RhoGAM Ultra-Filtered PLUS
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use RhoGAM Ultra-Filtered PLUS (RhoGAM) and MICRhoGAM Ultra-Filtered PLUS (MICRhoGAM) safely and effectively. See full prescribing information for RhoGAM and MICRhoGAM. • Rh(D) Immune Globulin (Human) RhoGAM Ultra-Filtered PLUS (300 μg) (1500 IU) Initial U.S. Approval: 1968• Rh(D) Immune Globulin (Human) MICRhoGAM Ultra-Filtered PLUS (50 μg) (250 IU) Initial U.S. Approval: 1979INDICATIONS AND USAGEFor use in preventing Rh immunization. Pregnancy and other obstetrical conditions in Rh-negative women unless the father or baby are conclusively Rh-negative, e.g. delivery of an Rh-positive baby irrespective of the ABO groups of the mother and baby, any antepartum fetal-maternal hemorrhage (suspected or proven), actual or threatened pregnancy loss at any stage of gestation and ectopic pregnancy. (1.1) Prevention of Rh immunization in any Rh-negative person after incompatible transfusion of Rh-positive blood or blood products (1.2) DOSAGE AND ADMINISTRATION For intramuscular use only, do not administer intravenously. Pregnancy and other obstetrical conditions (2.1) RhoGAM (300 μg) (1500 IU) Postpartum – if the newborn is Rh-positive. Administer within 72 hours of delivery. Antepartum – Prophylaxis at 26 – 28 weeks gestation. At or beyond thirteen weeks gestation: administer within 72 hours when suspected or proven exposure to Rh-positive red blood cells occurs resulting from invasive procedures, abdominal trauma or obstetrical manipulation, ectopic pregnancy, pregnancy termination or threatened termination. Administer every 12 weeks starting from first injection to maintain a level of passively acquired anti-D. If delivery occurs within three weeks after the last antepartum dose, the postpartum dose may be withheld, but a test for fetal-maternal hemorrhage should be performed to determine if exposure to > 15 mL of red blood cells has occurred.MICRhoGAM (50 µg) (250 IU) Administer within 72 hours of actual or threatened termination of pregnancy (spontaneous or induced) up to and including 12 weeks gestation. Transfusion of Rh-incompatible blood or blood products (2.1) Administer within 72 hours.RhoGAM (300 μg) (1500 IU) 2.5 - 15.0 mL Rh-positive red blood cells > 15.0 mL Rh-positive red blood cells (multiple syringes) MICRhoGAM (50 µg) (250 IU) < 2.5 mL Rh-positive red blood cells DOSAGE FORMS AND STRENGTHSRho(D) Immune Globulin (Human) RhoGAM® Ultra-Filtered PLUS - 300 μg (1500 IU) – Prefilled Syringes (3) MICRhoGAM® Ultra-Filtered PLUS - 50 μg (250 IU) – Prefilled Syringes (3) CONTRAINDICATIONS Rh-positive individuals. (4) WARNINGS AND PRECAUTIONS Do not inject intravenously. (5.1) In the case of postpartum use, the product is intended for maternal use only. (5.1) Do not inject the newborn infant. (5.1) May carry a risk of transmitting infectious agents because it is made from human plasma. (5.2) Administer with caution to patients who have had prior severe systemic allergic reactions to human immune globulin. (5.1) Contains a small quantity of immunoglobulin A (IgA), there is a potential risk of hypersensitivity in IgA deficient individuals. (5.1) Patients treated for Rh-incompatible transfusion should be monitored by clinical and laboratory means for signs and symptoms of a hemolytic reaction. (5.1) Side EffectsMost common are: Injection site reactions that include swelling, induration, redness and mild pain or warmth. (6) Systemic reactions that include skin rash, body aches or a slight elevation in temperature. Patients should be observed for at least 20 minutes after administration. Severe systemic allergic reactions are extremely rare. (6) Anti-D formation is rarely reported after proper administration of RhoGAM. (6). To report SUSPECTED ADVERSE REACTIONS, contact: Ortho-Clinical Diagnostics, Inc. at 1-800-421-3311 in the United States. Outside of the United States, the company distributing these products should be contacted. Voluntary reporting of adverse reactions may also be made to the FDA through MedWatch at 1-800-FDA-1088 or on the Internet at www.fda.gov/medwatch. DRUG INTERACTIONS May impair the efficacy of live vaccines such as measles, mumps and varicella. Administration of live vaccines should generally be delayed until 12 weeks after the final dose of immune globulin. If administered within 14 days after administration of a live vaccine, the efficacy of the vaccination may be impaired. (7) The postpartum vaccination of rubella-susceptible women with rubella or MMR vaccine should not be delayed because of the receipt of Rho(D) Immune Globulin (Human). (7) USE IN SPECIFIC POPULATIONS Administer only to Rh-negative patients exposed or potentially exposed to Rh-positive red blood cells to prevent Rh immunization. (8)
FULL PRESCRIBING INFORMATION: CONTENTS*
- 1 RHOGAM ULTRA-FILTERED PLUS INDICATIONS AND USAGE
- 2 RHOGAM ULTRA-FILTERED PLUS DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORMS AND STRENGTHS
- 4 RHOGAM ULTRA-FILTERED PLUS CONTRAINDICATIONS
- 5 WARNINGS AND PRECAUTIONS
- 6 RHOGAM ULTRA-FILTERED PLUS ADVERSE REACTIONS
- 7 DRUG INTERACTIONS
- 8 USE IN SPECIFIC POPULATIONS
- 10 OVERDOSAGE
- 11 RHOGAM ULTRA-FILTERED PLUS DESCRIPTION
- 12 CLINICAL PHARMACOLOGY
- 14 CLINICAL STUDIES
- 15 REFERENCES
- 16 HOW SUPPLIED / STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
- Principal Display Panel - 300 µg Carton
- Principal Display Panel - 50 µg Carton
FULL PRESCRIBING INFORMATION
Rho(D) Immune Globulin (Human)
RhoGAM®
Ultra-Filtered PLUS
(300 μg) (1500 IU)
MICRhoGAM® Ultra-Filtered PLUS (50 μg) (250 IU)
Rx Only
For Intramuscular Injection Only
Prefilled syringes, preservative-free (thimerosal free), latex-free delivery system
1 INDICATIONS AND USAGE
1.1. Pregnancy and other obstetrical conditions
For administration to Rh-negative women not previously sensitized to the Rho(D) factor, unless the father or baby are conclusively Rh-negative.
