine, stavudine, tenofovir, and zidovudine; the non-nucleoside reverse transcriptase inhibitors (NNRTIs) delavirdine and efavirenz; and the protease inhibitors atazanavir and saquinavir. Amprenavir exhibited additive anti–HIV–1 activity in combination with the NNRTI nevirapine, the protease inhibitors indinavir, lopinavir, nelfinavir, and ritonavir; and the fusion inhibitor enfuvirtide. These drug combinations have not been adequately studied in humans.
Resistance: HIV-1 isolates with decreased susceptibility to amprenavir have been selected in cell culture and obtained from subjects treated with fosamprenavir. Genotypic analysis of isolates from treatment-naive subjects failing amprenavir-containing regimens showed substitutions in the HIV-1 protease gene resulting in amino acid substitutions primarily at positions V32I, M46I/L, I47V, I50V, I54L/M, and I84V, as well as substitutions in the p7/p1 and p1/p6 Gag and Gag-Pol polyprotein precursor cleavage sites. Some of these amprenavir resistance-associated substitutions have also been detected in HIV-1 isolates from antiretroviral-naive subjects treated with LEXIVA. Of the 488 antiretroviral-naive subjects treated with LEXIVA 1,400 mg twice daily or LEXIVA 1,400 mg plus ritonavir 200 mg once daily in Trials APV30001 and APV30002, respectively, 61 subjects (29 receiving LEXIVA and 32 receiving LEXIVA/ritonavir) with virologic failure (plasma HIV-1 RNA greater than 1,000 copies per mL on 2 occasions on or after Week 12) were genotyped. Five of the 29 antiretroviral-naive subjects (17%) receiving LEXIVA without ritonavir in Trial APV30001 had evidence of genotypic resistance to amprenavir: I54L/M (n = 2), I54L + L33F (n = 1), V32I + I47V (n = 1), and M46I + I47V (n = 1). No amprenavir resistance-associated substitutions were detected in antiretroviral-naive subjects treated with LEXIVA/ritonavir for 48 weeks in Trial APV30002. However, the M46I and I50V substitutions were detected in isolates from 1 virologic failure subject receiving LEXIVA/ritonavir once daily at Week 160 (HIV-1 RNA greater than 500 copies per mL). Upon retrospective analysis of stored samples using an ultrasensitive assay, these resistant substitutions were traced back to Week 84 (76 weeks prior to clinical virologic failure).
Cross-Resistance: Varying degrees of cross-resistance among HIV-1 protease inhibitors have been observed. An association between virologic response at 48 weeks (HIV-1 RNA level less than 400 copies per mL) and protease inhibitor-resistance substitutions detected in baseline HIV-1 isolates from protease inhibitor-experienced subjects receiving LEXIVA/ritonavir twice daily (n = 88), or lopinavir/ritonavir twice daily (n = 85) in Trial APV30003 is shown in Table 14. The majority of subjects had previously received either one (47%) or 2 protease inhibitors (36%), most commonly nelfinavir (57%) and indinavir (53%). Out of 102 subjects with baseline phenotypes receiving twice-daily LEXIVA/ritonavir, 54% (n = 55) had resistance to at least one protease inhibitor, with 98% (n = 54) of those having resistance to nelfinavir. Out of 97 subjects with baseline phenotypes in the lopinavir/ritonavir arm, 60% (n = 58) had resistance to at least one protease inhibitor, with 97% (n = 56) of those having resistance to nelfinavir.
|
Protease Inhibitor Resistance-Associated Substitutionsb |
LEXIVA/Ritonavir b.i.d. (n = 88) |
Lopinavir/Ritonavir b.i.d. (n = 85) |
||
|
D30N |
21/22 |
95% |
17/19 |
89% |
|
N88D/S |
20/22 |
91% |
12/12 |
100% |
|
L90M |
16/31 |
52% |
17/29 |
59% |
|
M46I/L |
11/22 |
50% |
12/24 |
50% |
|
V82A/F/T/S |
2/9 |
22% |
6/17 |
35% |
|
I54V |
2/11 |
18% |
6/11 |
55% |
|
I84V |
1/6 |
17% |
2/5 |
40% |
aResults should be interpreted with caution because the subgroups were small.
bMost subjects had greater than 1 protease inhibitor resistance-associated substitution at baseline.
