Pentazocine and naloxone
Pentazocine and Naloxone Tablets
FULL PRESCRIBING INFORMATION
Pentazocine and Naloxone Tablets USP C-IV
Revised: October 2013
Analgesic for Oral Use Only
Pentazocine and naloxone tablets are intended for oral use only. Severe, potentially lethal, reactions may result from misuse of pentazocine and naloxone tablets by injection either alone or in combination with other substances. (See DRUG ABUSE AND DEPENDENCE section.)
Pentazocine and naloxone tablets USP contain pentazocine hydrochloride, USP, equivalent to 50 mg base and is a member of the benzazocine series (also known as the benzomorphan series), and naloxone hydrochloride, USP, equivalent to 0.5 mg base.
Pentazocine and naloxone tablets USP are an analgesic for oral administration.
Chemically, pentazocine hydrochloride, USP is (2R*,6R*,11R*)-1,2,3,4,5,6-Hexahydro-6,11-dimethyl-3-(3-methyl-2-butenyl)-2,6-methano-3-benzazocin-8-ol hydrochloride, a white, crystalline substance soluble in acidic aqueous solutions, and has the following structural formula:

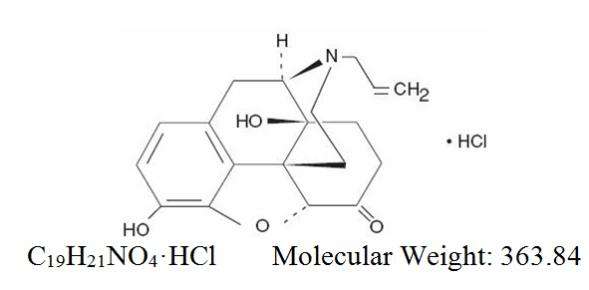
Chemically, naloxone hydrochloride, USP is Morphinan-6-one,4,5-epoxy-3,14-dihydroxy-17-(2-propenyl)-, hydrochloride, (5α)-. It is a slightly off-white powder, and is soluble in water and dilute acids, and has the following structural formula:
Inactive Ingredients: colloidal silicon dioxide, dibasic calcium phosphate, D&C Yellow No. 10 Al-lake, FD&C Blue No. 1 Al-lake, FD&C Yellow No. 6 Al-lake, magnesium stearate, microcrystalline cellulose, pregelatinized starch, and sodium lauryl sulfate.
Pentazocine is a Schedule IV opioid analgesic which when administered orally in a 50 mg dose appears equivalent in analgesic effect to 60 mg of codeine.
Pentazocine weakly antagonizes the analgesic effects of morphine and meperidine; in addition, it produces incomplete reversal of cardiovascular, respiratory, and behavioral depression induced by morphine and meperidine. Pentazocine has about 1/50 the antagonistic activity of nalorphine. It also has sedative activity.
Onset of significant analgesia usually occurs between 15 and 30 minutes after oral administration, and duration of action is usually three hours or longer.
Pentazocine is well absorbed from the gastrointestinal tract. Concentrations in plasma coincide closely with the onset, duration, and intensity of analgesia. The time to mean peak concentration in 24 normal volunteers was 1.7 hours (range 0.5 to 4 hours) after oral administration and the mean plasma elimination half-life was 3.6 hours (range 1.5 to 10 hours).
Pentazocine is metabolized in the liver and excreted primarily in the urine. The products of the oxidation of the terminal methyl groups and glucuronide conjugates are excreted by the kidney. Elimination of approximately 60% of the total dose occurs within 24 hours. Pentazocine passes into the fetal circulation.
Naloxone when administered orally at 0.5 mg has no pharmacologic activity. Naloxone hydrochloride administered parenterally at the same dose is an antagonist to pentazocine and a pure antagonist to narcotic analgesics.
Pentazocine and naloxone tablets are a potent analgesic when administered orally. However, the presence of naloxone in pentazocine and naloxone tablets is intended to prevent the effect of pentazocine if the product is misused by injection.
Studies in animals indicate that the presence of naloxone does not affect pentazocine analgesia when the combination is given orally. If the combination is given by injection the action of pentazocine is neutralized.
Pentazocine and naloxone tablets USP are indicated for the relief of moderate to severe pain.
