Mirtazapine
FULL PRESCRIBING INFORMATION: CONTENTS*
- Suicidality and Antidepressant Drugs
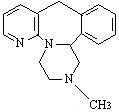
- MIRTAZAPINE DESCRIPTION
- CLINICAL PHARMACOLOGY
- MIRTAZAPINE INDICATIONS AND USAGE
- MIRTAZAPINE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- MIRTAZAPINE ADVERSE REACTIONS
- Associated with Discontinuation of Treatment
- Commonly Observed Adverse Events in U.S. Controlled Clinical Trials
- Adverse Events Occurring at an Incidence of 1% or More Among Mirtazapine-Treated Patients
- ECG Changes
- Other Adverse Events Observed During the Premarketing Evaluation of Mirtazapine
- Other Adverse Events Observed During Postmarketing Evaluation of Mirtazapine
- DRUG ABUSE AND DEPENDENCE
- OVERDOSAGE
- MIRTAZAPINE DOSAGE AND ADMINISTRATION
- Initial Treatment
- Elderly and Patients with Renal or Hepatic Impairment
- Maintenance/Extended Treatment
- Switching a Patient To or From a Monoamine Oxidase Inhibitor (MAOI) Intended to Treat Psychiatric Disorders
- Use of Mirtazapine Tablets With Other MAOIs, Such as Linezolid or Methylene Blue
- Discontinuation of Mirtazapine Tablets Treatment
- HOW SUPPLIED
- Medication Guide
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 7.5 mg (100 Tablet Bottle)
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 15 mg (100 Tablet Bottle)
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 15 mg Unit-of-Use Pack (30 Tablets)
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 30 mg (100 Tablet Bottle)
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 30 mg Unit-of-Use Pack (30 Tablets)
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 45 mg (100 Tablet Bottle)
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 45 mg Unit-of-Use Pack (30 Tablets)
FULL PRESCRIBING INFORMATION
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of mirtazapine tablets or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Mirtazapine is not approved for use in pediatric patients. (See WARNINGS: Clinical Worsening and Suicide Risk, PRECAUTIONS: Information for Patients, and PRECAUTIONS: Pediatric Use).
MIRTAZAPINE DESCRIPTION
17193
CLINICAL PHARMACOLOGY
Pharmacodynamics
2
2 31A1B
1
1
Pharmacokinetics
In vitro
Special Populations
Geriatric
PRECAUTIONSDOSAGE AND ADMINISTRATION
Pediatrics
PRECAUTIONS
Gender
Pharmacokinetics
Race
Renal Insufficiency
22PRECAUTIONSDOSAGE AND ADMINISTRATION
Hepatic Insufficiency
PRECAUTIONSDOSAGE AND ADMINISTRATION
Clinical Trials Showing Effectiveness
MIRTAZAPINE INDICATIONS AND USAGE
CLINICAL PHARMACOLOGY
CLINICAL PHARMACOLOGY
MIRTAZAPINE CONTRAINDICATIONS
WARNINGSDOSAGE AND ADMINISTRATION
WARNINGSDOSAGE AND ADMINISTRATION
WARNINGS
Clinical Worsening and Suicide Risk
| Age Range |
Drug-Placebo Difference in Number of Cases of Suicidality per 1000 Patients Treated |
| Increases Compared to Placebo |
|
| <18 |
14 additional cases |
| 18-24 |
5 additional cases |
| Decreases Compared to Placebo |
|
| 25-64 |
1 fewer case |
|
>65 |
6 fewer cases |
All patients being treated with antidepressants for any indication should be monitored appropriately and observed closely for clinical worsening, suicidality, and unusual changes in behavior, especially during the initial few months of a course of drug therapy, or at times of dose changes, either increases or decreases.
Families and caregivers of patients being treated with antidepressants for major depressive disorder or other indications, both psychiatric and nonpsychiatric, should be alerted about the need to monitor patients for the emergence of agitation, irritability, unusual changes in behavior, and the other symptoms described above, as well as the emergence of suicidality, and to report such symptoms immediately to health care providers. Such monitoring should include daily observation by families and caregivers.