The virologic response based upon baseline phenotype was assessed. Baseline isolates from protease inhibitor-experienced subjects responding to LEXIVA/ritonavir twice daily had a median shift in susceptibility to amprenavir relative to a standard wild-type reference strain of 0.7 (range: 0.1 to 5.4, n = 62), and baseline isolates from individuals failing therapy had a median shift in susceptibility of 1.9 (range: 0.2 to 14, n = 29). Because this was a select patient population, these data do not constitute definitive clinical susceptibility break points. Additional data are needed to determine clinically relevant break points for LEXIVA.
Isolates from 15 of the 20 subjects receiving twice-daily LEXIVA/ritonavir up to Week 48 and experiencing virologic failure/ongoing replication were subjected to genotypic analysis. The following amprenavir resistance-associated substitutions were found either alone or in combination: V32I, M46I/L, I47V, I50V, I54L/M, and I84V. Isolates from 4 of the 16 subjects continuing to receive twice-daily LEXIVA/ritonavir up to Week 96 who experienced virologic failure underwent genotypic analysis. Isolates from 2 subjects contained amprenavir resistance-associated substitutions: V32I, M46I, and I47V in 1 isolate and I84V in the other.
NONCLINICAL TOXICOLOGY
In long-term carcinogenicity studies, fosamprenavir was administered orally for up to 104 weeks at doses of 250, 400, or 600 mg per kg per day in mice and at doses of 300, 825, or 2,250 mg per kg per day in rats. Exposures at these doses were 0.3- to 0.7-fold (mice) and 0.7- to 1.4-fold (rats) those in humans given 1,400 mg twice daily of fosamprenavir alone, and 0.2- to 0.3-fold (mice) and 0.3- to 0.7-fold (rats) those in humans given 1,400 mg once daily of fosamprenavir plus 200 mg ritonavir once daily. Exposures in the carcinogenicity studies were 0.1- to 0.3-fold (mice) and 0.3- to 0.6-fold (rats) those in humans given 700 mg of fosamprenavir plus 100 mg ritonavir twice daily. There was an increase in hepatocellular adenomas and hepatocellular carcinomas at all doses in male mice and at 600 mg per kg per day in female mice, and in hepatocellular adenomas and thyroid follicular cell adenomas at all doses in male rats, and at 835 mg per kg per day and 2,250 mg per kg per day in female rats. The relevance of the hepatocellular findings in the rodents for humans is uncertain. Repeat dose studies with fosamprenavir in rats produced effects consistent with enzyme induction, which predisposes rats, but not humans, to thyroid neoplasms. In addition, in rats only there was an increase in interstitial cell hyperplasia at 825 mg per kg per day and 2,250 mg per kg per day, and an increase in uterine endometrial adenocarcinoma at 2,250 mg per kg per day. The incidence of endometrial findings was slightly increased over concurrent controls, but was within background range for female rats. The relevance of the uterine endometrial adenocarcinoma findings in rats for humans is uncertain.
Fosamprenavir was not mutagenic or genotoxic in a battery of in vitro and in vivo assays. These assays included bacterial reverse mutation (Ames), mouse lymphoma, rat micronucleus, and chromosome aberrations in human lymphocytes.
The effects of fosamprenavir on fertility and general reproductive performance were investigated in male (treated for 4 weeks before mating) and female rats (treated for 2 weeks before mating through postpartum day 6). Systemic exposures (AUC0-24 hr) to amprenavir in these studies were 3 (males) to 4 (females) times higher than exposures in humans following administration of the MRHD of fosamprenavir alone or similar to those seen in humans following administration of fosamprenavir in combination with ritonavir. Fosamprenavir did not impair mating or fertility of male or female rats and did not affect the development and maturation of sperm from treated rats.
CLINICAL STUDIES
APV30001: A randomized, open-label trial evaluated treatment with LEXIVA Tablets (1,400 mg twice daily) versus nelfinavir (1,250 mg twice daily) in 249 antiretroviral treatment-naive subjects. Both groups of subjects also received abacavir (300 mg twice daily) and lamivudine (150 mg twice daily).