- Delivery of an Rh-positive baby irrespective of the ABO groups of the mother and baby
- Antepartum prophylaxis at 26 to 28 weeks gestation
- Antepartum fetal-maternal hemorrhage (suspected or proven) as a result of placenta previa, amniocentesis, chorionic villus sampling, percutaneous umbilical blood sampling, other obstetrical manipulative procedure (e.g., version) or abdominal trauma
- Actual or threatened pregnancy loss at any stage of gestation
- Ectopic pregnancy
1.2. Transfusion of Rh-incompatible blood or blood products
- Prevention of Rh immunization in any Rh-negative person after incompatible transfusion of Rh-positive blood or blood products (e.g., red blood cells, platelet concentrates, granulocyte concentrates)
2 DOSAGE AND ADMINISTRATION
For intramuscular use only. Do not inject RhoGAM Ultra-Filtered PLUS (RhoGAM) or MICRhoGAM Ultra-Filtered PLUS (MICRhoGAM) intravenously. In the case of postpartum use, the product is intended for maternal administration. Do not inject the newborn infant. Inject the entire contents of the syringe(s). For single use only. (See WARNINGS AND PRECAUTIONS)
RhoGAM or MICRhoGAM should be administered within 72 hours of delivery or known or suspected exposure to Rh-positive red blood cells. There is little information concerning the effectiveness of Rho(D) Immune Globulin (Human) when given beyond this 72 hour period. In one study, Rho(D) Immune Globulin (Human) provided protection against Rh immunization in about 50% of subjects when given 13 days after exposure to Rh-positive red blood cells.1 Administer every 12 weeks starting from first injection to maintain a level of passively acquired anti-D. If delivery occurs within three weeks after the last antepartum dose, the postpartum dose may be withheld, but a test for fetal-maternal hemorrhage should be performed to determine if exposure to > 15 mL of red blood cells has occurred.2
Parenteral drug products should be inspected visually for particulate matter, discoloration and syringe damage prior to administration. Do not use if particulate matter and / or discoloration are observed. The solution should appear clear or slightly opalescent.
2.1 Indications and Recommended Dosage
| Indication | Dose | Notes |
|---|---|---|
| Postpartum (if the newborn is Rh-positive) | RhoGAM (300 μg) (1500 IU) |
Additional doses of RhoGAM are indicated when the patient has been exposed to > 15 mL of Rh-positive red blood cells. This may be determined by use of qualitative or quantitative tests for fetal-maternal hemorrhage. |
| Administer within 72 hours of delivery. | ||
| Antepartum: | ||
|
If antepartum prophylaxis is indicated, it is essential that the mother receive a postpartum dose if the infant is Rh-positive. | |
| If RhoGAM is administered early in pregnancy (before 26 to 28 weeks), there is an obligation to maintain a level of passively acquired anti-D by administration of RhoGAM at 12-week intervals. | ||
|
MICRhoGAM (50 μg) (250 IU) |
RhoGAM may be administered if MICRhoGAM is not available. |
| Transfusion of Rh-incompatible blood or blood products | Administer within 72 hours of suspected or proven exposure to Rh-positive red blood cells. | |
|
MICRhoGAM (50 μg) (250 IU) |
RhoGAM may be administered if MICRhoGAM is not available. |
|
RhoGAM (300 μg) (1500 IU) |
|
|
RhoGAM (300 μg) (1500 IU) (multiple syringes) |
Additional doses of RhoGAM are indicated when the patient has been exposed to > 15 mL of Rh-positive red blood cells. Administer 20 μg of RhoGAM per mL of Rh-positive red blood cell exposure. Multiple doses may be administered at the same time or at spaced intervals, as long as the total dose is administered within three days of exposure. |
2.2 RhoGAM Administration
Each single dose prefilled syringe of RhoGAM contains 300 μg (1500 IU) of Rho(D) Immune Globulin (Human). This is the dose for the indications associated with pregnancy at or beyond 13 weeks unless there is clinical or laboratory evidence of a fetal-maternal hemorrhage (FMH) in excess of 15 mL of Rh-positive red blood cells.