Screening Patients for Bipolar Disorder
Agranulocytosis
In premarketing clinical trials, 2 (1 with Sjögren’s Syndrome) out of 2796 patients treated with mirtazapine tablets developed agranulocytosis [absolute neutrophil count (ANC) < 500/mm3 with associated signs and symptoms, e.g., fever, infection, etc.] and a third patient developed severe neutropenia (ANC < 500/mm3 without any associated symptoms). For these 3 patients, onset of severe neutropenia was detected on days 61, 9, and 14 of treatment, respectively. All 3 patients recovered after mirtazapine was stopped. These 3 cases yield a crude incidence of severe neutropenia (with or without associated infection) of approximately 1.1 per thousand patients exposed, with a very wide 95% confidence interval, i.e., 2.2 cases per 10,000 to 3.1 cases per 1000. If a patient develops a sore throat, fever, stomatitis, or other signs of infection, along with a low WBC count, treatment with mirtazapine should be discontinued and the patient should be closely monitored.
Serotonin Syndrome
The development of a potentially life-threatening serotonin syndrome has been reported with SNRIs and SSRIs, including mirtazapine, alone but particularly with concomitant use of other serotonergic drugs (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, and St. John's wort), and with drugs that impair metabolism of serotonin (in particular, MAOIs, both those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue).
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Patients should be monitored for the emergence of serotonin syndrome.
The concomitant use of mirtazapine with MAOIs intended to treat psychiatric disorders is contraindicated. Mirtazapine should also not be started in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue. All reports with methylene blue that provided information on the route of administration involved intravenous administration in the dose range of 1 mg/kg to 8 mg/kg. No reports involved the administration of methylene blue by other routes (such as oral tablets or local tissue injection) or at lower doses. There may be circumstances when it is necessary to initiate treatment with an MAOI such as linezolid or intravenous methylene blue in a patient taking mirtazapine. Mirtazapine should be discontinued before initiating treatment with the MAOI (see CONTRAINDICATIONS and DOSAGE AND ADMINISTRATION).
If concomitant use of mirtazapine with other serotonergic drugs, including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, buspirone, tryptophan, and St. John's wort, is clinically warranted, be aware of a potential increased risk for serotonin syndrome, particularly during treatment initiation and dose increases.
Treatment with mirtazapine and any concomitant serotonergic agents should be discontinued immediately if the above events occur and supportive symptomatic treatment should be initiated.
PRECAUTIONS
General
Discontinuation Symptoms
Akathisia/Psychomotor Restlessness
Hyponatremia
Somnolence
PRECAUTIONS: Information for Patients
Dizziness
Increased Appetite/Weight Gain
PRECAUTIONS: Pediatric Use
Cholesterol/Triglycerides
Transaminase Elevations
CLINICAL PHARMACOLOGYDOSAGE AND ADMINISTRATION
Activation of Mania/Hypomania
Seizure
Use in Patients with Concomitant Illness
22CLINICAL PHARMACOLOGYDOSAGE AND ADMINISTRATION
Information for Patients
Clinical Worsening and Suicide Risk
Agranulocytosis
Interference with Cognitive and Motor Performance
Completing Course of Therapy
Concomitant Medication
Alcohol
Pregnancy
Nursing
Laboratory Tests
Drug Interactions
CLINICAL PHARMACOLOGY
Monoamine Oxidase Inhibitors
CONTRAINDICATIONSWARNINGSDOSAGE AND ADMINISTRATION
Serotonergic Drugs
CONTRAINDICATIONSWARNINGS
Drugs Affecting Hepatic Metabolism