The mean age of the subjects in this trial was 37 years (range: 17 to 70 years); 69% of the subjects were male, 20% were CDC Class C (AIDS), 24% were Caucasian, 32% were black, and 44% were Hispanic. At baseline, the median CD4+ cell count was 212 cells per mm3 (range: 2 to 1,136 cells per mm3; 18% of subjects had a CD4+ cell count of less than 50 cells per mm3 and 30% were in the range of 50 to less than 200 cells per mm3). Baseline median HIV-1 RNA was 4.83 log10 copies per mL (range: 1.69 to 7.41 log10 copies per mL; 45% of subjects had greater than 100,000 copies per mL).
The outcomes of randomized treatment are provided in Table 15.
|
Outcome (Rebound or discontinuation = failure) |
LEXIVA 1,400 mg b.i.d. (n = 166) |
Nelfinavir 1,250 mg b.i.d. (n = 83) |
|
Respondera |
66% (57%) |
52% (42%) |
|
Virologic failure |
19% |
32% |
|
Rebound |
16% |
19% |
|
Never suppressed through Week 48 |
3% |
13% |
|
Clinical progression |
1% |
1% |
|
Death |
0% |
1% |
|
Discontinued due to adverse reactions |
4% |
2% |
|
Discontinued due to other reasonsb |
10% |
10% |
aSubjects achieved and maintained confirmed HIV-1 RNA less than 400 copies per mL (less than 50 copies per mL) through Week 48 (Roche AMPLICOR HIV-1 MONITOR Assay Version 1.5).
bIncludes consent withdrawn, lost to follow up, protocol violations, those with missing data, and other reasons.
Treatment response by viral load strata is shown in Table 16.
|
Screening Viral Load HIV-1 RNA (copies/mL) |
LEXIVA 1,400 mg b.i.d. |
Nelfinavir 1,250 mg b.i.d. |
||
|
<400 copies/mL |
n |
<400 copies/mL |
n |
|
|
100,000 |
65% |
93 |
65% |
46 |
|
>100,000 |
67% |
73 |
36% |
37 |
Through 48 weeks of therapy, the median increases from baseline in CD4+ cell counts were 201 cells per mm3 in the group receiving LEXIVA and 216 cells per mm3 in the nelfinavir group.
APV30002: A randomized, open-label trial evaluated treatment with LEXIVA Tablets (1,400 mg once daily) plus ritonavir (200 mg once daily) versus nelfinavir (1,250 mg twice daily) in 649 treatment-naive subjects. Both treatment groups also received abacavir (300 mg twice daily) and lamivudine (150 mg twice daily).
The mean age of the subjects in this trial was 37 years (range: 18 to 69 years); 73% of the subjects were male, 22% were CDC Class C, 53% were Caucasian, 36% were black, and 8% were Hispanic. At baseline, the median CD4+ cell count was 170 cells per mm3 (range: 1 to 1,055 cells per mm3; 20% of subjects had a CD4+ cell count of less than 50 cells per mm3 and 35% were in the range of 50 to less than 200 cells per mm3). Baseline median HIV-1 RNA was 4.81 log10 copies per mL (range: 2.65 to 7.29 log10 copies per mL; 43% of subjects had greater than 100,000 copies per mL).
The outcomes of randomized treatment are provided in Table 17.
Table 17. Outcomes of Randomized Treatment Through Week 48 (APV30002)
|
Outcome (Rebound or discontinuation = failure) |
LEXIVA 1,400 mg q.d./ Ritonavir 200 mg q.d. (n = 322) |
Nelfinavir 1,250 mg b.i.d. (n = 327) |
|
Respondera |
69% (58%) |
68% (55%) |
|
Virologic failure |
6% |
16% |
|
Rebound |
5% |
8% |
|
Never suppressed through Week 48 |
1% |
8% |
|
Death |
1% |
0% |
|
Discontinued due to adverse reactions |
9% |
6% |
|
Discontinued due to other reasonsb |
15% |
10% |
aSubjects achieved and maintained confirmed HIV-1 RNA less than 400 copies per mL (less than 50 copies per mL) through Week 48 (Roche AMPLICOR HIV-1 MONITOR Assay Version 1.5).
bIncludes consent withdrawn, lost to follow up, protocol violations, those with missing data, and other reasons.