Pentazocine and naloxone tablets USP is indicated for oral use only.
Pentazocine and naloxone tablets are contraindicated in patients who are hypersensitive to either pentazocine or naloxone.
Pentazocine can cause a physical and psychological dependence. (See DRUG ABUSE AND DEPENDENCE .)
In the presence of head injury, intracranial lesions or a preexisting increase in intracranial pressure, the possible respiratory depressant effects of pentazocine and its potential to elevate cerebrospinal fluid pressure (resulting from vasodilation following CO2 retention) may be markedly increased. Furthermore, pentazocine can produce effects on pupillary response and consciousness, which may obscure neurologic signs of further increases in intracranial pressure in patients with head injuries. In such patients, pentazocine must be used with extreme caution and only if its use is deemed essential.
Pentazocine may be expected to have additive effects when used in conjunction with alcohol, other opioids, or illicit drugs that cause central nervous system depression because respiratory depression, hypotension, profound sedation, coma or death may result.
Pentazocine is a mild narcotic antagonist. Some patients previously given narcotics, including methadone for the daily treatment of narcotic dependence, have experienced withdrawal symptoms after receiving pentazocine.
Respiratory depression occurs more frequently in elderly or debilitated patients and in those suffering from conditions accompanied by hypoxia, hypercapnia, or upper airway obstruction, in whom even moderate therapeutic doses may significantly decrease pulmonary ventilation. Use pentazocine and naloxone tablets with extreme caution in patients with chronic obstructive pulmonary disease or cor pulmonale and in patients having a substantially decreased respiratory reserve (e.g., severe kyphoscoliosis), hypoxia, hypercapnia, or pre-existing respiratory depression. Alternative non-opioid analgesics should be considered, and pentazocine and naloxone tablets should be employed only under careful medical supervision at the lowest effective dose in such patients.
Patients receiving therapeutic doses of pentazocine and naloxone tablets have experienced hallucinations (usually visual), disorientation, and confusion which have cleared spontaneously within a period of hours. The mechanism of this reaction is not known. Such patients should be very closely observed and vital signs checked. If the drug is reinstituted, it should be done with caution since these acute CNS manifestations may recur.
Pentazocine and naloxone tablets are a Schedule IV controlled substance.
Abuse and addiction are separate and distinct from physical dependence and tolerance. Abuse is characterized by misuse of a drug for non-medical purposes, often in combination with other psychoactive substances. Addiction is a disease of repeated drug abuse. Addiction is a primary, chronic, neurobiologic disease, with genetic, psychosocial, and environmental factors influencing its development and manifestations. Addiction is characterized by behaviors that include one or more of the following: impaired control over drug use, compulsive use, continued use despite harm, and craving. Drug addiction is a treatable disease, utilizing a multidisciplinary approach, but relapse is common. Physical dependence is a state of adaptation that is manifested by a specific withdrawal syndrome that can be produced by abrupt cessation, rapid dose reduction, decreasing blood level of the drug, and/or administration of an antagonist. Tolerance is a state of adaptation in which exposure to a drug induces changes that result in a diminution of one or more of the drug’s effects over time. Tolerance may occur to both the desired and undesired effects of drugs, and may develop at different rates for different effects.
Physicians should be aware that addiction may not be accompanied by concurrent tolerance and symptoms of physical dependence in all addicts. In addition, abuse of opioids can occur in the absence of addiction and is characterized by misuse of the drug for non-medical purposes, and often in combination with other psychoactive substances.
There have been some reports of dependence and of withdrawal symptoms with pentazocine and naloxone tablets. Patients with a history of drug dependence should be under close supervision while receiving pentazocine and naloxone tablets. There have been rare reports of possible abstinence syndromes in newborns after prolonged use of pentazocine and naloxone tablets during pregnancy.
There have been reports of development of addiction and physical dependence in patients receiving parenteral pentazocine. People with a history of drug abuse or alcohol abuse may have a higher chance of becoming addicted to opioid medicines.