Drugs that are Metabolized by and/or Inhibit Cytochrome P450 Enzymes
CYP Enzyme Inducers
Phenytoin
Carbamazepine
CYP Enzyme Inhibitors
Cimetidine
Ketoconazole
Paroxetine
in vivo
Other Drug-Drug Interactions
Amitriptyline
Warfarin
Lithium
Risperidone
in vivo
Alcohol
Diazepam
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
2
Mutagenesis
in vitro in vitro in vivo
Impairment of Fertility
2
Pregnancy
Teratogenic Effects
Pregnancy Category C
22
Nursing Mothers
Pediatric Use
BOXED WARNINGWARNINGS: Clinical Worsening and Suicide Risk
PRECAUTIONS: Increased Appetite/Weight Gain
Geriatric Use
CLINICAL PHARMACOLOGYDOSAGE AND ADMINISTRATION
MIRTAZAPINE ADVERSE REACTIONS
Associated with Discontinuation of Treatment
|
Adverse Event
|
Percentage of Patients Discontinuing With Adverse Event
|
|
|
Mirtazapine
(n=453) |
Placebo
(n=361) |
|
| Somnolence |
10.4% |
2.2% |
| Nausea |
1.5% |
0% |
Commonly Observed Adverse Events in U.S. Controlled Clinical Trials
|
Adverse Event
|
Percentage of Patients Reporting Adverse Event
|
|
|
Mirtazapine
(n=453) |
Placebo
(n=361) |
|
| Somnolence |
54% |
18% |
| Increased Appetite |
17% |
2% |
| Weight Gain |
12% |
2% |
| Dizziness |
7% |
3% |
Adverse Events Occurring at an Incidence of 1% or More Among Mirtazapine-Treated Patients
| 1Events reported by at least 1% of patients treated with mirtazapine are included, except the following events, which had an incidence on placebo grater than or equal to mirtazapine: headache, infection, pain, chest pain, palpitation, tachycardia, postural hypotension, nausea, dyspepsia, diarrhea, flatulence, insomnia, nervousness, libido decreased, hypertonia, pharyngitis, rhinitis, sweating, amblyopia, tinnitus, taste perversion. | |||
|
Body System
Adverse Clinical Experience |
Mirtazapine
(n=453) |
Placebo
(n=361) |
|
|
Body as a Whole
|
|||
| Asthenia |
8% |
5% |
|
| Flu Syndrome |
5% |
3% |
|
| Back Pain |
2% |
1% |
|
|
Digestive System
|
|||
| Dry Mouth |
25% |
15% |
|
| Increased Appetite |
17% |
2% |
|
| Constipation |
13% |
7% |
|
|
Metabolic and Nutritional Disorders
|
|||
| Weight Gain |
12% |
2% |
|
| Peripheral Edema |
2% |
1% |
|
| Edema |
1% |
0% |
|
| Musculoskeletal System | |||
| Myalgia |
2% |
1% |
|
| Nervous System | |||
| Somnolence |
54% |
18% |
|
| Dizziness |
7% |
3% |
|
| Abnormal Dreams |
4% |
1% |
|
| Thinking Abnormal |
3% |
1% |
|
| Tremor |
2% |
1% |
|
| Confusion |
2% |
0% |
|
| Respiratory System | |||
| Dyspnea |
1% |
0% |
|
| Urogenital System | |||
| Urinary Frequency |
2% |
1% |
|
ECG Changes
Other Adverse Events Observed During the Premarketing Evaluation of Mirtazapine
WARNINGSPRECAUTIONS
frequent:infrequent: rare:
frequent: infrequent:rare:
frequent: infrequent: rare:
rare:
rare:
frequent: infrequent: rare:
frequent: infrequent: rare:
frequent: infrequent: rare:
frequent: infrequent: rare:
frequent: infrequent: rare:
infrequent: rare:
frequent:infrequent: rare:
Other Adverse Events Observed During Postmarketing Evaluation of Mirtazapine
DRUG ABUSE AND DEPENDENCE
Controlled Substance Class
Physical and Psychological Dependence
OVERDOSAGE
Human Experience
Overdose Management
Physicians’ Desk Reference (
MIRTAZAPINE DOSAGE AND ADMINISTRATION
Initial Treatment
Elderly and Patients with Renal or Hepatic Impairment
PRECAUTIONSCLINICAL PHARMACOLOGY
Maintenance/Extended Treatment
CLINICAL PHARMACOLOGY
Switching a Patient To or From a Monoamine Oxidase Inhibitor (MAOI) Intended to Treat Psychiatric Disorders
At least 14 days should elapse between discontinuation of an MAOI intended to treat psychiatric disorders and initiation of therapy with mirtazapine tablets. Conversely, at least 14 days should be allowed after stopping mirtazapine tablets before starting an MAOI intended to treat psychiatric disorders (see CONTRAINDICATIONS).
