Reaphirm Plant Source DHA
Everett Laboratories, Inc.
Everett Laboratories, Inc.
Product Information.
FULL PRESCRIBING INFORMATION: CONTENTS*
- Contraindications
- Boxed Warning
- WARNINGS/PRECAUTIONS:
- Dosage and Administration
- How Supplied
- Principal Display Panel
FULL PRESCRIBING INFORMATION
Contraindications
Reaphirm Plant Source DHA is contraindicated in patients with hypersensitivity to any of its components or color additives.
Folic acid is contraindicated in patients with untreated and uncomplicated pernicious anemia, and in those with anaphylactic sensitivity to folic acid.
Iron Therapy is contraindicated in patients with hemochromatosis and patients with iron storage disease or the potential for iron storage disease due to chronic hemolytic anemia (e.g., inherited anomalies of hemoglobin structure or synthesis and/or red cell enzyme deficiencies, etc.), pyridoxine responsive anemia, or cirrhosis of the liver.
Cyanocobalamin is contraindicated in patients with sensitivity to cobalt or to cyanocobalamin (Vitamin B12).
Boxed Warning
WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of the reach of children. In case of accidental overdose, call a doctor or a Poison Control Center immediately.
WARNINGS/PRECAUTIONS:
Drug Interactions:
Information for Patients:
Pediatric Use:
Side Effects
Adverse reactions have been reported with specific vitamins and minerals, but generally at doses substantially higher than those in Reaphirm Plant Source DHA. Reported adverse events include skin ailments, gastrointestinal complaints, glucose abnormalities, and visual problems.
Dosage and Administration
Before, during and after pregnancy, one softgel capsule daily, or as directed by a physician.
How Supplied
Reaphirm Plant Source DHA is available as a dark blue, oval shaped softgel capsule imprinted "EV3010". Available in Box of Unit-Dose pack of 30 (5 child resistant blister cards of 6 softgel capsules).
Store at room temperature, approximately 15-30C (59-86F), avoid excessive heat.
Rx Only
Manufactured for
EVERETT LABORATORIES, INC.
West Orange, NJ 07052
1-877-324-9349
Principal Display Panel
reaphirm111128carton_lcr
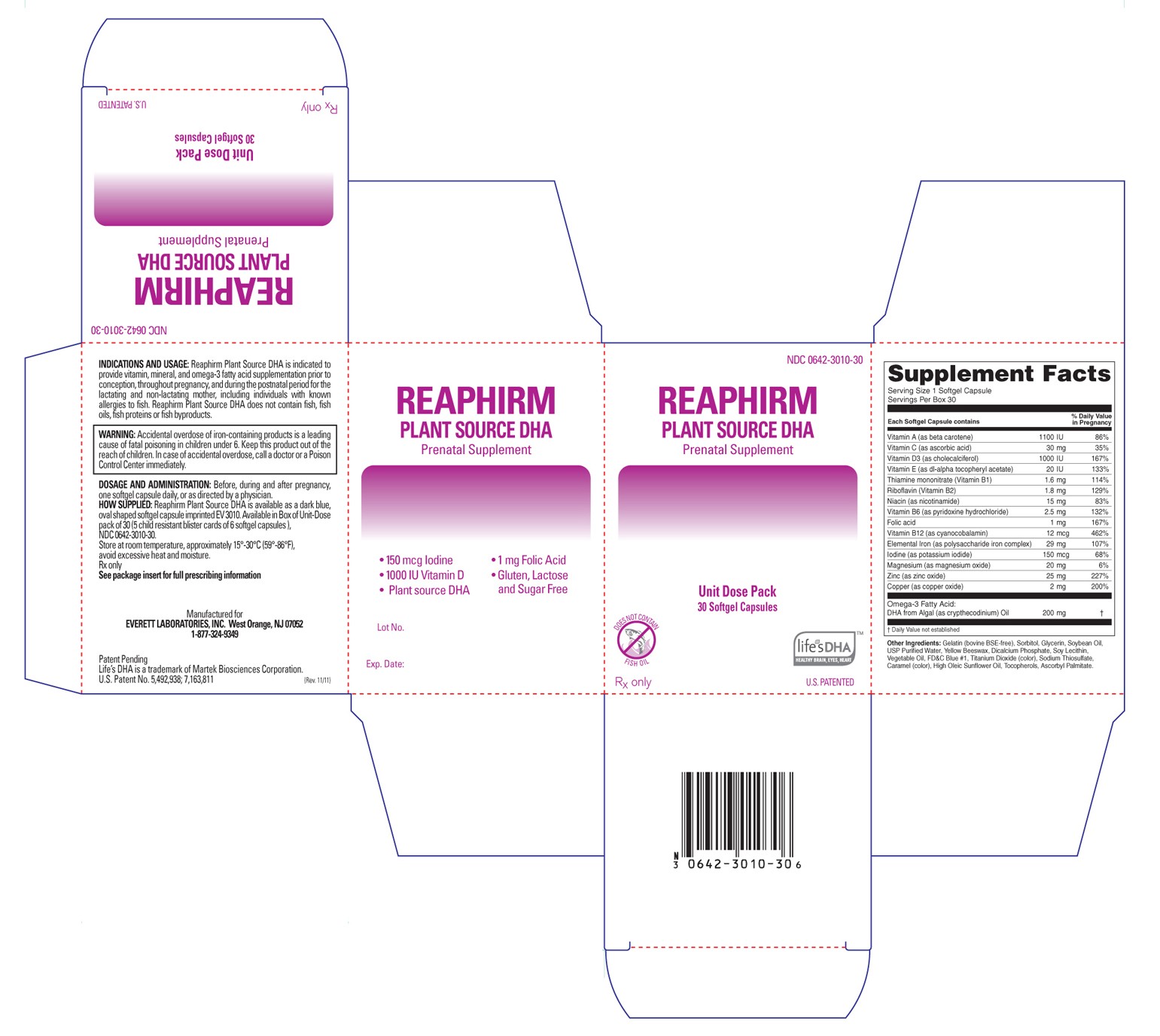
Reaphirm Plant Source DHAPrenatal Supplement CAPSULE, GELATIN COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||