Senna Plus
SENNA PLUS
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient
- Purpose
- Senna Plus Uses
- Warnings
- Ask a doctor or pharmacist before use if you have
- Stop use and ask a doctor if
- If pregnant or breast-feeding
- Keep out of the reach of children.
- Directions
- Inactive ingredients
- Package/Label Principal Display Panel
FULL PRESCRIBING INFORMATION
Active ingredient
Sennosides form Senna Concentrate 8.6mg
Sodium Dioctylsulfosuccinate 50 mg
Purpose
Senna Plus Uses
For the short term relief of constipation, produce bowel movement in 6 to 12 hours.
Warnings
Do not use for longer than one week, when abdominal pain, nausea or vomiting are present.
Ask a doctor or pharmacist before use if you have
noticed a sudden change in bowel habits that lasts over a period of two weeks
Stop use and ask a doctor if
you have rectal bleeding, you fail to have a bowel movement after use of this product, may indicate a serious condition. You may report side effects to FDA at 1-800-FDA-1088.
If pregnant or breast-feeding
ask a health professional before use.
Keep out of the reach of children.
In case of overdose, get medical help or contact a Poision Control Center right away.
Directions
Take preferably at bedtime or as directed by a doctor, if you do not have a comfortable bowel movement by the second day, increase dose by one tablet (do not exceed the maximum dosage) or decrease dose until you are comfortable
| age | starting dose | maximum dose |
| adults and children over 12 years | 2 tablets once a day | 4 tablets twice a day |
| children 6 to under 12 years | 1 tablet once a day | 2 tablets twice a day |
| children 2 to under 6 years | 1/2 tablet once a day | 1 tablet twice a day |
| children under 2 years | ask a doctor |
Inactive ingredients
Croscarmellose sodium, Dicalcium Phosphate, FD&C Yellow #5,Lake, FD&C Yellow #6 Lake, Hydroxypropylmethylcellulose, Magensium Silicate, Magnesium Stearate, Microcrystalline Cellulose, Mineral Oil, Polyethylene Glycol, Sodium Benzoate, Sodium Lauryl Sulfate, Starch, Stearic cid and Titanium Dioxide.
Store at controlled room temperature 20°C to 25°C (68°-77°F), excursions permitted to 15°C to 30°C (598°-86°F).
Mfg by
Contract Pharmacal Corp
Hauppauge, NY 11788 USA
www.CPCHealth.com
Package/Label Principal Display Panel
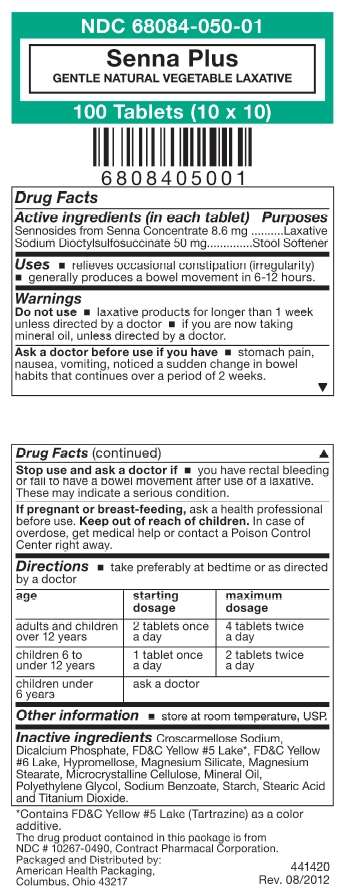
Senna PlusSenna Plus TABLET
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||