Losartan Potassium
FULL PRESCRIBING INFORMATION: CONTENTS*
- BOXED WARNING
- LOSARTAN POTASSIUM DESCRIPTION
- CLINICAL PHARMACOLOGY
- PHARMACOKINETICS
- PHARMACODYNAMICS AND CLINICAL EFFECTS
- INDICATIONS & USAGE
- LOSARTAN POTASSIUM CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- INFORMATION FOR PATIENTS
- DRUG INTERACTIONS
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- PREGNANCY
- NURSING MOTHERS
- PEDIATRIC USE
- GERIATRIC USE
- LOSARTAN POTASSIUM ADVERSE REACTIONS
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- INFORMATION FOR PATIENTS
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
BOXED WARNING
USE IN PREGNANCYWhen used in pregnancy during the second and third trimesters, drugs that act directly on the renin-angiotensin system can cause injury and even death to the developing fetus. When pregnancy is detected, losartan potassium tablets should be discontinued as soon as possible. See WARNINGS, Fetal/Neonatal Morbidity and Mortality.
LOSARTAN POTASSIUM DESCRIPTION

CLINICAL PHARMACOLOGY
Mechanism of ActionPHARMACOKINETICS
GeneralSpecial Populations
Pediatric
****
Geriatric and gender
Race
Renal insufficiency
Hepatic insufficiency
Drug Interactions
PHARMACODYNAMICS AND CLINICAL EFFECTS
Adult HypertensionPediatric Hypertension
Reduction in the Risk of Stroke

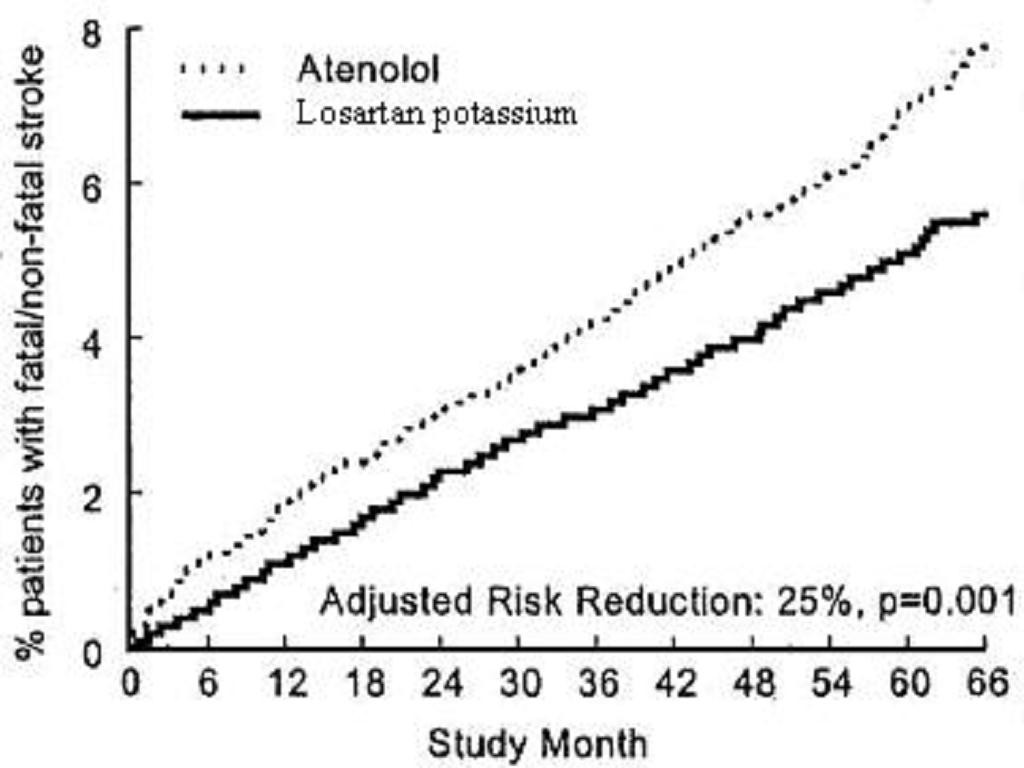
**
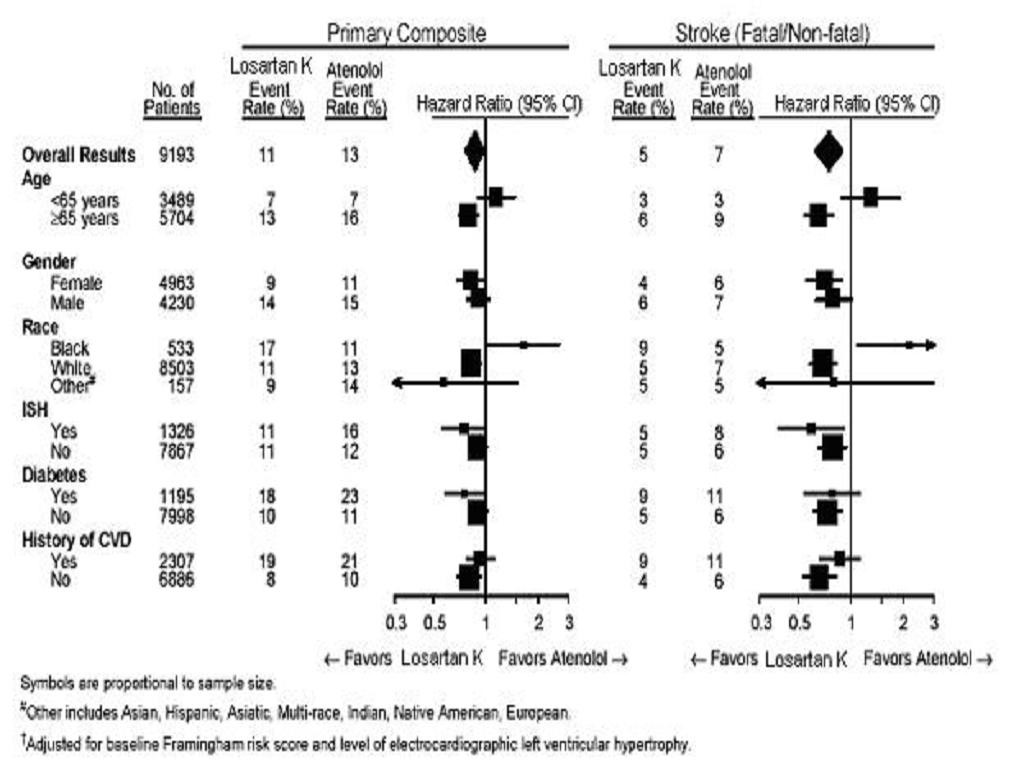
Race
Nephropathy in Type 2 Diabetic Patients

