Prometh with Codeine
PROMETHAZINE HYDROCHLORIDE AND CODEINE PHOSPHATE SYRUP
FULL PRESCRIBING INFORMATION: CONTENTS*
- SPL UNCLASSIFIED
- PROMETH WITH CODEINE DESCRIPTION
- CLINICAL PHARMACOLOGY
- INDICATIONS & USAGE
- PROMETH WITH CODEINE CONTRAINDICATIONS
- WARNINGS
- BOXED WARNING
- SPL UNCLASSIFIED
- PRECAUTIONS
- PROMETH WITH CODEINE ADVERSE REACTIONS
- DRUG ABUSE AND DEPENDENCE
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
SPL UNCLASSIFIED
CV
FORM NO. 1627
Rev. 1/09
VC3277
Rx Only
PROMETH WITH CODEINE DESCRIPTION
Each 5 mL (one teaspoonful), for oral administration contains: Promethazine hydrochloride 6.25 mg; codeine phosphate 10 mg. Alcohol 7%.
Inactive Ingredients: Citric acid, corn syrup, D&C Red #33, FD&C Blue #1, flavor, methylparaben, propylene glycol, propylparaben, purified water, saccharin sodium, sodium benzoate, sodium citrate.
Codeine is one of the naturally occurring phenanthrene alkaloids of opium derived from the opium poppy; it is classified pharmacologically as a narcotic analgesic. Codeine phosphate may be chemically designated as 7,8-Didehydro-4,5α-epoxy-3-methoxy-17-methylmorphinan-6α -ol phosphate (1:1)(salt) hemihydrate.
The phosphate salt of codeine occurs as white, needle-shaped crystals or white crystalline powder. Codeine phosphate is freely soluble in water and slightly soluble in alcohol. It has a molecular weight of 406.37, a molecular formula of C18H21NO3•H3PO4•½H2O, and the following structural formula:

Promethazine hydrochloride, a phenothiazine derivative, is chemically designated as (±)-10-[2-(Dimethylamino)propyl] phenothiazine monohydrochloride.
Promethazine hydrochloride occurs as a white to faint yellow, practically odorless, crystalline powder which slowly oxidizes and turns blue on prolonged exposure to air. It is soluble in water and freely soluble in alcohol. It has a molecular weight of 320.88, a molecular formula of C17H20N2S•HCl, and the following structural formula:

CLINICAL PHARMACOLOGY
Codeine: Narcotic analgesics, including codeine, exert their primary effects on the central nervous system and gastrointestinal tract. The analgesic effects of codeine are due to its central action; however, the precise sites of action have not been determined, and the mechanisms involved appear to be quite complex. Codeine resembles morphine both structurally and pharmacologically, but its actions at the doses of codeine used therapeutically are milder, with less sedation, respiratory depression and gastrointestinal, urinary, and pupillary effects. Codeine produces an increase in biliary tract pressure, but less than morphine or meperidine. Codeine is less constipating than morphine.
Codeine has good antitussive activity, although less than that of morphine at equal doses. It is used in preference to morphine, because side effects are infrequent at the usual antitussive dose of codeine.
Codeine in oral therapeutic dosage does not usually exert major effects on the cardiovascular system.
Narcotic analgesics may cause nausea and vomiting by stimulating the chemoreceptor trigger zone (CTZ); however, they also depress the vomiting center, so that subsequent doses are unlikely to produce vomiting. Nausea is minimal after usual oral doses of codeine.
Narcotic analgesics cause histamine release, which appears to be responsible for wheals or urticaria sometimes seen at the site of injection on parenteral administration. Histamine release may also produce dilation of cutaneous blood vessels, with resultant flushing of the face and neck, pruritus, and sweating.
Codeine and its salts are well absorbed following both oral and parenteral administration. Codeine is about 2/3 as effective orally as parenterally. Codeine is metabolized primarily in the liver by enzymes of the endoplasmic reticulum, where it undergoes O-demethylation, N-demethylation, and partial conjugation with glucuronic acid. The drug is excreted primarily in the urine, largely as inactive metabolites and small amounts of free and conjugated morphine. Negligible amounts of codeine and its metabolites are found in the feces.
