Non-Habit Forming Stool Softener
Rebel Distributors Corp
PSS World Medical, Inc.
Non-Habit Forming Stool Softener Laxative
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active Ingredient (in each softgel capsule)
- Non-Habit Forming Stool Softener Uses
- Warnings - Do not use
- Directions
- Non-Habit Forming Stool Softener Other information
- Inactive Ingredients
- Questions or comments?
- Principal Display Panel
FULL PRESCRIBING INFORMATION
Active Ingredient (in each softgel capsule)
Docusate Sodium 100mg
Purpose
Stool softener
Non-Habit Forming Stool Softener Uses
- Relieves occasional constipation.
- Generally produces bowel movement within 12 to72 hours.
Warnings - Do not use
- for longer than one week
- if you are taking mineral oil
- when abdominal pain, nausea, or vomiting are present
Ask a doctor before use if
you have a sudden change in bowel habits that lasts over two weeks
Ask a doctor or pharmacist before use if you are taking any other drug. Take this product two or more hours before or after other drugs. Laxatives may affect how other drugs work.
Stop use and ask a doctor if
- you experience rectal bleeding
- you fail to have a bowel movement after using this product
These may indicate a serious condition.
If pregnant or breastfeeding
ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- adults and children over 12 years and over:1 capsule once daily or in divided doses
- children 6 to under 12 years: 1 capsule once daily
- children under 6 years: ask a doctor
Non-Habit Forming Stool Softener Other information
- store at controlled room temperature 15 - 30 degrees C (59-86 degrees F)
Each capsule contains sodium 6mg
Inactive Ingredients
edible white ink, FDandC Red No. 40, FDandC Yellow No. 6, gelatin, glycerin, polyethylene glycol, propylene glycol, purified water, sorbitol special.
Questions or comments?
Call 1-800-645-2158, 9am - 5am ET Monday - Friday
Principal Display Panel
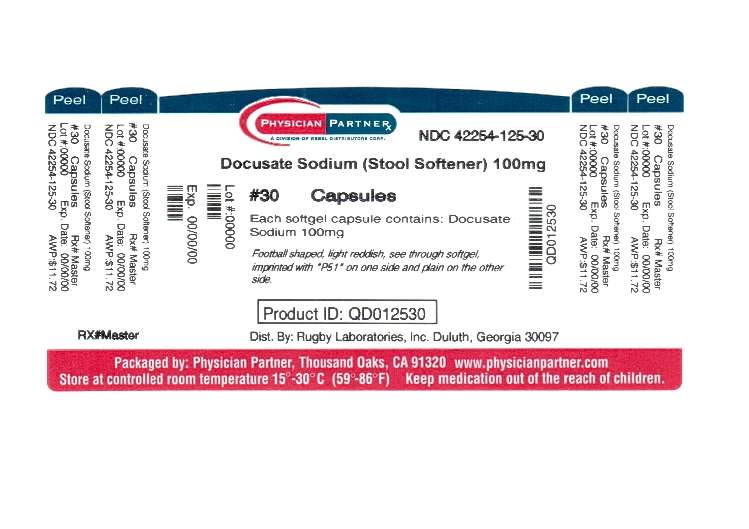
Non-Habit Forming Stool SoftenerDOCUSATE SODIUM CAPSULE, LIQUID FILLED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||