Proparacaine Hydrochloride
Falcon Pharmaceuticals, Ltd.
Alcon Laboratories, Inc.
FULL PRESCRIBING INFORMATION: CONTENTS*
- PROPARACAINE HYDROCHLORIDE DESCRIPTION
- CLINICAL PHARMACOLOGY
- PROPARACAINE HYDROCHLORIDE INDICATIONS AND USAGE
- PROPARACAINE HYDROCHLORIDE CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
- PROPARACAINE HYDROCHLORIDE ADVERSE REACTIONS
- PROPARACAINE HYDROCHLORIDE DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- PRINCIPAL DISPLAY PANEL
FULL PRESCRIBING INFORMATION
DESCRIPTION
Proparacaine hydrochloride ophthalmic solution 0.5% is a topical local anesthetic for ophthalmic use. The active ingredient is represented by the structural formula:
Established name: Proparacaine Hydrochloride
Chemical name: Benzoic acid, 3-amino-4-propoxy-,2-(diethylamino) ethyl ester, monohydrochloride.
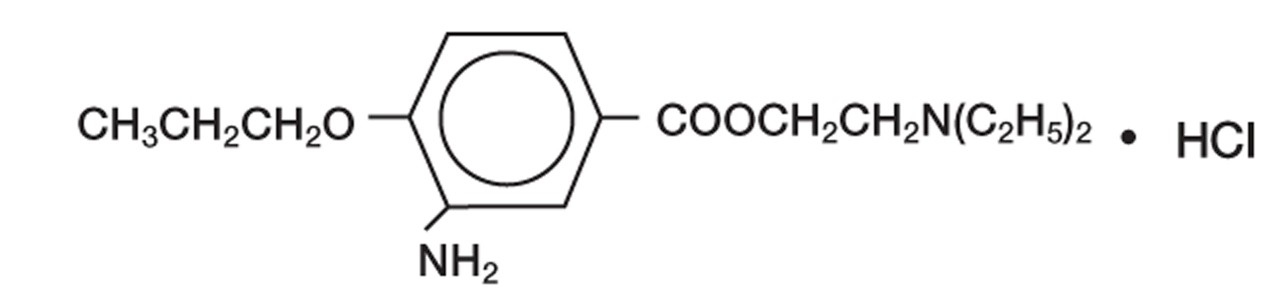
Molecular Weight: 330.85
Each mL contains: Active: proparacaine hydrochloride 5mg 0.5%. Preservative: benzalkonium chloride (0.01%). Inactives: glycerin; and purified water. The pH may be adjusted with hydrochloric acid and/or sodium hydroxide.
CLINICAL PHARMACOLOGY
Proparacaine hydrochloride ophthalmic solution is a rapidly-acting topical anesthetic, with induced anesthesia lasting approximately 10-20 minutes.
INDICATIONS AND USAGE
Proparacaine hydrochloride ophthalmic solution is indicated for procedures in which a topical ophthalmic anesthetic is indicated: corneal anesthesia of short duration, e.g. tonometry, gonioscopy, removal of corneal foreign bodies, and for short corneal and conjunctival procedures.
CONTRAINDICATIONS
Proparacaine hydrochloride ophthalmic solution should be considered contraindicated in patients with known hypersensitivity to any of the ingredients of this preparation.
WARNINGS
NOT FOR INJECTION - FOR TOPICAL OPHTHALMIC USE ONLY. Prolonged use of a topical ocular anesthetic is not recommended. It may produce permanent corneal opacification with accompanying visual loss.
PRECAUTIONS
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic potential, mutagenicity, or possible impairment of fertility in males or females.
Pregnancy
Pregnancy Category C: Animal reproduction studies have not been conducted with proparacaine hydrochloride ophthalmic solution 0.5%. It is also not known whether proparacaine hydrochloride can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Proparacaine hydrochloride should be administered to a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when proparacaine hydrochloride is administered to a nursing woman.
Pediatric Use
Safety and effectiveness of proparacaine hydrochloride ophthalmic solution in pediatric patients have been established. Use of proparacaine hydrochloride is supported by evidence from adequate and well-controlled studies in adults and children over the age of twelve, and safety information in neonates and other pediatric patients.
Geriatric Use
No overall clinical differences in safety or effectiveness have been observed between the elderly and other adult patients
ADVERSE REACTIONS
Occasional temporary stinging, burning and conjunctival redness may occur with the use of proparacaine. A rare, severe, immediate-type, apparently hyperallergic corneal reaction characterized by acute, intense and diffuse epithelial keratitis, a gray, ground glass appearance, sloughing of large areas of necrotic epithelium, corneal filaments and, sometimes, iritis with descemetitis has been reported.
Allergic contact dermatitis from proparacaine with drying and fissuring of the fingertips has also been reported.
DOSAGE AND ADMINISTRATION
Usual Dosage: Removal of foreign bodies and sutures, and for tonometry: 1 to 2 drops (in single instillations) in each eye before operating.
Short corneal and conjunctival procedures: 1 drop in each eye every 5 to 10 minutes for 5 to 7 doses.
NOTE: Proparacaine hydrochloride ophthalmic solution should be clear to straw-color. If the solution becomes darker, discard the solution.
HOW SUPPLIED
Proparacaine hydrochloride ophthalmic solution 0.5% is supplied in 15 mL DROP-TAINER® dispensers.
NDC 61314-016-01
Storage: Bottle must be stored in unit carton to protect contents from light. Store bottles under refrigeration at 2° - 8°C (36° - 46°F).
Rx Only
©2004 Falcon Pharmaceuticals, Ltd.
345491-0404
Dist by:
FALCON Pharmaceuticals, Ltd.
Fort Worth, TX 76134 USA
Mfd by:
ALCON LABORATORIES, INC.
Fort Worth, TX 76134 USA
Printed in USA
PRINCIPAL DISPLAY PANEL
NDC 61314-016-01 Rx Only
FALCON PHARMACEUTICALS®
Proparacaine
Hydrochloride
Ophthalmic
Solution, USP
0.5%
15 mL STERILE
AFFILIATE OF
ALCON LABORATORIES, INC.
QUALITY RX


Proparacaine HydrochlorideProparacaine Hydrochloride SOLUTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||