Following oral or subcutaneous administration of codeine, the onset of analgesia occurs within 15 to 30 minutes and lasts for four to six hours.
The cough-depressing action, in animal studies, was observed to occur 15 minutes after oral administration of codeine, peak action at 45 to 60 minutes after ingestion. The duration of action, which is dose-dependent, usually did not exceed 3 hours.
Promethazine: Promethazine is a phenothiazine derivative which differs structurally from the antipsychotic phenothiazines by the presence of a branched side chain and no ring substitution. It is thought that this configuration is responsible for its relative lack (1/10 that of chlorpromazine) of dopamine antagonist properties.
Promethazine is an H1 receptor blocking agent. In addition to its antihistaminic action, it provides clinically useful sedative and antiemetic effects.
Promethazine is well absorbed from the gastrointestinal tract. Clinical effects are apparent within 20 minutes after oral administration and generally last four to six hours, although they may persist as long as 12 hours. Promethazine is metabolized by the liver to a variety of compounds; the sulfoxides of promethazine and N-demethylpromethazine are the predominant metabolites appearing in the urine.
INDICATIONS & USAGE
Promethazine hydrochloride and codeine phosphate syrup is indicated for the temporary relief of coughs and upper respiratory symptoms associated with allergy or the common cold.
PROMETH WITH CODEINE CONTRAINDICATIONS
The combination of promethazine hydrochloride and codeine phosphate is contraindicated in pediatric patients less than 6 years of age, because the combination may cause fatal respiratory depression in this age population.
Codeine is contraindicated in patients with a known hypersensitivity to the drug.
Promethazine is contraindicated in comatose states, and in individuals known to be hypersensitive or to have had an idiosyncratic reaction to promethazine or to other
phenothiazines.
Antihistamines and codeine are both contraindicated for use in the treatment of lower respiratory tract symptoms, including asthma.
WARNINGS
WARNINGS
BOXED WARNING
The combination of promethazine hydrochloride and codeine phosphate is contraindicated in pediatric patients less than 6 years of age. Concomitant administration of promethazine products with other respiratory depressants has an association with respiratory depression, and sometimes death, in pediatric patients.
Postmarketing cases of respiratory depression, including fatalities, have been reported with use of promethazine hydrochloride in pediatric patients less than 2 years of age. A wide range of weight-based doses of promethazine hydrochloride have resulted in respiratory depression in these patients.
SPL UNCLASSIFIED
Codeine: Dosage of codeine SHOULD NOT BE INCREASED if cough fails to respond; an unresponsive cough should be reevaluated in 5 days or sooner for possible underlying pathology, such as foreign body or lower respiratory tract disease.
Codeine may cause or aggravate constipation.
Respiratory depression leading to arrest, coma, and death has occurred with the use of codeine antitussives in young children, particularly in the under-one-year infants whose ability to deactivate the drug is not fully developed.
Administration of codeine may be accompanied by histamine release and should be used with caution in atopic children.
Head Injury And Increased Intracranial Pressure: The respiratory-depressant effects of narcotic analgesics and their capacity to elevate cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, intracranial lesions or a preexisting increase in intracranial pressure. Narcotics may produce adverse reactions which may obscure the clinical course of patients with head injuries.
Asthma And Other Respiratory Conditions: Narcotic analgesics or cough suppressants, including codeine, should not be used in asthmatic patients (see CONTRAINDICATIONS). Nor should they be used in acute febrile illness associated with productive cough or in chronic respiratory disease where interference with ability to clear the tracheobronchial tree of secretions would have a deleterious effect on the patient’s respiratory function.
Hypotensive Effect: Codeine may produce orthostatic hypotension in ambulatory patients.
CNS Depression - Promethazine may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a vehicle or operating machinery. The impairment may be amplified by concomitant use of other central-nervous-system depressants such as alcohol, sedatives/hypnotics (including barbiturates), narcotics, narcotic analgesics, general anesthetics, tricyclic antidepressants, and tranquilizers; therefore such agents should either be eliminated or given in reduced dosage in the presence of promethazine HCl (see PRECAUTIONS-Information for Patients and Drug Interactions).
Respiratory Depression - Promethazine may lead to potentially fatal respiratory depression.
Use of Promethazine in patients with compromised respiratory function (e.g., COPD, sleep apnea) should be avoided.