Treatment response by viral load strata is shown in Table 18.
|
Screening Viral Load HIV-1 RNA |
LEXIVA 1,400 mg q.d./ Ritonavir 200 mg q.d. |
Nelfinavir 1,250 mg b.i.d. |
||
|
(copies/mL) |
<400 copies/mL |
n |
<400 copies/mL |
n |
|
100,000 |
72% |
197 |
73% |
194 |
|
>100,000 |
66% |
125 |
64% |
133 |
Through 48 weeks of therapy, the median increases from baseline in CD4+ cell counts were 203 cells per mm3 in the group receiving LEXIVA and 207 cells per mm3 in the nelfinavir group.
APV30003: A randomized, open-label, multicenter trial evaluated 2 different regimens of LEXIVA plus ritonavir (LEXIVA Tablets 700 mg twice daily plus ritonavir 100 mg twice daily or LEXIVA Tablets 1,400 mg once daily plus ritonavir 200 mg once daily) versus lopinavir/ritonavir (400 mg/100 mg twice daily) in 315 subjects who had experienced virologic failure to 1 or 2 prior protease inhibitor-containing regimens.
The mean age of the subjects in this trial was 42 years (range: 24 to 72 years); 85% were male, 33% were CDC Class C, 67% were Caucasian, 24% were black, and 9% were Hispanic. The median CD4+ cell count at baseline was 263 cells per mm3 (range: 2 to 1,171 cells per mm3). Baseline median plasma HIV-1 RNA level was 4.14 log10 copies per mL (range: 1.69 to 6.41 log10 copies per mL).
The median durations of prior exposure to NRTIs were 257 weeks for subjects receiving LEXIVA/ritonavir twice daily (79% had greater than or equal to 3 prior NRTIs) and 210 weeks for subjects receiving lopinavir/ritonavir (64% had greater than or equal to 3 prior NRTIs). The median durations of prior exposure to protease inhibitors were 149 weeks for subjects receiving LEXIVA/ritonavir twice daily (49% received greater than or equal to 2 prior protease inhibitors) and 130 weeks for subjects receiving lopinavir/ritonavir (40% received greater than or equal to 2 prior protease inhibitors).
The time-averaged changes in plasma HIV-1 RNA from baseline (AAUCMB) at 48 weeks (the endpoint on which the trial was powered) were -1.4 log10 copies per mL for twice-daily LEXIVA/ritonavir and -1.67 log10 copies per mL for the lopinavir/ritonavir group.
The proportions of subjects who achieved and maintained confirmed HIV-1 RNA less than 400 copies per mL (secondary efficacy endpoint) were 58% with twice-daily LEXIVA/ritonavir and 61% with lopinavir/ritonavir (95% CI for the difference: -16.6, 10.1). The proportions of subjects with HIV-1 RNA less than 50 copies per mL with twice-daily LEXIVA/ritonavir and with lopinavir/ritonavir were 46% and 50%, respectively (95% CI for the difference: -18.3, 8.9). The proportions of subjects who were virologic failures were 29% with twice-daily LEXIVA/ritonavir and 27% with lopinavir/ritonavir.
The frequency of discontinuations due to adverse events and other reasons, and deaths were similar between treatment arms.
Through 48 weeks of therapy, the median increases from baseline in CD4+ cell counts were 81 cells per mm3 with twice-daily LEXIVA/ritonavir and 91 cells per mm3 with lopinavir/ritonavir.
This trial was not large enough to reach a definitive conclusion that LEXIVA/ritonavir and lopinavir/ritonavir are clinically equivalent.
Once-daily administration of LEXIVA plus ritonavir is not recommended for protease inhibitor-experienced patients. Through Week 48, 50% and 37% of subjects receiving LEXIVA 1,400 mg plus ritonavir 200 mg once daily had plasma HIV-1 RNA less than 400 copies per mL and less than 50 copies per mL, respectively.