Use of Mirtazapine Tablets With Other MAOIs, Such as Linezolid or Methylene Blue
Do not start mirtazapine tablets in a patient who is being treated with linezolid or intravenous methylene blue because there is an increased risk of serotonin syndrome. In a patient who requires more urgent treatment of a psychiatric condition, other interventions, including hospitalization, should be considered (see CONTRAINDICATIONS).
In some cases, a patient already receiving therapy with mirtazapine tablets may require urgent treatment with linezolid or intravenous methylene blue. If acceptable alternatives to linezolid or intravenous methylene blue treatment are not available and the potential benefits of linezolid or intravenous methylene blue treatment are judged to outweigh the risks of serotonin syndrome in a particular patient, mirtazapine tablets should be stopped promptly, and linezolid or intravenous methylene blue can be administered. The patient should be monitored for symptoms of serotonin syndrome for 2 weeks or until 24 hours after the last dose of linezolid or intravenous methylene blue, whichever comes first. Therapy with mirtazapine tablets may be resumed 24 hours after the last dose of linezolid or intravenous methylene blue (see WARNINGS).
The risk of administering methylene blue by non-intravenous routes (such as oral tablets or by local injection) or in intravenous doses much lower than 1 mg/kg with mirtazapine tablets is unclear. The clinician should, nevertheless, be aware of the possibility of emergent symptoms of serotonin syndrome with such use (see WARNINGS).
Discontinuation of Mirtazapine Tablets Treatment
PRECAUTIONSADVERSE REACTIONS
HOW SUPPLIED
7.5 mg Tablets
15 mg Tablets
30 mg Tablets
45 mg Tablets
Storage
Store at
Aurobindo Pharma USA, Inc.
Aurobindo Pharma Limited
Medication Guide
Mirtazapine Tablets, USP
What is the most important information I should know about mirtazapine tablets?
1. Suicidal thoughts or actions:
- Mirtazapine tablets and other antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers, or young adults within the first few months of treatment or when the dose is changed.
- Depression or other serious mental illnesses are the most important causes of suicidal thoughts or actions.
- Watch for these changes and call your healthcare provider right away if you notice:
- New or sudden changes in mood, behavior, actions, thoughts, or feelings, especially if severe.
- Pay particular attention to such changes when mirtazapine tablets are started or when the dose is changed.
Call your healthcare provider right away if you have any of the following symptoms, or call 911 if an emergency, especially if they are new, worse, or worry you:
- attempts to commit suicide
- acting on dangerous impulses
- acting aggressive or violent
- thoughts about suicide or dying
- new or worse depression
- new or worse anxiety or panic attacks
- feeling agitated, restless, angry or irritable
- trouble sleeping
- an increase in activity or talking more than what is normal for you
- other unusual changes in behavior or mood
Call your healthcare provider right away if you have any of the following symptoms, or call 911 if an emergency. Mirtazapine tablets may be associated with these serious side effects:
2. Manic episodes:
- greatly increased energy
- severe trouble sleeping
- racing thoughts
- reckless behavior
- unusually grand ideas
- excessive happiness or irritability
- talking more or faster than usual
3. Decreased White Blood Cells
4. Serotonin Syndrome. This condition can be life-threatening and may include:
- agitation, hallucinations, coma or other changes in mental status
- coordination problems or muscle twitching (overactive reflexes)