Lower Seizure Threshold - Promethazine may lower seizure threshold. It should be used with caution in persons with seizure disorders or in persons who are using concomitant medications, such as narcotics or local anesthetics, which may also affect seizure threshold.
Bone-Marrow Depression - Promethazine should be used with caution in patients with bone-marrow depression. Leukopenia and agranulocytosis have been reported, usually when promethazine HCl has been used in association with other known marrow-toxic agents.
Neuroleptic Malignant Syndrome - A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with promethazine HCl alone or in combination with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmias).
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (e.g. pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever and primary central nervous system (CNS) pathology.
The management of NMS should include 1) immediate discontinuation of promethazine HCl, antipsychotic drugs, if any, and other drugs not essential to concurrent therapy, 2) intensive symptomatic treatment and medical monitoring, and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
Since recurrences of NMS have been reported with phenothiazines, the reintroduction of
promethazine HCl should be carefully considered.
The combination of promethazine hydrochloride and codeine phosphate is contraindicated in pediatric patients less than 6 years of age. Concomitant administration of promethazine products with other respiratory depressants has an association with respiratory depression, and sometimes death, in pediatric patients. The association does not directly relate to individualized weight-based dosing, which might otherwise permit safe administration.
Excessively large dosages of antihistamines, including promethazine hydrochloride, in pediatric patients may cause sudden death (see OVERDOSAGE). Hallucinations and convulsions have occurred with therapeutic doses and overdoses of promethazine hydrochloride in pediatric patients. In pediatric patients who are acutely ill associated with dehydration, there is an increased susceptibility to dystonias with the use of promethazine HCl.
Administration of promethazine has been associated with reported cholestatic jaundice.
PRECAUTIONS
Animal reproduction studies have not been conducted with the drug combination-promethazine and codeine. It is not known whether this drug combination can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Promethazine hydrochloride and codeine phosphate should be given to a pregnant woman only if clearly needed.
Narcotic analgesics, including codeine, should be administered with caution and the initial dose reduced in patients with acute abdominal conditions, convulsive disorders, significant hepatic or renal impairment, fever, hypothyroidism, Addison’s disease, ulcerative colitis, prostatic hypertrophy, in patients with recent gastrointestinal or urinary tract surgery, and in the very young or elderly or debilitated patients.
Drugs having anticholinergic properties should be used with caution in patients with
narrow-angle glaucoma, prostatic hypertrophy, stenosing peptic ulcer, pyloroduodenal
obstruction, and bladder-neck obstruction.
Promethazine should be used cautiously in persons with cardiovascular disease or with impairment of liver function.
Some individuals may be ultra-rapid metabolizers due to a specific CYP2D6*2x2 genotype. These individuals convert codeine into its a active metabolite, morphine, more rapidly and completely than other people. This rapid conversion results in higher than expected serum morphine levels. Even at labeled dosage regiments, individuals who are ultra-rapid metabolizers may experience overdose symptoms such as extreme sleepiness, confusion, and shallow breathing.
The prevalence of this CYP2D6 phenotype varies widely and has been estimated at 0.5 to 1% in Chinese and Japanese, 0.5 to 1% in Hispanics, 1-10% in Caucasians, 3% in African Americans, and 16-28% in North Africans, Ethiopians and Arabs. Data is not available for other ethnic groups.
When physicians prescribe codeine-containing drugs, they should choose the lowest effective dose for the shortest period of time and should inform their patients about these risks and the signs of morphine overdose. (See Precautions-Nursing Mothers).
Patients should be advised to measure Promethazine with Codeine Oral Solution with an accurate measuring device. A household teaspoon is not an accurate measuring device and could lead to overdosage, especially when a half a teaspoon is measured. A pharmacist can recommend an appropriate measuring device and can provide instructions for measuring the correct dose.
Promethazine and codeine may cause marked drowsiness or may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a vehicle or operating machinery. Ambulatory patients should be told to avoid engaging in such activities until it is known that they do not become drowsy or dizzy from promethazine and codeine therapy. Pediatric patients should be supervised to avoid potential harm in bike riding or in other hazardous activities.
