Metoprolol Tartrate
FULL PRESCRIBING INFORMATION: CONTENTS*
- BOXED WARNING
- METOPROLOL TARTRATE DESCRIPTION
- CLINICAL PHARMACOLOGY
- PHARMACOKINETICS
- INDICATIONS & USAGE
- METOPROLOL TARTRATE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- INFORMATION FOR PATIENTS
- DRUG INTERACTIONS
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- PREGNANCY
- NURSING MOTHERS
- PEDIATRIC USE
- GERIATRIC USE
- METOPROLOL TARTRATE ADVERSE REACTIONS
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- STORAGE AND HANDLING
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
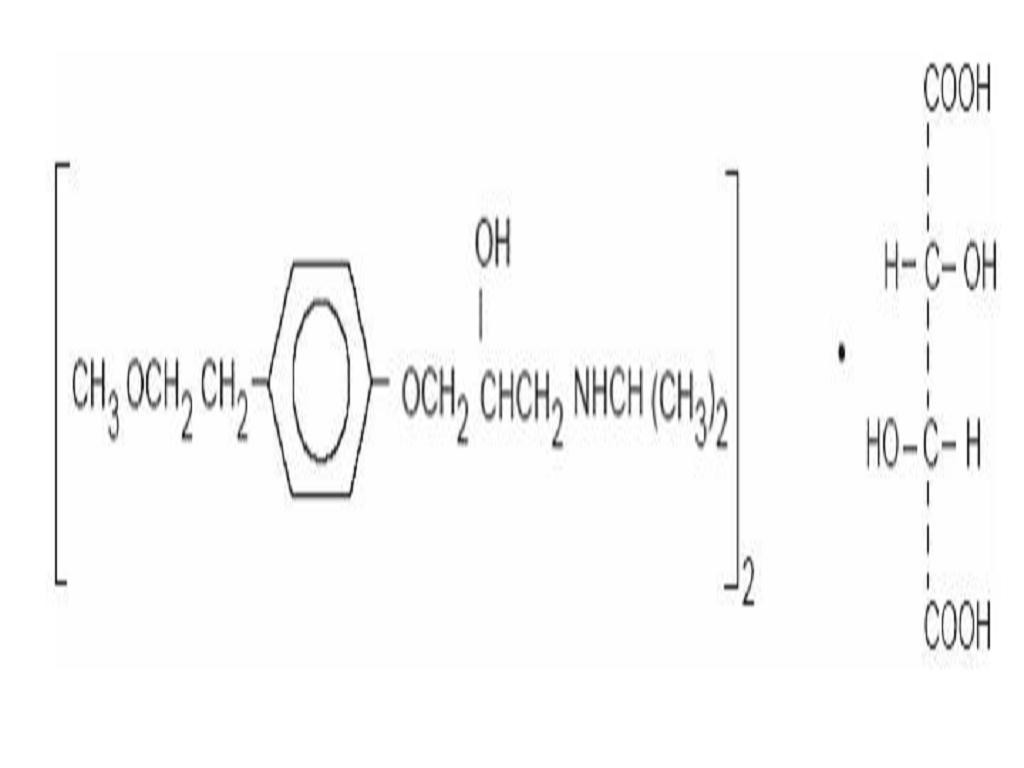
METOPROLOL TARTRATE DESCRIPTION
CLINICAL PHARMACOLOGY
PHARMACOKINETICS
INDICATIONS & USAGE
HypertensionAngina Pectoris
Myocardial Infarction
DOSAGE AND ADMINISTRATIONCONTRAINDICATIONSWARNINGSDOSAGE AND ADMINISTRATION
METOPROLOL TARTRATE CONTRAINDICATIONS
Hypertension and AnginaWARNINGS
Myocardial Infarction
WARNINGS
WARNINGS
Hypertension and AnginaCardiac Failure
In Patients Without a History of Cardiac Failure
Bronchospastic Diseases
PATIENTS WITH BRONCHOSPASTIC DISEASES SHOULD, IN GENERAL, NOT RECEIVE BETA BLOCKERS, including Metoprolol tartrate. Because of its relative beta1 selectivity, however, metoprolol may be used with caution in patients with bronchospastic disease who do not respond to, or cannot tolerate, other antihypertensive treatment. Since beta1 selectivity is not absolute, a beta2-stimulating agent should be administered concomitantly, and the lowest possible dose of metoprolol tartrate should be used. In these circumstances it would be prudent initially to administer metoprolol in smaller doses three times daily, instead of larger doses two times daily, to avoid the higher plasma levels associated with the longer dosing interval (seeDOSAGE AND ADMINISTRATION).
Major Surgery
Diabetes and Hypoglycemia
Pheochromocytoma
Thyrotoxicosis
Myocardial Infarction
Cardiac Failure
Bradycardia
AV Block
Hypotension
above
Bronchospastic Diseases
PATIENTS WITH BRONCHOSPASTIC DISEASES SHOULD, IN GENERAL, NOT RECEIVE BETA BLOCKERS, including Metoprolol tartrate. Because of its relative beta1 selectivity, metoprolol may be used with extreme caution in patients with bronchospastic disease. Because it is unknown to what extent beta2-stimulating agents may exacerbate myocardial ischemia and the extent of infarction, these agents should not be used prophylactically. If bronchospasm not related to congestive heart failure occurs, metoprolol should be discontinued. A theophylline derivative or a beta2 agonist may be administered cautiously, depending on the clinical condition of the patient. Both theophylline derivatives and beta2 agonists may produce serious cardiac arrhythmias.
PRECAUTIONS
GeneralINFORMATION FOR PATIENTS
DRUG INTERACTIONS
Risk of Anaphylactic Reaction
General Anesthetics
WARNINGS, Major Surgery
CYP2D6 Inhibitors
Pharmacokinetics
Clonidine
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
PREGNANCY
NURSING MOTHERS
PEDIATRIC USE
GERIATRIC USE
METOPROLOL TARTRATE ADVERSE REACTIONS
Hypertension and AnginaCentral Nervous System:
Cardiovascular:CONTRAINDICATIONSWARNINGSPRECAUTIONS
Respiratory:WARNINGS
Gastrointestinal:
Hypersensitive Reactions:
Miscellaneous:
Myocardial Infarction
Central Nervous System:
Cardiovascular:
metoprololPlaceboRespiratory:
Gastrointestinal:
Dermatologic:
Miscellaneous:
Potential Adverse Reactions
Central Nervous System:
Cardiovascular:CONTRAINDICATIONS
Hematologic:
Hypersensitive Reactions:
Postmarketing Experience
OVERDOSAGE
Acute Toxicity
Signs and Symptoms
Treatment
WARNINGS, Myocardial Infarction
Elimination of the Drug:
Bradycardia:
Hypotension:
Bronchospasm:
Cardiac Failure:
DOSAGE & ADMINISTRATION
HypertensionAngina Pectoris
WARNINGS
Myocardial Infarction
Early Treatment
Late Treatment
WARNINGS
Late Treatment
HOW SUPPLIED
STORAGE AND HANDLING
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION

Metoprolol TartrateMetoprolol Tartrate TABLET
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!