- racing heartbeat, high or low blood pressure
- sweating or fever
- nausea, vomiting, or diarrhea
- muscle rigidity
5. Seizures
6. Low salt (sodium) levels in the blood.
- headache
- weakness or feeling unsteady
- confusion, problems concentrating or thinking or memory problems
7. Sleepiness. mirtazapine tablets
8. Severe skin reactions:
- severe rash with skin swelling (including on the palms of the hands and soles of the feet)
- painful reddening of the skin and/or blisters/ulcers on the body or in the mouth
9. Severe allergic reactions: trouble breathing, swelling of the face, tongue, eyes or mouth
- rash, itchy welts (hives) or blisters, alone or with fever or joint pain
10. Increases in appetite or weight.
11. Increased cholesterol and triglyceride levels in your blood
Do not stop mirtazapine tablets without first talking to your healthcare provider
- dizziness
- abnormal dreams
- agitation
- anxiety
- fatigue
- confusion
- headache
- shaking
- tingling sensation
- nausea, vomiting
- sweating
What are mirtazapine tablets?
Who should not take mirtazapine tablets?
Do not take mirtazapine tablets:
- if you are allergic to mirtazapine or any of the ingredients in mirtazapine tablets. See the end of this Medication Guide for a complete list of ingredients in mirtazapine tablets.
- if you take a monoamine oxidase inhibitor (MAOI). Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including the antibiotic linezolid.
- Do not take an MAOI within 2 weeks of stopping mirtazapine tablets unless directed to do so by your physician.
- Do not start mirtazapine tablets if you stopped taking an MAOI in the last 2 weeks unless directed to do so by your physician.
People who take mirtazapine tablets close in time to an MAOI may have serious or even life-threatening side effects. Get medical help right away if you have any of these symptoms:
- high fever
- uncontrolled muscle spasms
- stiff muscles
- rapid changes in heart rate or blood pressure
- confusion
- loss of consciousness (pass out)
What should I tell my healthcare provider before taking mirtazapine tablets? Ask if you are not sure.
- Are taking certain drugs such as:
- Triptans used to treat migraine headache
- Medicines used to treat mood, anxiety, psychotic or thought disorders, including tricyclics, lithium, SSRIs, SNRIs, or antipsychotics
- Tramadol used to treat pain
- Over-the-counter supplements such as tryptophan or St. John’s wort
- Phenytoin, carbamazepine, or rifampicin (these drugs can decrease your blood level of mirtazapine)
- Cimetidine or ketoconazole (these drugs can increase your blood level of mirtazapine)
- Have or had:
- liver problems
- kidney problems
- heart problems
- seizures or convulsions
- bipolar disorder or mania
- a tendency to get dizzy or faint
- are pregnant or plan to become pregnant. It is not known if mirtazapine tablets will harm your unborn baby. Talk to your healthcare provider about the benefits and risks of treating depression during pregnancy
- are breastfeeding or plan to breastfeed. Some mirtazapine may pass into your breast milk. Talk to your healthcare provider about the best way to feed your baby while taking mirtazapine tablets
Tell your healthcare provider about all the medicines that you take,
| If you take mirtazapine tablets, you should not take any other medicines that contain mirtazapine including mirtazapine orally disintegrating tablets. |
How should I take mirtazapine tablets?
- Take mirtazapine tablets exactly as prescribed. Your healthcare provider may need to change the dose of mirtazapine tablets until it is the right dose for you.
- Take mirtazapine tablets at the same time each day, preferably in the evening at bedtime.
- Swallow mirtazapine tablets as directed.
- It is common for antidepressant medicines such as mirtazapine tablets to take up to a few weeks before you start to feel better. Do not stop taking mirtazapine tablets if you do not feel results right away.
- Do not stop taking or change the dose of mirtazapine tablets without first talking to your doctor, even if you feel better.
- Mirtazapine tablets may be taken with or without food.
- If you miss a dose of mirtazapine tablets, take the missed dose as soon as you remember. If it is almost time for the next dose, skip the missed dose and take your next dose at the regular time. Do not take two doses of mirtazapine tablets at the same time.
- If you take too much mirtazapine, call your healthcare provider or poison control center right away, or get emergency treatment.
What should I avoid while taking mirtazapine tablets?