The concomitant use of alcohol or other central-nervous-system depressants, such as sedatives/hypnotics (including barbiturates), narcotics, narcotic analgesics, general anesthetics, tricyclic antidepressants, and tranquilizers may enhance impairment and should be avoided or their dosage reduced (see WARNINGS-CNS Depression and PRECAUTIONS-Drug Interactions)
Patients should be advised to report any involuntary muscle movements.
Avoid prolonged exposure to the sun.
Codeine, like other narcotic analgesics, may produce orthostatic hypotension in some ambulatory patients. Patients should be cautioned accordingly.
Caution patients that some people have a variation in a liver enzyme and change codeine into morphine more rapidly and completely than other people. These people are ultra-rapid metabolizers and are more likely to have higher-than-normal levels of morphine in their blood after taking codeine, which can result in overdose symptoms such as extreme sleepiness, confusion, or shallow breathing. In most cases, it is unknown if someone is an ultra-rapid codeine metabolizer.
Nursing mothers taking codeine can also have higher morphine levels in their breast milk if they are ultra-rapid metabolizers. These higher levels of morphine in breast milk may lead to life-threatening or fatal side effects in nursing babies. Instruct nursing mothers to watch for signs of morphine toxicity in their infants including increased sleepiness (more than usual), difficulty breastfeeding, breathing difficulties, or limpness. Instruct nursing mothers to talk to the baby’s doctor immediately if they notice these signs and, if they cannot reach the doctor right away, to take the baby to an emergency room or call 911 (or local emergency services).
Codeine: In patients receiving MAO inhibitors, an initial small test dose is advisable to allow observation of any excessive narcotic effects or MAOI interaction.
CNS depressants - Promethazine may increase, prolong, or intensify the sedative action of other central-nervous-system depressants, such as alcohol, sedatives/hypnotics (including barbiturates), narcotics, narcotic analgesics, general anesthetics, tricyclic antidepressants, and tranquilizers; therefore, such agents should be avoided or administered in reduced dosage to patients receiving promethazine HCl. When given concomitantly with promethazine, the dose of barbiturates should be reduced by at least one-half, and the dose of narcotics should be reduced by one-quarter to one-half. Dosage must be individualized. Excessive amounts of promethazine HCl relative to a narcotic may lead to restlessness and motor hyperactivity in the patient with pain; these symptoms usually disappear with adequate control of the pain.
Epinephrine - Because of the potential for promethazine to reverse epinephrine’s vasopressor effect, epinephrine should NOT be used to treat hypotension associated with promethazine overdose.
Anticholinergics - Concomitant use of other agents with anticholinergic properties should be undertaken with caution.
Monoamine oxidase inhibitors (MAOI) - Drug interactions, including an increased incidence of extrapyramidal effects, have been reported when some MAOI and phenothiazines are used concomitantly.
Because narcotic analgesics may increase biliary tract pressure with resultant increases in plasma amylase or lipase levels, determination of these enzyme levels may be unreliable for 24 hours after a narcotic analgesic has been given.
The following laboratory tests may be affected in patients who are receiving therapy with promethazine hydrochloride.
Pregnancy Tests: Diagnostic pregnancy tests based on immunological reactions between HCG and anti-HCG may result in false-negative or false-positive interpretations.
Glucose Tolerance Test: An increase in blood glucose has been reported in patients receiving promethazine.
Long-term animal studies have not been performed to assess the carcinogenic potential of codeine or of promethazine, nor are there other animal or human data concerning carcinogenicity, mutagenicity, or impairment of fertility with these agents. Codeine has been reported to show no evidence of carcinogenicity or mutagenicity in a variety of test systems, including the micronucleus and sperm abnormality assays and the Salmonella assay. Promethazine was nonmutagenic in the Salmonella test system of Ames.
Pregnancy Category C.
Codeine: A study in rats and rabbits reported no teratogenic effect of codeine administered during the period of organogenesis in doses ranging from 5 to 120 mg/kg. In the rat, doses at the 120-mg/kg level, in the toxic range for the adult animal, were associated with an increase in embryo resorption at the time of implantation. In another study a single 100-mg/kg dose of codeine administered to pregnant mice reportedly resulted in delayed ossification in the offspring.
There are no studies in humans, and the significance of these findings to humans, if any, is not known.