2.3 MICRhoGAM Administration
Each single dose prefilled syringe of MICRhoGAM contains 50 μg (250 IU) of Rho(D) Immune Globulin (Human). This dose will suppress the immune response to up to 2.5 mL of Rh-positive red blood cells. MICRhoGAM is indicated within 72 hours after termination of pregnancy up to and including 12 weeks gestation. At or beyond 13 weeks gestation, RhoGAM should be administered instead of MICRhoGAM.
2.4 Multiple Dosage
Multiple doses of RhoGAM are required if a FMH exceeds 15 mL, an event that is possible but unlikely prior to the third trimester of pregnancy and is most likely at delivery. Patients known or suspected to be at increased risk of FMH should be tested for FMH by qualitative or quantitative methods.3 In efficacy studies, RhoGAM was shown to suppress Rh immunization in all subjects when given at a dose of ≥ 20 μg per mL of Rh-positive red blood cells.4 Thus, a single dose of RhoGAM will suppress the immune response after exposure to≤ 15 mL of Rh-positive red blood cells. However, in clinical practice, laboratory methods used to determine the amount of exposure (volume of transfusion or FMH) to Rh-positive red blood cells are imprecise.5,6 Therefore, administration of more than 20 μg of RhoGAM per mL of Rh-positive red blood cells should be considered whenever a large FMH or red blood cell exposure is suspected or documented.6 Multiple doses may be administered at the same time or at spaced intervals, as long as the total dose is administered within three days of exposure.7
2.5 Dosage Frequency
To maintain an adequate level of anti-D, RhoGAM should be administered every 12 weeks. The exact timing for the injection is based on 12 week intervals starting from the administration of the first injection. If delivery of the baby does not occur 12 weeks after the administration of the standard antepartum dose (at 26 to 28 weeks), a second dose is recommended to maximize protection antepartum. If delivery occurs within three weeks after the last antepartum dose, the postpartum dose may be withheld, but a test for FMH should be performed to determine if exposure to > 15 mL of red blood cells has occurred.2
2.6 Administration
| Administer injection per standard protocol. |
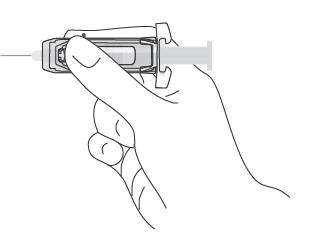
|
| Note: When administering an intramuscular injection, place fingers in contact with syringe barrel through windows in shield to prevent possible premature activation of safety guard. |
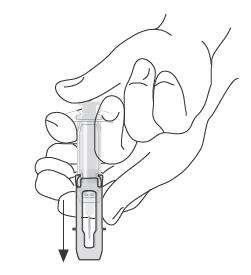
|
Slide safety guard over needle. |
| After injection, use free hand to slide safety guard over needle. An audible "click" indicates proper activation. Keep hands behind needle at all times. Dispose of the syringe in accordance with local regulations. |
3 DOSAGE FORMS AND STRENGTHS
- RhoGAM® Ultra-Filtered PLUS - 300 μg (1500 IU)
The anti-D content of RhoGAM / MICRhoGAM is expressed as μg per dose or as International Units (IU) per dose. The conversion factor is 1 μg = 5 IU.8 – Prefilled Syringes - MICRhoGAM® Ultra-Filtered PLUS - 50 μg (250 IU)

The brands listed are the registered trademarks of their respective owners and are not trademarks of sanofi-aventis U.S. LLC – Prefilled Syringes
4 CONTRAINDICATIONS
The use of RhoGAM and MICRhoGAM is contraindicated in Rh-positive individuals.
5 WARNINGS AND PRECAUTIONS
5.1 Warnings
- For intramuscular use only, do not inject intravenously.
- In the case of postpartum use, the product is intended for maternal administration.
- Do not inject the newborn infant.
- Patients should be observed for at least 20 minutes after administration.
- Administer with caution to patients who have had prior severe systemic allergic reactions to human immune globulin.
- RhoGAM / MICRhoGAM contain a small quantity og IgA. There is a potential risk of hypersensitivity in IgA deficient individuals.
- Patients treated for Rh-incompatible transfusion should be monitored by clinical and laboratory means for signs and symptoms of a hemolytic reaction.
- Store at 2 to 8°C. Do not store frozen.
- Do not use after the expiration date printed on the syringe.
5.2 Use of Plasma Derived Products
RhoGAM and MICRhoGAM are made from human plasma and may carry a risk of transmitting infectious agents, e.g., viruses, and theoretically the Creutzfeldt-Jakob disease (CJD) agent. The risk that such products will transmit an infectious agent has been reduced by screening plasma donors for prior exposure to certain viruses, by testing plasma for the presence of certain current virus infections and by using pathogen removal and inactivation techniques during the manufacturing process. All of the above steps are designed to increase product safety by reducing the risk of pathogen transmission. Despite these measures, such products can still potentially transmit disease. There is also the possibility that unknown infectious agents may be present in such products. All infections thought by a physician possibly to have been transmitted by these products should be reported by the physician or other healthcare provider in the United States to Ortho-Clinical Diagnostics, Inc. at 1-800-421-3311. Outside the United States, the company distributing these products should be contacted. The physician should discuss the risks and benefits of these products with the patient.