Abrupt dose cessation or rapid dose reduction following the extended use of parenteral pentazocine has resulted in withdrawal symptoms such as abdominal cramps, nausea, vomiting, elevated temperature, chills, rhinorrhea, restlessness, anxiety, or lacrimation. In general opioid therapy should not be abruptly discontinued. When the patient no longer requires treatment with pentazocine and naloxone tablets, the drug should be tapered gradually to prevent signs and symptoms of withdrawal in patients who have been receiving opioids for an extended period of time and might have become physically dependent.
In prescribing pentazocine and naloxone tablets for chronic use, the physician should take under consideration that proper assessment of the patient, proper prescribing practices, periodic re-evaluation of therapy, and proper dispensing and storage are appropriate measures that help to identify and decrease misuse and abuse of opioid drugs.
The amount of naloxone present in pentazocine and naloxone tablets (0.5 mg per tablet) has no action when taken orally and will not interfere with the pharmacologic action of pentazocine. However, this amount of naloxone given by injection has profound antagonistic action to narcotic analgesics.
Severe, even lethal, consequences may result from misuse of tablets by injection either alone or in combination with other substances, such as pulmonary emboli, vascular occlusion, ulceration and abscesses, and withdrawal symptoms in narcotic dependent individuals.
Caution should be used when pentazocine and naloxone tablets is administered to patients prone to seizures; seizures have occurred in a few such patients in association with the use of pentazocine though no cause and effect relationship has been established.
Particular caution should be exercised in administering pentazocine to patients with porphyria since it may provoke an acute attack in susceptible individuals.
Pentazocine can elevate blood pressure, possibly through the release of endogenous catecholamines. Particular caution should be exercised in conditions where alterations in vascular resistance and blood pressure might be particularly undesirable, such as in the acute phase of myocardial infarction.
Pentazocine and naloxone tablets should be used with caution in patients with myocardial infarction who have nausea or vomiting.
Decreased metabolism of pentazocine by the liver in extensive liver disease may predispose to accentuation of side effects. Although laboratory tests have not indicated that pentazocine causes or increases renal or hepatic impairment, the drug should be administered with caution to patients with such impairment.
Caution should also be observed when administering pentazocine and naloxone tablets in patients with hypothyroidism, adrenocortical insufficiency, prostate hypertrophy, inflammatory or obstructive bowel disease, acute abdominal syndromes of unknown etiology, cholecystitis, pancreatitis, or acute alcohol intoxication and delirium tremens.
Narcotic drug products are generally considered to elevate biliary tract pressure for varying periods following their administration. Some evidence suggests that pentazocine may differ from other marketed narcotics in this respect (i.e., it causes little or no elevation in biliary tract pressures). The clinical significance of these findings, however, is not yet known.
Patients receiving pentazocine and naloxone tablets should be given the following instructions by the physician:
- Patients should be advised that pentazocine and naloxone tablets are a narcotic pain reliever, and should be taken only as directed.
- The dose of pentazocine and naloxone tablets should not be adjusted without consulting with a physician or other healthcare professional.
- Patients should be advised that pentazocine and naloxone tablets may cause drowsiness, dizziness, or lightheadedness and may impair mental and/or physical ability required for the performance of potentially hazardous tasks (e.g., driving, operating machinery). Patients started on pentazocine and naloxone tablets or patients whose dose has been adjusted should refrain from any potentially dangerous activity until it is established that they are not adversely affected.
- Pentazocine and naloxone tablets will add to the effect of alcohol and other CNS depressants (such as antihistamines, sedatives, hypnotics, tranquilizers, general anesthetics, phenothiazines, other opioids, and monoamine oxidase [MAO] inhibitors).
- Patients should not combine pentazocine and naloxone tablets with alcohol or other central nervous system depressants (sleep aids, tranquilizers) except by the orders of the prescribing physician, because dangerous additive effects may occur, resulting in serious injury or death.
- Women of childbearing potential who become or are planning to become pregnant should consult a physician prior to initiating or continuing therapy with pentazocine and naloxone tablets.
- Safe use in pregnancy has not been established. Prolonged use of opioid analgesics during pregnancy may cause neonatal physical dependence, and neonatal withdrawal may occur.
- If patients have been receiving treatment with pentazocine and naloxone tablets for more than a few weeks and cessation of therapy is indicated, they should be counseled on the importance of safely tapering the dose and that abruptly discontinuing the medication could precipitate withdrawal symptoms. The physician should provide a dose schedule to accomplish a gradual discontinuation of the medication.
