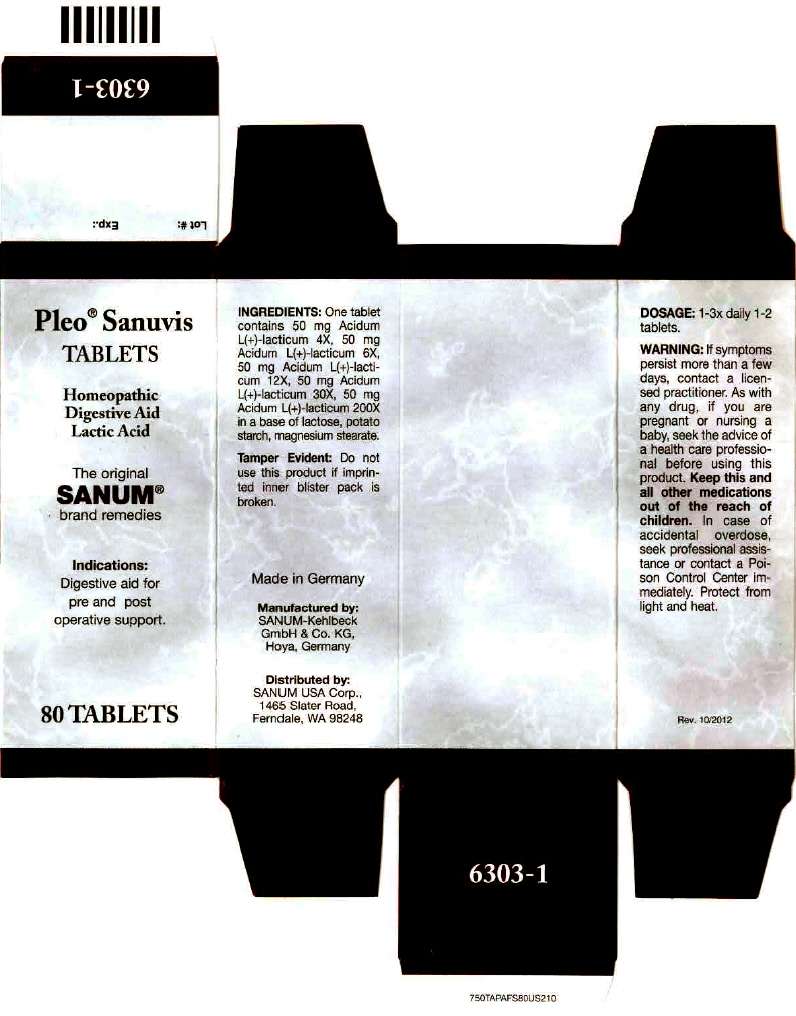
Pleo Sanuvis
Pleo Sanuvis TABLETS
FULL PRESCRIBING INFORMATION: CONTENTS*
FULL PRESCRIBING INFORMATION
Purpose
Homeopathic Digestive Aid Lactic Acid
80 Tablets
Indications
Digestive aid for pre and post operative support.
INGREDIENTS
One tablet contains 50 mg Acidum L(+)-lacticum 4X, 50 mg Acidum L(+)-lacticum 6X , 50 mg Acidum L(+)-lacticum 12X, 50 mg Acidum L(+)-lacticum 30X, 50 mg Acidum L(+)-lacticum 200X in a base of lactose, potato starch, magnesium stearate.
Tamper Evident
Do not use this product if imprinted inner blister pack is broken.
DOSAGE
1-3x daily 1-2 tablet.
WARNING
If symptoms persist more than a few days, contact a licensed practitioner. As with any drug, if you are pregnant or nursing a baby, seek the advice of a health care professional before using this product.
Keep this and all other medications out of the reach of children. In case of accidental overdose, seek professional assistance or contact a Poison Control Center immediately.
Protect fom light and heat.
Made in Germany
Distributed by:
SANUM USA Corp.
1465 Slater Road,
Ferndale, WA 98248
Manufactured by:
Sanum-Kehlbeck GmbH & Co. KG
Hoya, Germany
Rev. 10/2012
PRINCIPAL DISPLAY PANEL - 80 Tablet Carton
Pleo
®
Sanuvis
TABLETS
Homeopathic
Digestive Aid
Lactic Acid
The original
SANUM
®
brand remedies
Indications:
Digestive aid for
pre and post
operative support.
80 TABLETS
Pleo Sanuvislactic acid, l- TABLET
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||