Three open-label trials in pediatric subjects aged at least 4 weeks to 18 years were conducted. In one trial (APV29005), twice-daily dosing regimens (LEXIVA with or without ritonavir) were evaluated in combination with other antiretroviral agents in pediatric subjects aged 2 to 18 years. In a second trial (APV20002), twice-daily dosing regimens (LEXIVA with ritonavir) were evaluated in combination with other antiretroviral agents in pediatric subjects aged at least 4 weeks to less than 2 years. A third trial (APV20003) evaluated once-daily dosing of LEXIVA with ritonavir; the pharmacokinetic data from this trial did not support a once-daily dosing regimen in any pediatric patient population.
APV29005: LEXIVA: Twenty (18 therapy-naive and 2 therapy-experienced) pediatric subjects received LEXIVA Oral Suspension without ritonavir twice daily. At Week 24, 65% (13/20) achieved HIV-1 RNA less than 400 copies per mL, and the median increase from baseline in CD4+ cell count was 350 cells per mm3.
LEXIVA plus Ritonavir: Forty-nine protease inhibitor-naive and 40 protease inhibitor-experienced pediatric subjects received LEXIVA Oral Suspension or Tablets with ritonavir twice daily. At Week 24, 71% of protease inhibitor-naive (35/49) and 55% of protease inhibitor-experienced (22/40) subjects achieved HIV-1 RNA less than 400 copies per mL; median increases from baseline in CD4+ cell counts were 184 cells per mm3 and 150 cells per mm3 in protease inhibitor-naive and experienced subjects, respectively.
APV20002: Fifty-four pediatric subjects (49 protease inhibitor-naive and 5 protease inhibitor-experienced) received LEXIVA Oral Suspension with ritonavir twice daily. At Week 24, 72% of subjects achieved HIV-1 RNA less than 400 copies per mL. The median increases from baseline in CD4+ cell counts were 400 cells per mm3 in subjects aged at least 4 weeks to less than 6 months and 278 cells per mm3 in subjects aged 6 months to 2 years.
HOW SUPPLIED
LEXIVA Tablets, 700 mg, are pink, film-coated, capsule-shaped, biconvex tablets, with “GX LL7” debossed on one face.
Bottle of 60 with child-resistant closure (NDC 49702-207-18).
Store at controlled room temperature of 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) (see USP Controlled Room Temperature). Keep container tightly closed.
LEXIVA Oral Suspension, a white to off-white grape-bubblegum-peppermint–flavored suspension, contains 50 mg of fosamprenavir as fosamprenavir calcium equivalent to approximately 43 mg of amprenavir in each 1 mL.
Bottle of 225 mL with child-resistant closure (NDC 49702-208-53).
This product does not require reconstitution.
Store in refrigerator or at room temperature (5° to 30°C; 41° to 86°F). Shake vigorously before using. Do not freeze.
INFORMATION FOR PATIENTS
See FDA-approved Patient Labeling (Patient Information)
A statement to patients and healthcare providers is included on the product’s bottle label: ALERT: Find out about medicines that should NOT be taken with LEXIVA.
LEXIVA may interact with many drugs; therefore, patients should be advised to report to their healthcare provider the use of any other prescription or nonprescription medication or herbal products, particularly St. John’s wort.
Patients receiving PDE5 inhibitors should be advised that they may be at an increased risk of PDE5 inhibitor-associated adverse events, including hypotension, visual changes, and priapism, and should promptly report any symptoms to their healthcare provider.
Patients receiving hormonal contraceptives should be instructed to use alternate contraceptive measures during therapy with LEXIVA because hormonal levels may be altered, and if used in combination with LEXIVA and ritonavir, liver enzyme elevations may occur.
Patients should inform their healthcare provider if they have a sulfa allergy. The potential for cross-sensitivity between drugs in the sulfonamide class and fosamprenavir is unknown.
Patients should be informed that redistribution or accumulation of body fat may occur in patients receiving antiretroviral therapy, including LEXIVA, and that the cause and long-term health effects of these conditions are not known at this time.
LEXIVA is not a cure for HIV-1 infection and patients may continue to experience illnesses associated with HIV-1 infection, including opportunistic infections. Patients should remain under the care of a physician when using LEXIVA.
Patients should be advised to avoid doing things that can spread HIV-1 infection to others.
•
Do not share needles or other injection equipment.