- Patients should be advised that pentazocine and naloxone tablets are a potential drug of abuse. They should protect it from theft. It should never be given to anyone other than the individual for whom it was prescribed.
- Patients should be instructed to keep pentazocine and naloxone tablets in a secure place out of the reach of children. When pentazocine and naloxone tablets are no longer needed, please consult your pharmacist for proper disposal instructions.
- As with other opioids, patients taking pentazocine and naloxone tablets should be advised of the potential for severe constipation; appropriate laxatives and/or stool softeners as well as other appropriate treatments should be initiated from the onset of opioid therapy.
- Patients should be advised of the most common adverse events that may occur while taking pentazocine and naloxone tablets: constipation, nausea, somnolence, lightheadedness, dizziness, sedation, vomiting, and sweating.
CNS Depressants
Other central nervous system (CNS) depressants including sedatives, hypnotics, general anesthetics, antiemetics, phenothiazines, or other tranquilizers or alcohol increases the risk of respiratory depression, hypotension, profound sedation, or coma. Use morphine sulfate with caution and in reduced dosages in patients taking these agents.
Pentazocine and naloxone tablets can antagonize the effects of a pure opioid agonist analgesic and/or may precipitate withdrawal symptoms.
Concomitant use of monoamine oxidase inhibitors (MAOIs) with pentazocine and naloxone tablets may cause CNS excitation and hypertension through their respective effects on catecholamines. Caution should therefore be observed in administering pentazocine and naloxone tablets to patients who are currently receiving MAOIs or who have received them within the preceding 14 days
Anticholinergics or other medications with anticholinergic activity when used concurrently with opioid analgesics may result in increased risk of urinary retention and/or severe constipation, which may lead to paralytic ileus.
Smoking tobacco could enhance the metabolic clearance rate of pentazocine reducing the clinical effectiveness of a standard dose of pentazocine.
No long-term studies in animals to test for carcinogenesis have been performed with the components of pentazocine and naloxone tablets.
Studies to evaluate the mutagenic potential of the components of pentazocine and naloxone tablets have not been conducted.
Pentazocine, when administered orally or parenterally, had no adverse effect on either the reproductive capabilities or the course of pregnancy in rabbits and rats. Embryotoxic effects on the fetuses were not shown.
The daily administration of 4 mg/kg to 20 mg/kg pentazocine subcutaneously to female rats during a 14 day pre-mating period and until the 13th day of pregnancy did not have any adverse effects on the fertility rate.
Teratogenic
Effects
Pregnancy Category C
There are no adequate and well-controlled studies in pregnant women. Pentazocine and naloxone tablets should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
In a published report, a single dose of pentazocine administered to pregnant hamsters on gestation day 8 increased the incidence of exencephaly and cranioschisis at a dose of 196 mg/kg, SC (0.4-times the maximum daily human dose of pentazocine via 12 tablets on a mg/m2 basis).
Animal reproduction studies testing the combination of pentazocine and naloxone during organogenesis have been completed in rats and rabbits. In rats, a pentazocine:naloxone dose of 64 mg/kg:0.64 mg/kg via oral gavage increased the incidences of resorptions and extra ribs (0.2times the maximum daily human dose of pentazocine via 12 tablets on a mg/m2 basis). There were no clear treatment related effects in rabbits treated with a pentazocine:naloxone dose of up to 64 mg/kg:0.64 mg/kg via oral gavage (0.3-times the maximum daily human dose of pentazocine via 12 tablets on a mg/m2 basis).
There has been no experience in this regard with pentazocine and naloxone tablets. However, there have been rare reports of possible abstinence syndromes in newborns after prolonged use of pentazocine during pregnancy.
Patients receiving pentazocine during labor have experienced no adverse effects other than those that occur with commonly used analgesics. However, pentazocine can cross the placental barrier and cause central nervous system depression in the newborn and, if used regularly throughout pregnancy, may lead to symptoms of withdrawal in the newborn. Pentazocine and naloxone tablets should be used with caution in women delivering premature infants. The effect of pentazocine and naloxone tablets on the mother and fetus, the duration of labor or delivery, the possibility that forceps delivery or other intervention or resuscitation of the newborn may be necessary, or the effect of pentazocine and naloxone tablets on the later growth, development, and functional maturation of the child are unknown at the present time.