Promethazine: Teratogenic effects have not been demonstrated in rat-feeding studies at doses of 6.25 and 12.5 mg/kg of promethazine HCl. These doses are from approximately 2.1 to 4.2 times the maximum recommended total daily dose of promethazine for a 50-kg subject, depending upon the indication for which the drug is prescribed. Daily doses of 25 mg/kg intraperitoneally have been found to produce fetal mortality in rats.
Specific studies to test the action of the drug on parturition, lactation, and development of the animal neonate were not done, but a general preliminary study in rats indicated no effect on these parameters. Although antihistamines have been found to produce fetal mortality in rodents, the pharmacological effects of histamine in the rodent do not parallel those in man. There are no adequate and well-controlled studies of promethazine in pregnant women.
Promethazine and codeine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Dependence has been reported in newborns whose mothers took opiates regularly during pregnancy. Withdrawal signs include irritability, excessive crying, tremors, hyperreflexia, fever, vomiting, and diarrhea. Signs usually appear during the first few days of life.
Promethazine administered to a pregnant woman within two weeks of delivery may inhibit platelet aggregation in the newborn.
Narcotic analgesics cross the placental barrier. The closer to delivery and the larger the dose used, the greater the possibility of respiratory depression in the newborn. Narcotic analgesics should be avoided during labor if delivery of a premature infant is anticipated. If the mother has received narcotic analgesics during labor, newborn infants should be observed closely for signs of respiratory depression. Resuscitation may be required (see OVERDOSAGE).
Limited data suggest that use of promethazine hydrochloride during labor and delivery does not have an appreciable effect on the duration of labor or delivery and does not increase the risk of need for intervention in the newborn.
The effect of promethazine and/or codeine on later growth and development of the newborn is unknown.
It is not known whether promethazine is excreted in human milk.
Codeine is secreted into human milk. In women with normal codeine metabolism (normal CYP2D6 activity), the amount of codeine secreted into human milk is low and dose-dependent. Despite the common use of codeine products to manage postpartum pain, reports of adverse events in infants are rare. However, some women are ultra-rapid metabolizers of codeine. These women achieve higher-than-expected serum levels of codeine’s active metabolite, morphine, leading to higher-than-expected levels of morphine in breast milk and potentially dangerously high serum morphine levels in their breastfed infants. Therefore, maternal use of codeine can potentially lead to serious adverse reactions, including death, in nursing infants.
The prevalence of this CYP2D6 phenotype varies widely and has been estimated at 0.5 to 1% in Chinese and Japanese, 0.5 to 1% in Hispanics, 1-10% in Caucasians, 3% in African Americans, and 16-28% in North Africans, Ethiopians and Arabs. Data is not available for other ethnic groups.
The risk of infant exposure to codeine and morphine through breast milk should be weighed against the benefits of breastfeeding for both the mother and baby. Caution should be exercised when codeine is administered to a nursing woman. If a codeine containing product is selected, the lowest dose should be prescribed for the shortest period of time to achieve the desired clinical effect. Mothers using codeine should be informed about when to receive immediate medical care and how to identify the signs and symptoms of neonatal toxicity, such as drowsiness or sedation, difficulty breastfeeding, breathing difficulties, and decreased tone, in their baby. Nursing mothers who are ultra-rapid metabolizers may also experience overdose symptoms such as extreme sleepiness, confusion or shallow breathing. Prescribers should closely monitor mothers-infant pairs and notify treating pediatricians about the use of codeine during breastfeeding. (See PRECAUTIONS – General – Ultra-rapid metabolizers of Codeine).
Caution should be exercised when promethazine hydrochloride and codeine phosphate syrup is administered to a nursing woman.
The combination of promethazine hydrochloride and codeine phosphate is contraindicated in pediatric patients less than 6 years of age, because the combination may cause fatal respiratory depression in this age population (see WARNINGS Box Warning and Use in Pediatric Patients).
The combination of promethazine hydrochloride and codeine phosphate should be used with caution in pediatric patient 6 years and older (see WARNINGS – Use in Pediatric Patients).
Clinical studies of promethazine hydrochloride and codeine phosphate syrup did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
Sedating drugs may cause confusion and over-sedation in the elderly; elderly patients generally should be started on low doses of Promethazine Hydrochloride and Codeine Phosphate Syrup and observed closely.