5.3 Laboratory Tests
Recovery of anti-D in plasma or serum after injection of RhoGAM or other Rho(D) Immune Globulin (Human) products is highly variable among individuals. Anti-D detection in a patient's plasma is dependent on assay sensitivity and time of sample collection post-injection. Currently there are no requirements or practice standards to test for the presence of anti-D in order to determine adequacy or efficacy of dose following an injection of RhoGAM.
The presence of passively acquired anti-D in the maternal serum may cause a positive antibody screening test. This does not preclude further antepartum or postpartum prophylaxis.
Some babies born to women given Rho(D) Immune Globulin (Human) antepartum have weakly positive direct antiglobulin (Coombs) tests at birth.
Fetal-maternal hemorrhage may cause false blood typing results in the mother. Late in pregnancy or following delivery, there may be sufficient fetal Rh-positive red blood cells in the circulation of the Rh-negative mother to cause a positive antiglobulin test for weak D (Du). In this instance if there is any doubt as to the patient's Rh type, RhoGAM or MICRhoGAM should be administered.9
6 ADVERSE REACTIONS
Adverse events (AE) after administration of RhoGAM and MICRhoGAM are rare.
The most frequently reported AEs are anti-D formation and injection site reactions, such as swelling, induration, redness and mild pain or warmth. Possible systemic reactions are skin rash, body aches or a slight elevation in temperature. Severe systemic allergic reactions are extremely rare. Patients should be observed for at least 20 minutes after administration. There have been no reported fatalities due to anaphylaxis or any other cause related to RhoGAM or MICRhoGAM administration.
As with any Rho(D) Immune Globulin (Human), administration to patients who are Rh-positive or have received Rh-positive red blood cells may result in signs and symptoms of a hemolytic reaction, including fever, back pain, nausea and vomiting, hypo- or hypertension, hemoglobinuria/emia, elevated bilirubin and creatinine and decreased haptoglobin.
RhoGAM and MICRhoGAM contain a small quantity of IgA (less than 15 μg per dose).10 Although high doses of intravenous immune globulin containing IgA at levels of 270-720 μg/mL have been given without incident during treatment of patients with high-titered antibodies to IgA,11 the attending physician must weigh the benefit against the potential risks of hypersensitivity reactions.
7 DRUG INTERACTIONS
Immune globulin preparations including Rho(D) Immune Globulin (Human) may impair the efficacy of live vaccines such as measles, mumps and varicella. Administration of live vaccines should generally be delayed until 12 weeks after the final dose of immune globulin. If an immune globulin is administered within 14 days after administration of a live vaccine, the immune response to the vaccination may be inhibited.12
Because of the importance of rubella immunity among women of childbearing age, the postpartum vaccination of rubella-susceptible women with rubella or MMR vaccine should not be delayed because of the receipt of Rho(D) Immune Globulin (Human) during the last trimester of pregnancy or at delivery. Vaccination should occur immediately after delivery and if possible, testing should be performed after 3 or more months to ensure immunity to rubella and if necessary, to measles.12
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
Animal reproduction studies have not been conducted with RhoGAM or MICRhoGAM. The available evidence suggests that Rho(D) Immune Globulin (Human) does not harm the fetus or affect future pregnancies or the reproduction capacity of the maternal recipient.13,14
8.6 Rh Blood Type
RhoGAM or MICRhoGAM Rho(D) Immune Globulin (Human) should only be administered to Rh-negative patients exposed or potentially exposed to Rh-positive red blood cells to prevent Rh immunization.
10 OVERDOSAGE
Repeated administration or increased dosage in Rh-negative individuals should not cause more severe or more frequent adverse reactions than the normal dose. Patients who receive RhoGAM or MICRhoGAM for Rh-incompatible transfusion should be monitored by clinical and laboratory means due to the risk of a hemolytic reaction.
11 DESCRIPTION
RhoGAM and MICRhoGAM Rho(D) Immune Globulin (Human) are sterile solutions containing immunoglobulin G (IgG) anti-D (anti-Rh) for use in preventing Rh immunization. They are manufactured from human plasma containing anti-D. A single dose of RhoGAM contains sufficient anti-D (300 μg or 1500 IU) to suppress the immune response to up to 15 mL of Rh-positive red blood cells.4,15 A single dose of MICRhoGAM contains sufficient anti-D (50 μg or 250 IU) to suppress the immune response to up to 2.5 mL of Rh-positive red blood cells. The anti-D dose is measured by comparison to the RhoGAM in-house reference standard, the potency of which is established relative to the U.S./World Health Organization/European Pharmacopoeia Standard Anti-D Immunoglobulin Rho(D) Immune Globulin (Human) CBER Lot 4: NIBSC Lot 01/572 (285 IU/ampoule).16
Plasma for RhoGAM is typically sourced from a donor center owned and operated by Ortho-Clinical Diagnostics. All donors are carefully screened by history and laboratory testing to reduce the risk of transmitting blood-borne pathogens from infected donors. Each plasma donation is tested and found to be non-reactive for the presence of hepatitis B surface antigen (HBsAg) and antibodies to hepatitis C (HCV) and human immunodeficiency viruses (HIV) 1 and 2. Additionally, plasma is tested by FDA licensed Nucleic Acid Testing (NAT) for HCV and HIV-1 and the results must be negative. Plasma is also tested by investigational NAT for hepatitis B (HBV) and must be non-reactive. However, the significance of a negative result has not been established. Plasma is tested by in-process NAT procedures for hepatitis A virus (HAV) and parvovirus B19 (B19) in a minipool format. Only plasma that has passed virus screening is used for production. The procedure for B19 detects all three genotypes based upon sequence alignment of known virus isolates. The limit of B19 DNA in the manufacturing pool is set not to exceed 104 IU per mL.