Pentazocine is excreted in human milk. Caution should be exercised when pentazocine and naloxone tablets are administered to a nursing woman.
Safety and effectiveness in pediatric patients below the age of 12 years have not been established.
Controlled clinical studies of pentazocine and naloxone tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses or effectiveness in analgesic activity between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Cardiovascular - Hypertension, hypotension, circulatory depression, tachycardia, syncope.
Respiratory - Rarely, respiratory depression.
Acute CNS Manifestations - Hallucinations (usually visual), disorientation, and confusion .
Other CNS Effects - Grand mal convulsions, increase in intracranial pressure, dizziness, lightheadedness, hallucinations, sedation, euphoria, headache, confusion, disorientation; infrequently weakness, disturbed dreams, insomnia, syncope, and depression; and rarely tremor, irritability, excitement, tinnitus.
Autonomic - Sweating; infrequently flushing; and rarely chills.
Gastrointestinal - Nausea, vomiting, constipation, diarrhea, anorexia, dry mouth, biliary tract spasm, and rarely abdominal distress.
Allergic - Edema of the face; anaphylactic shock; dermatitis, including pruritus; flushed skin, including plethora; infrequently rash, and rarely urticaria.
Ophthalmic - Visual blurring and focusing difficulty, miosis.
Hematologic - Depression of white blood cells (especially granulocytes), with rare cases of agranulocytosis, which is usually reversible, moderate transient eosinophilia.
Dependence and Withdrawal Symptoms - (See WARNINGS , PRECAUTIONS , and DRUG ABUSE AND DEPENDENCE Sections).
Other - Urinary retention, paresthesia, serious skin reactions, including erythema multiforme, Stevens-Johnson syndrome toxic epidermal necrolysis, and alterations in rate or strength of uterine contractions during labor.
For pentazocine alone in single doses above 60 mg there have been reports of the occurrence of nalorphine-like psychotomimetic effects such as anxiety, nightmares, strange thoughts, and hallucinations. Somnolence, marked respiratory depression associated with hypertension and tachycardia have also resulted as have seizures, hypotension, dizziness, nausea, vomiting, lethargy, and paresthesias. The respiratory depression is antagonized by naloxone (see Treatment ). Circulatory failure and deepening coma may occur in more severe cases, particularly in patients who have also ingested other CNS depressants such as alcohol, sedative/hypnotics, or antihistamines.
Adequate measures to maintain ventilation and general circulatory support should be employed. Assisted or controlled ventilation, intravenous fluids, vasopressors, and other supportive measures should be employed as indicated. Consideration should be given to gastric lavage and gastric aspiration. For respiratory depression due to overdosage or unusual sensitivity to pentazocine, parenteral naloxone is a specific and effective antagonist. Initial doses of 0.4 to 2.0 mg of naloxone are recommended, repeated at 2 to 3 minute intervals, if needed, up to a total of 10 mg. Anti-convulsant therapy may be necessary.
The usual initial adult dose is 1 tablet every three or four hours. This may be increased to 2 tablets when needed. Total daily dosage should not exceed 12 tablets.
Due to the potential for withdrawal symptoms associated with abrupt discontinuation, consideration should be given to tapering patients off pentazocine and naloxone tablets after prolonged periods of treatment with pentazocine and naloxone tablets (See PRECAUTIONS, Drug Abuse and Dependence ).
Pentazocine and naloxone tablets USP are light green, scored, capsule shaped tablets debossed 395 to the left of the score, 50 over 0.5 to the right of the score and WATSON on the reverse side supplied in bottles of 100.
Bottles of 100…………………………………..NDC 0591-0395-01
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP.
Manufactured by:
Watson Laboratories, Inc.
Corona, CA 92880 USA
Distributed by:
Watson
Pharma
, Inc.
Parsippany, NJ 07054 USA
Revised: October 2013
173017-1
NDC 0591-0395-01
CIV
Pentazocine and
Naloxone
Tablets USP
50 mg*/0.5mg*

Pentazocine and naloxonePentazocine hydrochloride and naloxone hydrochloride TABLET
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||