PROMETH WITH CODEINE ADVERSE REACTIONS
Nervous System - CNS depression, particularly respiratory depression, and to a lesser extent circulatory depression; light-headedness, dizziness, sedation, euphoria, dysphoria, headache, transient hallucination, disorientation, visual disturbances, and convulsions.
Cardiovascular - Tachycardia, bradycardia, palpitation, faintness, syncope, orthostatic hypotension (common to narcotic analgesics).
Gastrointestinal - Nausea, vomiting, constipation, and biliary tract spasm. Patients with chronic ulcerative colitis may experience increased colonic motility; in patients with acute ulcerative colitis, toxic dilation has been reported.
Genitourinary - Oliguria, urinary retention; antidiuretic effect has been reported (common to narcotic analgesics).
Allergic - Infrequent pruritus, giant urticaria, angioneurotic edema, and laryngeal edema.
Other - Flushing of the face, sweating and pruritus (due to opiate-induced histamine release); weakness.
Central Nervous System - Drowsiness is the most prominent CNS effect of this drug. Sedation, somnolence, blurred vision, dizziness; confusion, disorientation, and extrapyramidal symptoms such as oculogyric crisis, torticollis, and tongue protrusion; lassitude, tinnitus, incoordination, fatigue, euphoria, nervousness, diplopia, insomnia, tremors, convulsive seizures, excitation, catatonic-like states, hysteria. Hallucinations have also been reported.
Cardiovascular-Increased or decreased blood pressure, tachycardia, bradycardia, faintness.
Dermatologic-Dermatitis, photosensitivity, urticaria.
Hematologic-Leukopenia, thrombocytopenia, thrombocytopenic purpura, agranulocytosis.
Gastrointestinal-Dry mouth, nausea, vomiting, jaundice.
Respiratory-Asthma, nasal stuffiness, respiratory depression (potentially fatal) and apnea (potentially fatal). (See WARNINGS-Promethazine; Respiratory Depression.).
Other-Angioneurotic edema. Neuroleptic malignant syndrome (potentially fatal) has also been reported. (See WARNINGS-Promethazine; Neuroleptic Malignant Syndrome.)
Paradoxical Reactions - Hyperexcitability and abnormal movements have been reported in patients following a single administration of promethazine HCl. Consideration should be given to the discontinuation of promethazine HCl and to the use of other drugs if these reactions occur. Respiratory depression, nightmares, delirium, and agitated behavior have also been reported in some of these patients.
DRUG ABUSE AND DEPENDENCE
Promethazine hydrochloride and codeine phosphate syrup is a Schedule V Controlled Substance.
Codeine is known to be subject to abuse; however, the abuse potential of oral codeine appears to be quite low. Even parenteral codeine does not appear to offer the psychic effects sought by addicts to the same degree as heroin or morphine. However, codeine must be administered only under close supervision to patients with a history of drug abuse or dependence.
Psychological dependence, physical dependence, and tolerance are known to occur with codeine.
OVERDOSAGE
Codeine: Serious overdose with codeine is characterized by respiratory depression (a decrease in respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis), extreme somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, and sometimes bradycardia and hypotension. The triad of coma, pinpoint pupils, and respiratory depression is strongly suggestive of opiate poisoning. In severe overdosage, particularly by the intravenous route, apnea, circulatory collapse, cardiac arrest, and death may occur. Promethazine is additive to the depressant effects of codeine.
It is difficult to determine what constitutes a standard toxic or lethal dose. However, the lethal oral dose of codeine in an adult is reported to be in the range of 0.5 to 1.0 gram. Infants and children are believed to be relatively more sensitive to opiates on a body-weight basis. Elderly patients are also comparatively intolerant to opiates.
Promethazine: Signs and symptoms of overdosage with promethazine HCl range from mild depression of the central nervous system and cardiovascular system to profound hypotension,
respiratory depression, unconsciousness, and sudden death. Other reported reactions include hyperreflexia, hypertonia, ataxia, athetosis, and extensor-plantar reflexes (Babinski reflex).
Stimulation may be evident, especially in children and geriatric patients. Convulsions may rarely occur. A paradoxical-type reaction has been reported in children receiving single doses of 75 mg to 125 mg orally, characterized by hyperexcitability and nightmares.