Fractionation of the plasma is performed by a modification of the cold alcohol procedure that has been shown to significantly lower viral titers.10 Following plasma fractionation, a viral clearance filtration step and a viral inactivation step are performed. The viral filtration step removes viruses via a size-exclusion mechanism utilizing a patented Viresolve 180 ultrafiltration membrane with defined pore-size distribution of 12-18 nanometers to remove enveloped and non-enveloped viruses. Following viral filtration, quality control tests (CorrTest and diffusion test) are performed on the Viresolve 180 ultrafiltration membrane to insure filter integrity.17 The viral inactivation step utilizes Triton X-100 and tri-n-butyl phosphate (TNBP) to inactivate enveloped viruses such as HCV, HIV and West Nile Virus (WNV)10,18.
The donor selection process, the fractionation process, the viral filtration step and the viral inactivation process increase product safety by reducing the risk of transmission of enveloped and non-enveloped viruses. Rho(D) Immune Globulin (Human) intended for intramuscular use and prepared by cold alcohol fractionation has not been shown to transmit hepatitis or other infectious diseases.19 There have been no documented cases of infectious disease transmission by RhoGAM or MICRhoGAM.
Laboratory spiking studies10,20 have shown that the cumulative viral removal and inactivation capability of the RhoGAM / MICRhoGAM manufacturing process is as follows:
| Virus | HIV | BVDV | PRV | PPV | EMC | WNV | HAV |
|---|---|---|---|---|---|---|---|
| Units = log10 reduction HIV Human Immunodeficiency Virus, Model for HIV-1 and 2 and Human T-cell Lymphotropic Virus (HTLV) 1 and 2 BVDV Bovine Viral Diarrhea Virus, Model for Hepatitis C Virus PRV Pseudorabies Virus, Model for Herpes Viruses PPV Porcine Parvovirus, Model for Parvovirus B19 EMC Encephalomyocarditis Virus, Model for Hepatitis A Virus WNV West Nile Virus HAV Hepatitis A Virus ND Not Determined N/A Not Applicable |
|||||||
| Lipid Enveloped | Yes | Yes | Yes | No | No | Yes | No |
| Size (nm) | 80-120 | 40-70 | 120-200 | 18-24 | 25-30 | 40-60 | 27-32 |
| Genome | SS-RNA | SS-RNA | DS-DNA | SS-DNA | SS-RNA | SS-RNA | SS-RNA |
| Fractionation | ≥ 7.98 | 7.29 | ≥ 11.74 | 8.30 | ND | ND | ND |
| Viral Filtration | ≥ 5.60 | 5.40 | ≥ 6.20 | 3.30 | 4.16 | ND | ≥ 5.07 |
| Viral Inactivation | ≥ 4.28 | ≥ 4.90 | ≥ 5.58 | N/A | N/A | ≥ 7.05 | N/A |
| Total Viral Reduction | ≥ 17.86 | ≥ 17.59 | ≥ 23.52 | 11.60 | 4.16 | ≥ 7.05 | ≥ 5.07 |
The safety of Rho(D) Immune Globulin (Human) has been further shown in an empirical study of viral marker rates in female blood donors in the United States.21 This study revealed that Rh-negative donors, of whom an estimated 55-60% had received Rho(D) Immune Globulin (Human) for pregnancy-related indications, had prevalence and incidence viral marker rates similar to those of Rh-positive female donors who had not received Rho(D) Immune Globulin (Human).
The final product contains 5 ± 1% IgG, 2.9 mg/mL sodium chloride, 0.01% Polysorbate 80 (non-animal derived) and 15 mg/mL glycine. Small amounts of IgA, typically less than 15 μg per dose, are present.10 The pH range is 6.20 - 7.00 and IgG purity is ≥ 98%. The product contains no added human serum albumin (HSA), no thimerosal or other preservatives and utilizes a latex-free delivery system.
RhoGAM Ultra-Filtered PLUS and MICRhoGAM Ultra-Filtered PLUS are manufactured and distributed by Ortho-Clinical Diagnostics, Inc., Raritan, NJ 08869.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
RhoGAM and MICRhoGAM act by suppressing the immune response of Rh-negative individuals to Rh-positive red blood cells. The mechanism of action is unknown. RhoGAM, MICRhoGAM and other Rho(D) Immune Globulin (Human) products are not effective in altering the course or consequences of Rh immunization once it has occurred.