•
Do not share personal items that can have blood or body fluids on them, like toothbrushes and razor blades.
•
Do not have any kind of sex without protection.
Always practice safe sex by using a latex or polyurethane condom to lower the chance of sexual contact with semen, vaginal secretions, or blood.•
Do not breastfeed.
We do not know if LEXIVA can be passed to your baby in your breast milk and whether it could harm your baby. Also, mothers with HIV-1 should not breastfeed because HIV-1 can be passed to the baby in the breast milk.
Patients should be told that sustained decreases in plasma HIV-1 RNA have been associated with a reduced risk of progression to AIDS and death. Patients should be advised to take LEXIVA every day as prescribed. LEXIVA must always be used in combination with other antiretroviral drugs. Patients should not alter the dose or discontinue therapy without consulting their physician. If a dose is missed, patients should take the dose as soon as possible and then return to their normal schedule. However, if a dose is skipped, the patient should not double the next dose.
Patients should be instructed to shake the bottle vigorously before each use and that refrigeration of the oral suspension may improve the taste for some patients.
LEXIVA and AGENERASE are registered trademarks of ViiV Healthcare.
The brands listed are trademarks of their respective owners and are not trademarks of ViiV Healthcare. The makers of these brands are not affiliated with and do not endorse ViiV Healthcare or its products.
Manufactured for:
|
ViiV Healthcare Research Triangle Park, NC 27709 |
Vertex Pharmaceuticals Incorporated Cambridge, MA 02139 |
by:
GlaxoSmithKline
Research Triangle Park, NC 27709
©2013, ViiV Healthcare. All rights reserved.
LXV:17PI
PHARMACIST-DETACH HERE AND GIVE INSTRUCTIONS TO PATIENT
_ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ __ _ _ _ _ _ _ _ _
SPL PATIENT PACKAGE INSERT
PATIENT INFORMATION
LEXIVA® (lex-EE-vah)
(fosamprenavir calcium)
Tablets
and
Oral Suspension
Important: LEXIVA can interact with other medicines and cause serious side effects. It is important to know the medicines that should not be taken with LEXIVA. See the section “Who should not take LEXIVA?”
Read this Patient Information before you start taking LEXIVA and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment.
What is LEXIVA?
LEXIVA is a prescription anti-HIV medicine used with other anti-HIV medicines to treat human immunodeficiency (HIV-1) infections in adults and children 4 weeks of age and older. LEXIVA is a type of anti-HIV medicine called a protease inhibitor. HIV-1 is the virus that causes AIDS (Acquired Immune Deficiency Syndrome).
When used with other anti-HIV medicines, LEXIVA may help:
1.Reduce the amount of HIV-1 in your blood. This is called “viral load”.2.Increase the number of white blood cells called CD4 (T) cells, which help fight off other infections. Reducing the amount of HIV-1 and increasing the CD4 (T) cell count may improve your immune system. This may reduce your risk of death or infections that can happen when your immune system is weak (opportunistic infections).
It is not known if LEXIVA is safe and effective in children less than 4 weeks of age.
LEXIVA does not cure HIV-1 infection or AIDS. People taking LEXIVA may develop infections or other conditions associated with HIV-1 infection, including opportunistic infections (for example, pneumonia and herpes virus infections).
You should remain under the care of your healthcare provider when using LEXIVA.
Avoid doing things that can spread HIV-1 infection to others.
•
Do not share needles or other injection equipment.
•
Do not share personal items that can have blood or body fluids on them, like toothbrushes and razor blades.
•
Do not have any kind of sex without protection.
Always practice safe sex by using a latex or polyurethane condom to lower the chance of sexual contact with semen, vaginal secretions, or blood.
Ask your healthcare provider if you have any questions on how to prevent passing HIV to other people.
Who should not take LEXIVA?