Atropine-like signs and symptoms - dry mouth, fixed dilated pupils, flushing, as well as gastrointestinal symptoms may occur.
Treatment: The treatment of overdosage with promethazine and codeine is essentially symptomatic and supportive. Only in cases of extreme overdosage or individual sensitivity do vital signs including respiration, pulse, blood pressure, temperature, and EKG need to be monitored. Activated charcoal orally or by lavage may be given, or sodium or magnesium sulfate orally as a cathartic. Attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and institution of assisted or controlled ventilation. The narcotic antagonist, naloxone hydrochloride, may be administered when significant respiratory depression occurs with promethazine and codeine; any depressant effects of promethazine are not reversed with naloxone. Diazepam may be used to control convulsions. Avoid analeptics, which may cause convulsions. Acidosis and electrolyte losses should be corrected. A rise in temperature or pulmonary complications may signal the need for institution of antibiotic therapy.
Severe hypotension usually responds to the administration of norepinephrine or phenylephrine. EPINEPHRINE SHOULD NOT BE USED, since its use in a patient with partial adrenergic blockade may further lower the blood pressure.
Limited experience with dialysis indicates that it is not helpful.
DOSAGE & ADMINISTRATION
The combination of promethazine hydrochloride and codeine phosphate is contraindicated in pediatric patients less than 6 years of age, because the combination may cause fatal respiratory depression in this age population.
It is important that promethazine hydrochloride and codeine phosphate syrup is measured with an accurate measuring device (see PRECAUTIONS-Information for Patients). A household teaspoon is not an accurate measuring device and could lead to overdosage, especially when half a teaspoon is to be measured. It is strongly recommended that an accurate measuring device be used. A pharmacist can provide an appropriate device and can provide instructions for measuring the correct dose.
The average effective dose for adults and children (12 years and over) is: 1 teaspoonful (5mL) every 4 to 6 hours, not to exceed 30 mL in 24 hours.
The average effective dose for children 6 years to under 12 years of age is ½ to 1 teaspoonful (2.5 mL to 5 mL) every 4 to 6 hours, not to exceed 30 mL in 24 hours.
HOW SUPPLIED
Promethazine Hydrochloride and Codeine Phosphate Syrup contains promethazine hydrochloride 6.25 mg/5 mL, codeine phosphate 10 mg/5 mL and alcohol 7 percent, and is available in 4 fluid ounce (118 mL), 8 fluid ounce (237 mL), and pint (473 mL) bottles.
Store at 20 - 25°C (68 - 77°F) [see USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP.
Manufactured by
Actavis Mid Atlantic LLC
1877 Kawai Road
Lincolnton, NC 28092 USA
FORM NO. 1627
Rev. 1/09
VC3277
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
DRUG: Prometh with Codeine
GENERIC: promethazine hydrochloride and codeine phosphate
DOSAGE: SYRUP
ADMINSTRATION: ORAL
NDC: 52125-596-01
ACTIVE INGREDIENT(S):
- CODEINE PHOSPHATE 10mg in 5mL
- PROMETHAZINE HYDROCHLORIDE 6.25mg in 5mL
INACTIVE INGREDIENT(S):
- CITRIC ACID MONOHYDRATE
- WATER
- PROPYLPARABEN
- SACCHARIN SODIUM DIHYDRATE
- SODIUM BENZOATE
- PROPYLENE GLYCOL
- CORN SYRUP
- D&C RED NO. 33
- METHYLPARABEN
- FD&C BLUE NO. 1
- SODIUM CITRATE
PACKAGING: 118 mL in 1 BOTTLE

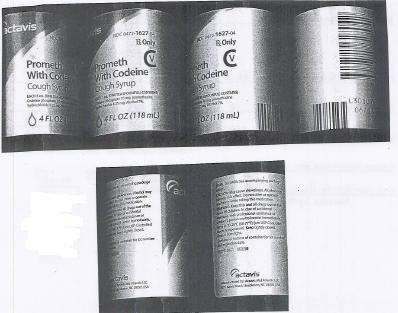
Prometh with Codeinepromethazine hydrochloride and codeine phosphate SYRUP
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||