- Mirtazapine tablets can cause sleepiness or may affect your ability to make decisions, think clearly, or react quickly. You should not drive, operate heavy machinery, or do other dangerous activities until you know how mirtazapine tablets affect you.
- Avoid drinking alcohol or taking diazepam (a medicine used for anxiety, insomnia and seizures, for example)or similar medicines while taking mirtazapine tablets. If you are uncertain about whether certain medication can be taken with mirtazapine tablets, please discuss with your doctor.
What are the possible side effects of mirtazapine tablets?
“What is the most important information I should know about mirtazapine tablets?"
- sleepiness
- increased appetite, weight gain
- dry mouth
- constipation
- dizziness
- abnormal dreams
CALL YOUR DOCTOR FOR MEDICAL ADVICE ABOUT SIDE EFFECTS. YOU MAY REPORT SIDE EFFECTS TO THE FDA AT 1-800-FDA-1088.
How should I store mirtazapine tablets?
- Store at 25° C (77° F); excursions permitted to 15° to 30° C (59° to 86° F) [see USP Controlled Room Temperature].
- Protect from light and moisture.
Keep mirtazapine tablets and all medicines out of the reach of children.
General information about mirtazapine tablets
What are the ingredients in mirtazapine tablets?
Aurobindo Pharma USA, Inc.
Aurobindo Pharma Limited
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 7.5 mg (100 Tablet Bottle)
NDC 65862-001-01
Mirtazapine Tablets, USP 7.5 mg
PHARMACIST: PLEASE DISPENSE WITH
MEDICATION GUIDE PROVIDED SEPARATELY
Rx only 100 Tablets
AUROBINDO

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 15 mg (100 Tablet Bottle)
NDC 65862-031-01
Mirtazapine Tablets, USP 15 mg
PHARMACIST: PLEASE DISPENSE WITH
MEDICATION GUIDE PROVIDED SEPARATELY
Rx only 100 Tablets
AUROBINDO

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 15 mg Unit-of-Use Pack (30 Tablets)
NDC 65862-031-32
Mirtazapine Tablets, USP
15 mg
PHARMACIST: PLEASE DISPENSE WITH
MEDICATION GUIDE PROVIDED SEPARATELY
Rx only 30 Tablets
PLACE PHARMACY LABEL HERE
DO NOT WRAP LABEL AROUND EDGES
AUROBINDO
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 30 mg (100 Tablet Bottle)
NDC 65862-003-01
Mirtazapine Tablets, USP 30 mg
PHARMACIST: PLEASE DISPENSE WITH
MEDICATION GUIDE PROVIDED SEPARATELY
Rx only 100 Tablets
AUROBINDO

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 30 mg Unit-of-Use Pack (30 Tablets)
NDC 65862-003-32
Mirtazapine Tablets, USP 30 mg
PHARMACIST: PLEASE DISPENSE WITH
MEDICATION GUIDE PROVIDED SEPARATELY
Rx only 30 Tablets
PLACE PHARMACY LABEL HERE
DO NOT WRAP LABEL AROUND EDGES
AUROBINDO
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 45 mg (100 Tablet Bottle)
NDC 65862-032-01
Mirtazapine Tablets, USP 45 mg
PHARMACIST: PLEASE DISPENSE WITH
MEDICATION GUIDE PROVIDED SEPARATELY
Rx only 100 Tablets
AUROBINDO

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 45 mg Unit-of-Use Pack (30 Tablets)
NDC 65862-032-32
Mirtazapine Tablets, USP 4 5 mg
PHARMACIST: PLEASE DISPENSE WITH
MEDICATION GUIDE PROVIDED SEPARATELY
Rx only 30 Tablets
PLACE PHARMACY LABEL HERE
DO NOT WRAP LABEL AROUND EDGES
AUROBINDO
MirtazapineMirtazapine TABLET, FILM COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
MirtazapineMirtazapine TABLET, FILM COATED
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
MirtazapineMirtazapine TABLET, FILM COATED
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
MirtazapineMirtazapine TABLET, FILM COATED
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||