Do not take LEXIVA if you take any of the following medicines:
•alfuzosin (UROXATRAL
®
)•flecainide (TAMBOCOR™)•propafenone (RYTHMOL SR
®
)•rifampin (RIFADIN
®
, RIFAMATE
®
, RIFATER
®
, RIMACTANE
®
)•ergot including:∘dihydroergotamine mesylate (D.H.E. 45
®
, MIGRANAL
®
)∘ergotamine tartrate (CAFERGOT
®
, MIGERGOT
®
, ERGOMAR
®
, MEDIHALER ERGOTAMINE
®
)∘methylergonovine (METHERGINE
®
)•St. John’s wort (
Hypericum perforatum
)•lovastatin (ADVICOR
®
, ALTOPREV
®
, MEVACOR
®
)•simvastatin (ZOCOR
®,
VYTORIN
®
, SIMCOR
®
)•pimozide (ORAP
®
)•delavirdine mesylate (RESCRIPTOR
®
)•sildenafil (REVATIO
®
), for treatment of pulmonary arterial hypertension•triazolam (HALCION
®
)
Serious problems can happen if you or your child take any of the medicines listed above with LEXIVA.
Do not take LEXIVA if you are allergic to AGENERASE® (amprenavir), fosamprenavir calcium, or any of the ingredients in LEXIVA. See the end of this leaflet for a complete list of ingredients in LEXIVA.
What should I tell my healthcare provider before taking LEXIVA?
Before taking LEXIVA, tell your healthcare provider if you:
•are allergic to medicines that contain sulfa•have liver problems, including hepatitis B or C•have kidney problems•have high blood sugar (diabetes)•have hemophilia•have any other medical condition•are pregnant or plan to become pregnant. It is not known if LEXIVA will harm your unborn baby.
Pregnancy Registry. There is a pregnancy registry for women who take antiviral medicines during pregnancy. The purpose of the registry is to collect information about the health of you and your baby. Talk to your healthcare provider about how you can take part in this registry.
•
Do not breastfeed.
We do not know if LEXIVA can be passed to your baby in your breast milk and whether it could harm your baby. Also, mothers with HIV-1 should not breastfeed because HIV-1 can be passed to the baby in the breast milk.
Tell your healthcare provider about all prescription and non-prescription medicines you take. Also tell your healthcare provider about any vitamins, herbal supplements, and dietary supplements you are taking.
Taking LEXIVA with certain other medicines may cause serious side effects. LEXIVA may affect the way other medicines work, and other medicines may affect how LEXIVA works.
Especially tell your healthcare provider if you take estrogen-based contraceptives (birth control pills). LEXIVA may reduce effectiveness of estrogen-based contraceptives. During treatment with LEXIVA, you should use a different contraceptive method.
Know all the medicines that you take. Keep a list of them with you to show healthcare providers and pharmacists when you get a new medicine.
How should I take LEXIVA?
•
Stay under the care of a healthcare provider while taking LEXIVA.
•Take LEXIVA exactly as prescribed by your healthcare provider.•Do not change your dose or stop taking LEXIVA without talking with your healthcare provider.•If your child is taking LEXIVA, your child’s healthcare provider will decide the right dose based on your child’s weight.•You can take LEXIVA Tablets with or without food.•
Adults should take LEXIVA Oral Suspension without food.
•
Children should take LEXIVA Oral Suspension with food.
If your child vomits within 30 minutes after taking a dose of LEXIVA, the dose should be repeated.•Shake LEXIVA Oral Suspension well before each use.•If you miss a dose of LEXIVA, take the next dose as soon as possible and then take your next dose at the regular time. Do not double the next dose. If you take too much LEXIVA, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of LEXIVA?
LEXIVA may cause serious side effects including:
•
Severe skin rash.
LEXIVA may cause severe or life-threatening skin reactions or rash.
If you get a rash with any of the following symptoms, stop taking LEXIVA and call your healthcare provider or get medical help right away:
∘hives or sores in your mouth, or your skin blisters and peels∘trouble swallowing or breathing∘swelling of your face, eyes, lips, tongue, or throat•
Liver problems.
Your healthcare provider should do blood tests before and during your treatment with LEXIVA to check your liver function. Some people with liver problems, including hepatitis B or C, may have an increased risk of developing worsening liver problem during treatment with LEXIVA.•
Diabetes and high blood sugar (hyperglycemia).
Some people who take protease inhibitors, including LEXIVA, can get high blood sugar, develop diabetes, or your diabetes can get worse. Tell your healthcare provider if you notice an increase in thirst or urinate often while taking LEXIVA.•
Changes in your immune system (Immune Reconstitution Syndrome)
can happen when you start taking HIV medicines. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Call your healthcare provider right away if you start having new symptoms after starting your HIV medicine.•
Changes in body fat.