INDICATIONS AND USAGE
INDICATIONS & USAGE
HypertensionHypertensive Patients With Left Ventricular Hypertrophy
Nephropathy in Type 2 Diabetic Patients
LOSARTAN POTASSIUM CONTRAINDICATIONS
WARNINGS
Fetal/Neonatal Morbidity and MortalityHypotensionVolume-Depleted Patients
PRECAUTIONS
GeneralHypersensitivity
Impaired Hepatic Function
Impaired Renal Function
Electrolyte Imbalance
INFORMATION FOR PATIENTS
PregnancyPotassium Supplements
DRUG INTERACTIONS
Lithium
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) Including Selective Cyclooxygenase-2 Inhibitors (COX-2 Inhibitors)
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
PREGNANCY
Teratogenic EffectsPregnancy categories C (first trimester) and D (second and third trimesters)
NURSING MOTHERS
PEDIATRIC USE
GERIATRIC USE
Race
LOSARTAN POTASSIUM ADVERSE REACTIONS
Hypertension**
Pediatric Patients
Hypertensive Patients With Left Ventricular Hypertrophy
Nephropathy in Type 2 Diabetic Patients
Postmarketing Experience
Laboratory Test Findings
Creatinine, Blood Urea Nitrogen
Hemoglobin and Hematocrit
Liver Function Tests
OVERDOSAGE
DOSAGE & ADMINISTRATION
Adult Hypertensive PatientsPediatric Hypertensive Patients6 Years of Age
Preparation of Suspension (for 200 mL of a 2.5 mg/mL Suspension)
Hypertensive Patients With Left Ventricular Hypertrophy
Nephropathy in Type 2 Diabetic Patients
HOW SUPPLIED
INFORMATION FOR PATIENTS
PATIENT INFORMATION ABOUT LOSARTAN POTASSIUM TABLETS USP-
● alone or with other blood pressure medicines to lower high blood pressure (hypertension).
-
● to lower the chance of stroke in patients with high blood pressure and a heart problem called left ventricular hypertrophy. Losartan potassium tablets USP may not help Black patients with this problem.
-
● to slow the worsening of diabetic kidney disease (nephropathy) in patients with type 2 diabetes who have or had high blood pressure.
-
● Do not take losartan potassium tablets USP if you are allergic to any of the ingredients in losartan potassium tablets USP. See the end of this leaflet for a complete list of ingredients in losartan potassium tablets USP.
-
● are pregnant or planning to become pregnant. SeeWhat is the most important information I should know about losartan potassium tablets USP?
-
● are breast-feeding. It is not known if losartan potassium passes into your breast milk. You should choose either to take losartan potassium tablets USP or breast-feed, but not both.
-
● are vomiting a lot or having a lot of diarrhea
-
● have liver problems
-
● have kidney problems
-
● potassium supplements
-
● salt substitutes containing potassium
-
● water pills (diuretics)
-
● medicines used to treat pain and arthritis, called non-steroidal anti-inflammatory drugs (NSAIDs), including COX-2 inhibitors.
-
● Take losartan potassium tablets USP exactly as prescribed by your doctor. Your doctor may change your dose if needed.
-
● Losartan potassium tablets USP can be taken with or without food.
-
● If you miss a dose, take it as soon as you remember. If it is close to your next dose, do not take the missed dose. Just take the next dose at your regular time.
-
● If you take too many losartan potassium tablets USP, call your doctor or Poison Control Center, or go to the nearest hospital emergency room right away.
-
● Injury or death of unborn babies. SeeWhat is the most important information I should know about losartan potassium tablets USP?
-
● Allergic reaction. Symptoms of an allergic reaction are swelling of the face, lips, throat or tongue. Get emergency medical help right away and stop taking losartan potassium tablets USP.
-
● Low blood pressure (hypotension). Low blood pressure may cause you to feel faint or dizzy. Lie down if you feel faint or dizzy. Call your doctor right away.
-
● For people who already have kidney problems, you may see a worsening in how well your kidneys work. Call your doctor if you get swelling in your feet, ankles, or hands, or unexplained weight gain.
-
● colds(upper respiratory infection)
-
● dizziness
-
● stuffy nose
-
● back pain
-
● diarrhea
-
● tiredness
-
● low blood sugar
-
● chest pain
-
● high blood potassium
-
● low blood pressure
-
● Store losartan potassium tablets USP at 20to 25(68to 77
-
● Keep losartan potassium tablets USP in a tightly closed container that protects the medicine from light.
-
● Keep losartan potassium tablets USP and all medicines out of the reach of children.
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION


Losartan PotassiumLosartan Potassium TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!