12.3 Pharmacokinetic Properties
Pharmacokinetic studies after intramuscular injection were performed on sixteen Rh-negative subjects receiving a single dose of (368 μg or 1840 IU) RhoGAM.10 Plasma anti-D levels were monitored for thirteen weeks using a validated Automated Quantitative Hemagglutination method with sensitivity of approximately 1 ng/mL. The following mean pharmacokinetic parameters were obtained from data collected over the first ten weeks of a thirteen-week study:
| Parameter | Mean | SD | Units |
|---|---|---|---|
| Maximum plasma concentration obtained (Cmax) | 54.0 | 13.0 | ng/mL |
| Time to attain Cmax (Tmax) | 4 | days | |
| Elimination half-life (T1/2) | 30.9 | 13.8 | days |
| Volume of distribution (Vd) | 7.3 | 1.5 | liters |
| Clearance (CL) | 150.4 | 53.3 | mL/day |
12.4 Obstetrical Use
The Rh-negative obstetrical patient may be exposed to red blood cells from her Rh-positive fetus during the normal course of pregnancy or after obstetrical procedures or abdominal trauma.
12.5 Use after Rh-Incompatible Transfusion
An Rh-negative individual transfused with one unit of Rh-positive red blood cells has about an 80% likelihood of producing anti-D.4 However, Rh immunization can occur after exposure to < 1 mL of Rh-positive red blood cells. Protection from Rh immunization is accomplished by administering ≥ 20 μg of RhoGAM or MICRhoGAM per mL of Rh-positive red blood cells within 72 hours of transfusion of incompatible red blood cells.13,22
14 CLINICAL STUDIES
Rho(D) Immune Globulin (Human) administered at 28 weeks, as well as within 72 hours of delivery, has been shown to reduce the Rh immunization rate to about 0.1-0.2%.23,24 Clinical studies demonstrated that administration of MICRhoGAM within three hours following pregnancy termination was 100% effective in preventing Rh immunization.25
Multiple studies have been performed that prove the safety and efficacy of RhoGAM in both the obstetrical and post transfusion settings.
Freda, Gorman and colleagues26, 27 studied the efficacy of RhoGAM in the postpartum setting in a randomized, controlled study completed in 1967. The control group received no immunoglobulin therapy after delivery, while the test group received 300 μg of RhoGAM intramuscularly within 72 hours of delivery of an Rh-positive infant. Six months after delivery, the incidence of Rh immunization in the control group was 6.4% (32/499) versus 0.13% (1/781) in the RhoGAM group (p < 0.001).
Pollack et al. performed two randomized, placebo-controlled studies in the post transfusion setting that were designed to establish the dose response relationship of RhoGAM. In the first study,15 178 (176 males, 2 females) Rh-negative volunteers received varying volumes of Rh- positive red cells; 92 subjects then received RhoGAM. A single dose of RhoGAM (1.1 mL @ 267 μg/mL) was shown to suppress anti-D formation after injection of up to 15.1 mL of Rh-positive red cells. In a companion study,4 Pollack administered 500 mL of Rh-positive whole blood to 44 Rh-negative male volunteers. Twenty-two (22) subjects received 20 μg RhoGAM per mL of Rh-positive red cells and 22 received no RhoGAM. None of the RhoGAM-treated subjects developed anti-D; 18/22 control arm subjects developed anti-D (p < 0.0001).
Human clinical studies10 were subsequently performed to prove the efficacy of MICRhoGAM and the low protein (5%) formulations. In the MICRhoGAM study, 81 Rh-negative male volunteers received an initial injection of 2.5 mL Rh-positive red cells, followed by a booster injection (0.1 mL) of red cells at 26 weeks; 40 subjects received an injection of MICRhoGAM after the initial red cell injection. None of the subjects who received MICRhoGAM developed anti-D, both before and after the booster red cell injection. A similar study was performed in 1985 using the low protein formulation of RhoGAM. None of the 30 Rh-negative male volunteers who received RhoGAM after injection of 15 mL of Rh-positive red cells developed anti-D.
15 REFERENCES
-
1 Samson D, Mollison PL. Effect on primary Rh immunization of delayed administration of anti-Rh. Immunol 1975;28:349-57. -
2 Garratty G, ed. Hemolytic disease of the newborn. Arlington, VA: American Association of Blood Banks, 1984:78. -
3 Urbaniak SJ. Statement from the Consensus Conference on Anti-D Prophylaxis, The Royal College of Physicians of Edinburgh & The Royal College of Obstetricians and Gynaecologists, UK. Vox Sang 1998;74:127-28. -
4 Pollack W, Ascari WQ, Crispen JF, O'Connor RR, Ho TY. Studies on Rh prophylaxis. II. Rh immune prophylaxis after transfusion with Rh-positive blood. Transfusion 1971;11:340-44. -
5 Bayliss KM, Kueck DB, Johnson ST, Fueger JT, McFadden PW, Mikulski D, Gottschall JL. Detecting fetomaternal hemorrhage: a comparison of five methods. Transfusion 1991;31:303-7. -
6 Kumpel BM. Quantification of anti-D and fetomaternal hemorrhage by flow cytometry (editorial). Transfusion 2000;40:6-9. -
7 AABB Technical Manual. 15th ed. Bethesda, Maryland: AABB, 2005. -
