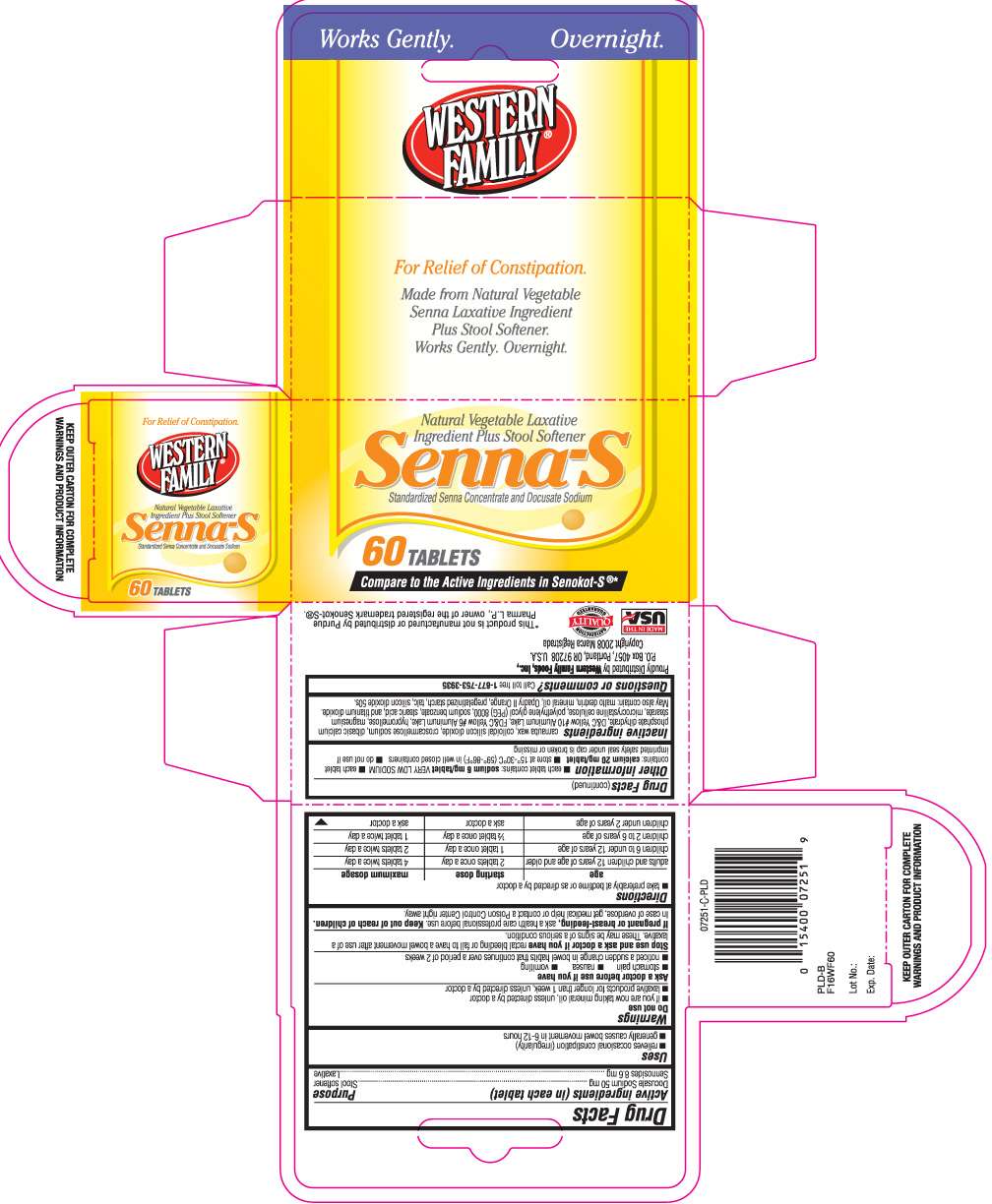
Senna- S
DRUG FACTS
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredients (in each tablet)
- Purpose
- Senna- S Uses
- Do not use
- Ask a doctor before use if you have
- Stop use and ask a doctor if you have
- If pregnant or breast-feeding,
- Keep out of reach of children.
- Directions
- Other Information
- Inactive ingredients
- Questions or comments?
- Principal Display Panel
- Product Label
FULL PRESCRIBING INFORMATION
Active ingredients (in each tablet)
Docusate sodium 50 mg
Sennosides 8.6 mg
Purpose
Stool softener
Laxative
Senna- S Uses
- relieves occasional constipation (irregularity)
- generally produces a bowel movement in 6-12 hours
Do not use
- if you are now taking mineral oil, unless directed by a doctor
- laxative products for longer than 1 week, unless directed by a doctor
Ask a doctor before use if you have
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that continues over a period of 2 weeks
Stop use and ask a doctor if you have
rectal bleeding or fail to have a bowel movement after use of a laxative. These maybe signs of a serious condition.
If pregnant or breast-feeding,
ask a health care professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- take preferably at bedtime or as directed by a doctor
| age | starting dose | maximum dosage |
| adults and children 12 years of age or older | 2 tablets once a day |
4 tablets twice a day |
| children 6 to under 12 years of age | 1 tablet once a day |
2 tablets twice a day |
| children 2 to under 6 years | 1/2 tablet once a day |
1 tablet twice a day |
| children under 2 years | ask a doctor | ask a doctor |
Other Information
- each tablet contains: sodium 6 mg/ tablet VERY LOW SODIUM
- each tablet contains: Calcium 20 mg/ tablet
- store at 15o- 30o C (59o- 86o F) in well closed containers
- do not use if imprinted safety seal under cap is broken or missing.
Inactive ingredients
carnauba wax, colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate dihydrate, D&C yellow #10 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol PEG 8000, sodium benzoate, stearic acid and titanium dioxide
May also contain: maltodextrin, mineral oil, opadry II orange, pregelatinized starch, talc, silicon dioxide 50s
Questions or comments?
Call toll free 1-877-753-3935
Principal Display Panel
Compare to the active ingredients in Senokot- S® *
Senna- S
Laxative plus stool softener
Standardized senna concentrate and Docusate sodium
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION
*This product is not manufactured or distributed by Purdue Pharma L.P., owner of the registered trademark SENOKOT-S®
Product Label
Senna- Sdocusate sodium and sennosides TABLET
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||