Medicated
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active Ingredients
- Purpose
- Medicated Uses
- Warnings
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children
- Directions
- Inactive ingredients
- disclaimer
- Side Effects
- Principal Display Panel
FULL PRESCRIBING INFORMATION
Active Ingredients
Menthol 0.15%
Zinc oxide 1.0%
Purpose
External analgesic
Skin protectant
Medicated Uses
Uses for the temporary relief of paina and itching due to:
- monor cuts
- sunburn
- insect bites
- poison ivy
- poison oak
- poison sumac
- scrapes
- minor burns
- minor skin irritations
dries the oozing and weeping of:
- poison ivy
- poison oak
- poison sumac
Warnings
For external use only
When using this product
avoid contact with the eyes
Stop use and ask a doctor if
condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away
Directions
adults and children 2 years of age and older - apply to affected area not more than 3 to 4 times daily
children under 2 years of age - do not use, consult a doctor
- for best results dry skin thoroughly before applying
Inactive ingredients
talc, acacia, eucalyptol, methyl salicylate, salicylic acid, thymol, zinc stearate
disclaimer
This product is sold by weight, not by volume. Some settling may occur during handling and shipping
Made in the USA with US and foreign components
Side Effects
Vi-Jon
One Swan Drive
Smyrna, TN 37167
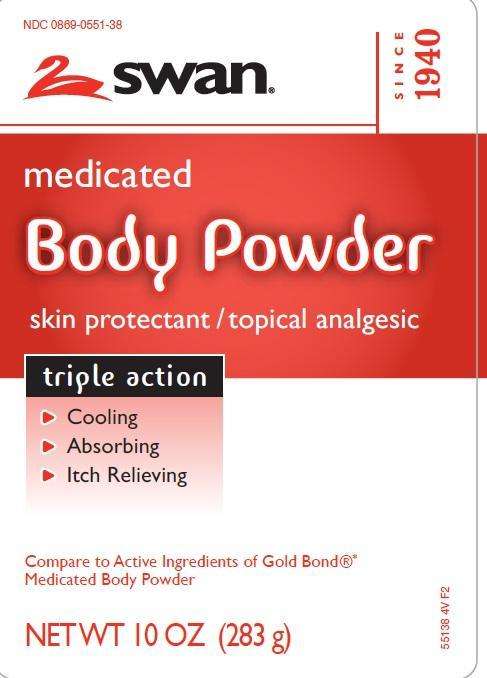
Principal Display Panel
NDC 0869-0551-38
SWAN
medicated
Body Powder
skin protectant/topical analgesic
triple action
- Cooling
- Absorbing
- Itch Relieving
Compare to Active Ingredients of Gold Bond Medicated Body Powder
NET WT 10 OZ (283 G)
Medicatedmenthol, zinc oxide POWDER
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||