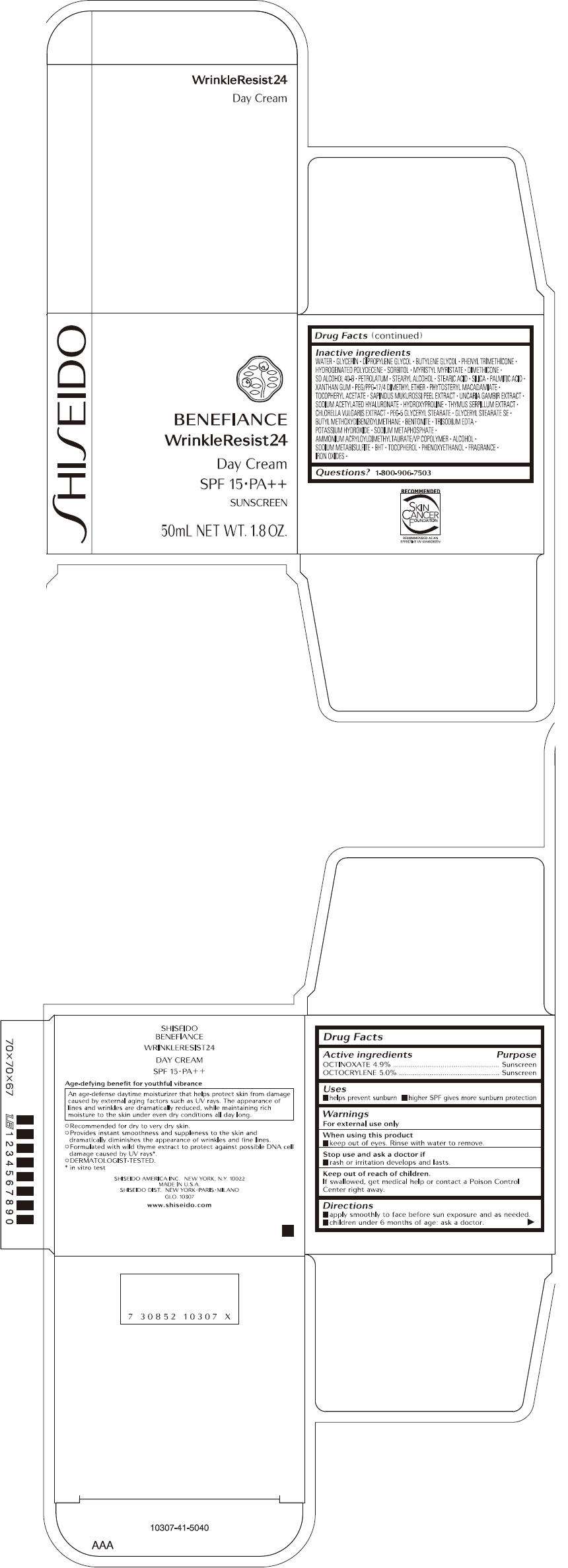
SHISEIDO BENEFIANCE WRINKLERESIST24 DAY
SHISEIDO BENEFIANCE WRINKLERESIST24 DAY CREAM
FULL PRESCRIBING INFORMATION: CONTENTS*
- SHISEIDO BENEFIANCE WRINKLERESIST24 DAY Uses
- Warnings
- Directions
- Inactive Ingredients
- Questions?
- PRINCIPAL DISPLAY PANEL - 50mL Carton
FULL PRESCRIBING INFORMATION
Drug Facts
Active ingredient
Purpose
| Active ingredients | Purpose |
|---|---|
| OCTINOXATE 4.9% | Sunscreen |
| OCTOCRYLENE 5.0% | Sunscreen |
SHISEIDO BENEFIANCE WRINKLERESIST24 DAY Uses
- helps prevent sunburn
- higher SPF gives more sunburn protection
Warnings
For external use only
When using this product
- keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if
- rash or irritation develops and lasts.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- apply smoothly to face before sun exposure and as needed.
- children under 6 months of age: ask a doctor.
Inactive Ingredients
WATER, GLYCERIN, DIPROPYLENE GLYCOL, BUTYLENE GLYCOL, PHENYL TRIMETHICONE, HYDROGENATED POLYDECENE, SORBITOL, MYRISTYL MYRISTATE, DIMETHICONE, SD ALCOHOL 40-B, PETROLATUM, STEARYL ALCOHOL, STEARIC ACID, SILICA, PALMITIC ACID, XANTHAN GUM, PEG/PPG-17/4 DIMETHYL ETHER, PHYTOSTERYL MACADAMIATE, TOCOPHERYL ACETATE, SAPINDUS MUKUROSSI PEEL EXTRACT, UNCARIA GAMBIR EXTRACT, SODIUM ACETYLATED HYALURONATE, HYDROXYPROLINE , THYMUS SERPILLUM EXTRACT, CHLORELLA VULGARIS EXTRACT, PEG-5 GLYCERYL STEARATE, GLYCERYL STEARATE SE, BUTYL METHOXYDIBENZOYLMETHANE, BENTONITE, TRISODIUM EDTA, POTASSIUM HYDROXIDE, SODIUM METAPHOSPHATE, AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER, ALCOHOL, SODIUM METABISULFITE, BHT, TOCOPHEROL, PHENOXYETHANOL, FRAGRANCE, IRON OXIDES
Questions?
1-800-906-7503
PRINCIPAL DISPLAY PANEL - 50mL Carton
SHISEIDO
BENEFIANCE
WrinkleResist24
Day Cream
SPF 15 • PA++
SUNSCREEN
50mL NET WT. 1.8 OZ.
SHISEIDO BENEFIANCE WRINKLERESIST24 DAYOctinoxate and Octocrylene CREAM
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||