These changes can happen in people who take antiretroviral therapy. The changes may include an increased amount of fat in the upper back and neck (“buffalo hump”), breast, and around the back, chest, and stomach area. Loss of fat from the legs, arms, and face may also happen. The exact cause and long-term health effects of these conditions are not known. •
Changes in blood tests
. Some people have changes in blood tests while taking LEXIVA. These include increases seen in liver function tests, blood fat levels, and decreases in white blood cells. Your healthcare provider should do regular blood tests before and during your treatment with LEXIVA.•
Increased bleeding problems in some people with hemophilia.
Some people with hemophilia have increased bleeding with protease inhibitors, including LEXIVA.•
Kidney stones.
Some people have developed kidney stones while taking LEXIVA. Tell your healthcare provider right away if you develop signs or symptoms of kidney stones:∘pain in your side∘blood in your urine∘pain when you urinate
The most common side effects of LEXIVA in adults include:
•nausea •vomiting•diarrhea•headache
Vomiting is the most common side effect in children when taking LEXIVA.
Tell your healthcare provider about any side effect that bothers you or that does not go away.
These are not all the possible side effects of LEXIVA. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store LEXIVA?
•Store LEXIVA Tablets at room temperature between 68°F to 77°F (20°C to 25°C).•Keep the bottle of LEXIVA Tablets tightly closed.•Store LEXIVA Oral Suspension between 41°F to 86°F (5°C to 30°C). Refrigeration of LEXIVA Oral Suspension may improve taste for some people.•Do not freeze.
Keep LEXIVA and all medicines out of the reach of children.
General information about LEXIVA
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use LEXIVA for a condition for which it was not prescribed. Do not give LEXIVA to other people, even if they have the same symptoms you have. It may harm them.
This leaflet summarizes the most important information about LEXIVA. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about LEXIVA that is written for health professionals.
For more information call 877-844-8872 or go to www.LEXIVA.com.
What are the ingredients in LEXIVA?
Tablets:
Active ingredient: fosamprenavir calcium
Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and povidone K30. The tablet film-coating contains the inactive ingredients hypromellose, iron oxide red, titanium dioxide, and triacetin.
Oral Suspension:
Active ingredient: fosamprenavir calcium
Inactive ingredients: artificial grape-bubblegum flavor, calcium chloride dihydrate, hypromellose, methylparaben, natural peppermint flavor, polysorbate 80, propylene glycol, propylparaben, purified water, and sucralose.
This Patient Information has been approved by the U.S. Food and Drug Administration.
LEXIVA and AGENERASE are registered trademarks of ViiV Healthcare.
The brands listed are trademarks of their respective owners and are not trademarks of ViiV Healthcare. The makers of these brands are not affiliated with and do not endorse ViiV Healthcare or its products.
Manufactured for:
|
ViiV Healthcare Research Triangle Park, NC 27709 |
Vertex Pharmaceuticals Incorporated Cambridge, MA 02139 |
by:
GlaxoSmithKline
Research Triangle Park, NC 27709
©2013, ViiV Healthcare. All rights reserved.
February 2013
LXV:14PIL
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
DRUG: LEXIVA
GENERIC: fosamprenavir calcium
DOSAGE: TABLET, FILM COATED
ADMINSTRATION: ORAL
NDC: 49349-811-02
ACTIVE INGREDIENT(S):
- FOSAMPRENAVIR CALCIUM 700mg in 1
INACTIVE INGREDIENT(S):
- FERRIC OXIDE RED
- HYPROMELLOSES
- Povidone K30
- TITANIUM DIOXIDE
- SILICON DIOXIDE
- CELLULOSE, MICROCRYSTALLINE
- CROSCARMELLOSE SODIUM
- MAGNESIUM STEARATE
- TRIACETIN
COLOR: pink
SHAPE: CAPSULE
SCORE: No score
SIZE: 21 mm
IMPRINT: GX;LL7
PACKAGING: 30 in 1 BLISTER PACK

LEXIVAfosamprenavir calcium TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||