8 HH, Bowell PJ, Kirkwood TBL. Collaborative study to recalibrate the International Reference Preparation of anti-D immunoglobulin. J Clin Pathol 1980;33:249-53. -
9 ACOG practice bulletin. Prevention of Rh D alloimmunization. Number 4, May 1999 (replaces educational bulletin Number 147, October 1990). Clinical management guidelines for obstetrician-gynecologists. American College of Obstetrics and Gynecology. Int J Gynaecol Obstet. 1999; 66(1):63-70. -
10 Data on file at Ortho-Clinical Diagnostics, Inc. -
11 Cunningham-Rundles C, Zhuo Z, Mankarious S, Courter S. Long-term use of IgA-depleted intravenous immunoglobulin in immunodeficient subjects with anti-IgA antibodies. J Clin Immunol 1993;13:272-78. -
12 Centers for Disease Control and Prevention. General recommendations on immunization: recommendations of the Advisory Committee on Immunization Practices and the American Academy of Family Physicians. MMWR 2002;51 (No. RR-2):6-7. -
13 Zipursky A, Israels LG. The pathogenesis and prevention of Rh immunization. Can Med Assoc J 1967;97:1245-56. -
14 Thornton JG, Page C, Foote G, Arthur GR, Tovey LAD, Scott JS. Efficacy and long term effects of antenatal prophylaxis with anti-D immunoglobulin. Brit Med J 1989;298:1671-73. -
15 Pollack W, Ascari WQ, Kochesky RJ, O'Connor RR, Ho TY, Tripodi D. Studies on Rh prophylaxis. I. Relationship between doses of anti-Rh and size of antigenic stimulus. Transfusion 1971;11:333-39. -
16 Thorpe SJ, Sands D, Fox B, Behr-Gross ME, Schaffner G, Yu MW. A global standard for anti-D immunoglobulin: international collaborative study to evaluate a candidate preparation. Vox Sang 2003;85:313-21. -
17 Phillips MW, DiLeo AJ. A Validatible Porosimetric Technique for verifying the integrity of virus-retentive membranes. Biologicals 1996;24:243-53. -
18 Horowitz B, Wiebe ME, Lippin A, Stryker MH. Inactivation of viruses in labile blood derivatives. I. Disruption of lipid-enveloped viruses by tri (n-butyl) phosphate detergent combinations. Transfusion 1985; 25(6):516-22. -
19 Tabor E. The epidemiology of virus transmission by plasma derivatives: clinical studies verifying the lack of transmission of hepatitis B and C viruses and HIV type 1. Transfusion 1999;39:1160-68. -
20 Van Holten RW, Ciavarella D, Oulundsen G, Harmon F, Riester S. Incorporation of an additional viral-clearance step into a human immunoglobulin manufacturing process. Vox Sang 2002;83:227-33. -
21 Watanabe KK, Busch MP, Schreiber GB, Zuck TF. Evaluation of the safety of Rh Immunoglobulin by monitoring viral markers among Rh-negative female blood donors. Vox Sang 2000;8:1-6. -
22 Crispen J. Immunosuppression of small quantities of Rh-positive blood with MICRhoGAM in Rh-negative male volunteers. In: Proceedings of a symposium on Rh antibody mediated immunosuppression. Raritan, NJ: Ortho Research Institute of Medical Sciences, 1975:51-54. -
23 Bowman JM, Chown B, Lewis M, Pollock JM. Rh isoimmunization during pregnancy: antenatal prophylaxis. Can Med Assoc J 1978;118:623-27. -
24 Bowman JM, Pollock JM. Antenatal prophylaxis of Rh isoimmunization: 28-weeks' gestation service program. Can Med Assoc J 1978;118:627-30. -
25 Stewart FH, Burnhill MS, Bozorgi N. Reduced dose of Rh immunoglobulin following first trimester pregnancy termination. Obstet Gynecol 1978;51:318-22. -
26 Pollack W, Gorman JG, Freda VJ, Ascari WQ, Allen AE, Baker WJ. Results of clinical trials of RhoGAM in women. Transfusion 1968;8:151-53. -
27 Freda VJ, Gorman JG, Pollack W, Bowe E. Prevention of Rh hemolytic disease – ten years' clinical experience with Rh immune globulin. New Engl J Med 1975; 292:1014-16.
16 HOW SUPPLIED / STORAGE AND HANDLING
16.1 RhoGAM Ultra-Filtered PLUS
- 1 Injection, Solution (Syringe) in 1 Pouch
NDC 0562-7805-00
16.2 RhoGAM Ultra-Filtered PLUS package sizes
- 1 prefilled single-dose syringe of RhoGAM (Product Code 780501)
NDC 0562-7805-01
1 package insert, 1 control form, 1 patient identification card - 5 prefilled single-dose syringes of RhoGAM (Product Code 780505)
NDC 0562-7805-05
5 package inserts, 5 control forms, 5 patient identification cards - 25 prefilled single-dose syringes of RhoGAM (Product Code 780525)
NDC 0562-7805-25
25 package inserts, 25 control forms, 25 patient identification cards
16.3 MICRhoGAM Ultra-Filtered PLUS
- 1 Injection, Solution (Syringe) in 1 Pouch
NDC 0562-7806-00
16.4 MICRhoGAM Ultra-Filtered PLUS package sizes
- 1 prefilled single-dose syringe of MICRhoGAM (Product Code 780601)
NDC 0562-7806-01
1 package insert, 1 control form, 1 patient identification card - 5 prefilled single-dose syringes of MICRhoGAM (Product Code 780605)
NDC 0562-7806-05
5 package inserts, 5 control forms, 5 patient identification cards - 25 prefilled single-dose syringes of MICRhoGAM (Product Code 780625)
NDC 0562-7806-25
25 package inserts, 25 control forms, 25 patient identification cards
Store at 2 to 8°C. Do not store frozen. Do not use after the expiration date printed on the syringe.
17 PATIENT COUNSELING INFORMATION
As with all immune globulin preparations, the physician should discuss the risks and benefits with the patient. The most common adverse reactions are local reactions including swelling, induration, redness and mild pain at the site of injection, and a small number of patients have noted a slight elevation in temperature.
Systemic reactions to RhoGAM or MICRhoGAM are extremely rare, however allergic responses to RhoGAM or MICRhoGAM may occur. Patients should be observed for at least 20 minutes after administration. Patients should be informed of the early signs of hypersensitivity reactions including hives, generalized urticaria, tightness of the chest, wheezing, hypotension and anaphylaxis.
The physician should provide the patient with a completed RhoGAM Patient Identification Card and advise the patient to retain the card and present it to other health care providers when appropriate.
| Section | Revision |
|---|---|
| HIGHLIGHTS OF PRESCRIBING INFORMATION - DOSAGE AND ADMINISTRATION | Corrected MICRhoGAM from 50 mg to read 50 µg. |
| HOW SUPPLIED / STORAGE AND HANDLING | Updated Section 16 for NDC barcode information. |
| Updated corporate logo. |
US LICENSE 1236
Ortho Clinical Diagnostics
PART OF THE Johnson & Johnson FAMILY OF COMPANIES
1001 US Highway 202, Raritan, NJ 08869 USA
©Ortho-Clinical Diagnostics, Inc. 2010
Printed in U.S.A.
Made by methods of
U.S. Pat. 6,096,872
U.S. Pat. 7,655,233
Revised July 2012
631203006



PATIENT IDENTIFICATION CARD
Name _____________________________________________________________________________________________________________
Address _____________________________________________________________________________________________________________
I AM Rh NEGATIVE. I have received a protective injection of RhoGAM® or MICRhoGAM® Rho(D) Immune Globulin (Human) Ultra-Filtered PLUS.
IMPORTANT: Anti-Rh antibody (also called anti-D) will be present in my blood for several weeks after the injection, and may be detectable by laboratory testing. The presence of this passive anti-Rh antibody does not disqualify me from receiving additional injections of RhoGAM or MICRhoGAM as indicated and prescribed by my physician.
©Ortho-Clinical Diagnostics, Inc. 2010
Rho(D) Immune Globulin (Human)
RhoGAM® and MICRhoGAM®
Ultra-Filtered PLUS
This 3-part form contains:
- Directions for Use
- Patient Control Form
- Patient Identification Card
US LICENSE 1236
Ortho Clinical Diagnostics
PART OF THE Johnson & Johnson FAMILY OF COMPANIES
1001 US Highway 202, Raritan, NJ 08869 USA
631203006
Date of Injection of RhoGAM or MICRhoGAM

Principal Display Panel - 300 µg Carton
Rho(D) Immune Globulin (Human)
RhoGAM®
Ultra-Filtered PLUS – 300 µg Dose (1500 IU*)
Thimerosal-Free
*International Units
Do not store frozen
See Directions for Use
Package Contains:
- 1 prefilled syringe containing a
single dose of RhoGAM - 1 control form
- 1 package insert
- 1 patient identification card
1 single dose
Active ingredient:
Anti-D Rho Immune Globulin (300 µg), the potency of
which is determined relative to the US/WHO/EP Standard
Anti-D Immunoglobulin Rho(D) Immune Globulin (Human)
CBER Lot 4: NIBSC Lot 01/572 (285 IU/ampoule)
Inactive ingredients:
2.9 mg/mL sodium chloride
0.01% polysorbate 80
15 mg/mL glycine
US LICENSE 1236
Ortho Clinical Diagnostics
PART OF THE Johnson & Johnson FAMILY OF COMPANIES
1001 US Highway 202, Raritan, NJ 08869 USA
REF
780501

Principal Display Panel - 50 µg Carton
Rho(D) Immune Globulin (Human)
MICRhoGAM®
Ultra-Filtered PLUS – 50 µg Dose (250 IU*)
Thimerosal-Free
*International Units
Do not store frozen
See Directions for Use
Package Contains:
- 1 prefilled syringe containing a
single dose of MICRhoGAM - 1 control form
- 1 package insert
- 1 patient identification card
1 single dose
Active ingredient:
Anti-D Rho Immune Globulin (50 µg), the potency of
which is determined relative to the US/WHO/EP Standard
Anti-D Immunoglobulin Rho(D) Immune Globulin (Human)
CBER Lot 4: NIBSC Lot 01/572 (285 IU/ampoule)
Inactive ingredients:
2.9 mg/mL sodium chloride
0.01% polysorbate 80
15 mg/mL glycine
US LICENSE 1236
Ortho Clinical Diagnostics
PART OF THE Johnson & Johnson FAMILY OF COMPANIES
1001 US Highway 202, Raritan, NJ 08869 USA
REF
780601

RhoGAM Ultra-Filtered PLUSHUMAN RHO(D) IMMUNE GLOBULIN INJECTION, SOLUTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
MICRhoGAM Ultra-Filtered PLUSHUMAN RHO(D) IMMUNE GLOBULIN INJECTION, SOLUTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||